Advanced Copper-Catalyzed Synthesis of 2,3,4-Trisubstituted Pyrroles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly polysubstituted pyrroles, which serve as critical building blocks in numerous bioactive natural products and synthetic drugs. Patent CN105884670B introduces a groundbreaking advancement in this domain by disclosing a novel class of 2,3,4-trisubstituted pyrrole compounds and an efficient synthesis method thereof. This technology leverages a copper-catalyzed cyclization reaction between isonitriles and allenes under remarkably mild conditions, addressing long-standing challenges in regioselectivity and functional group tolerance. The structural versatility of these compounds, characterized by an aryl sulfone functional group at the 3-position, opens new avenues for medicinal chemistry applications, particularly in the development of antitumor agents. As a leading entity in the chemical sector, we recognize the immense value of this intellectual property in streamlining the production of high-value intermediates.
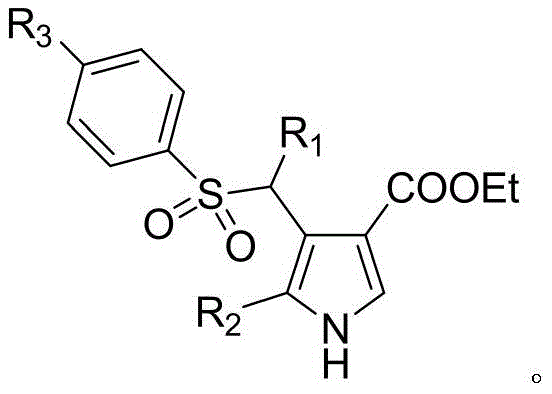
The core innovation lies in the ability to access these complex architectures through a straightforward catalytic cycle that avoids the extreme thermal or pressure conditions often associated with traditional heterocycle formation. By utilizing readily available starting materials and a cost-effective copper catalyst system, this patent provides a scalable route that aligns perfectly with the demands of modern green chemistry and industrial efficiency. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is crucial for securing a reliable supply chain of high-purity pharmaceutical intermediates. The following analysis delves deep into the mechanistic advantages and commercial implications of adopting this superior synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrole ring has relied on classical methodologies such as the Paal-Knorr, Knorr, Hantzsch, Van-Leusen, and Barton-Zard syntheses. While these methods have served the industry well for decades, they often suffer from significant limitations when applied to the synthesis of highly substituted or functionally complex pyrroles. Traditional routes frequently necessitate harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can lead to the decomposition of sensitive functional groups and result in poor yields. Furthermore, achieving specific substitution patterns, particularly trisubstituted variants with precise regiocontrol, remains a formidable challenge using these legacy techniques. The reliance on stoichiometric reagents and the generation of substantial waste streams further exacerbate the environmental and economic burdens associated with conventional pyrrole manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN105884670B represents a paradigm shift towards efficiency and selectivity. This novel approach utilizes a copper-catalyzed cyclization of allenes and isonitriles, enabling the formation of 2,3,4-trisubstituted pyrroles under exceptionally mild conditions. The reaction proceeds smoothly at a moderate temperature of 50°C in a mixed solvent system of 1,4-dioxane and water, eliminating the need for energy-intensive heating or high-pressure equipment. This温和 (mild) protocol not only enhances safety profiles for large-scale operations but also significantly broadens the scope of compatible substrates. The ability to introduce diverse electron-donating alkyl or aryl groups at the R1 and R2 positions, along with alkyl or halogen substituents at the R3 position, demonstrates the method's versatility.
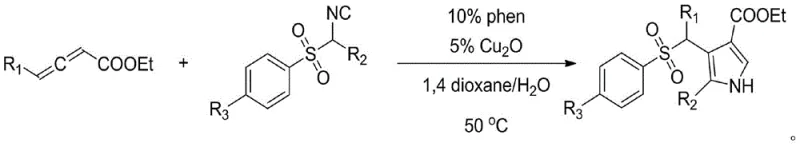
By employing a catalytic system comprising cuprous oxide (Cu2O) and 1,10-phenanthroline, the process achieves high atom economy and reduces the reliance on expensive noble metal catalysts. This transition from stoichiometric to catalytic processes is a key driver for cost reduction in pharmaceutical intermediate manufacturing. The streamlined workflow, which typically completes within one hour, allows for rapid iteration in drug discovery campaigns and facilitates the swift scale-up required for commercial production. For supply chain managers, this translates to shorter lead times and a more resilient sourcing strategy for critical heterocyclic building blocks.
Mechanistic Insights into Copper-Catalyzed Cyclization
The success of this synthesis hinges on the unique reactivity of the copper catalyst in mediating the interaction between the allene and isonitrile moieties. Mechanistically, the reaction is believed to proceed through a copper-mediated activation of the isonitrile, followed by a nucleophilic attack on the allene system to form a metallacycle intermediate. This intermediate subsequently undergoes cyclization and proton transfer to yield the final trisubstituted pyrrole scaffold. The presence of the 1,10-phenanthroline ligand is critical, as it stabilizes the copper species and modulates its electronic properties to favor the desired cyclization pathway over competing side reactions. This precise control over the catalytic cycle ensures high selectivity for the 2,3,4-substitution pattern, minimizing the formation of regioisomers that are difficult to separate.
From an impurity control perspective, the mild reaction conditions play a pivotal role in maintaining product integrity. High-temperature processes often promote polymerization of allenes or hydrolysis of sensitive ester groups, leading to complex impurity profiles that require extensive purification. By operating at 50°C, this method effectively suppresses thermal degradation pathways, resulting in cleaner crude reaction mixtures. The use of a biphasic or mixed solvent system involving water further aids in solubilizing polar byproducts while keeping the organic product in the organic phase, simplifying downstream processing. For quality assurance teams, this means that achieving stringent purity specifications becomes more attainable, reducing the risk of batch failures and ensuring consistent quality for downstream API synthesis.
How to Synthesize Ethyl 4-(tosylmethyl)-1H-pyrrole-3-carboxylate Efficiently
The practical implementation of this technology is straightforward and adaptable to standard laboratory and pilot plant equipment. The general procedure involves charging a pressure-resistant vessel with the requisite benzofuran derivative or allene precursor, the isonitrile component, and the catalytic system. Specifically, the protocol calls for the use of 5 mol% cuprous oxide and 10 mol% 1,10-phenanthroline, which are inexpensive and commercially available reagents. The reaction mixture is then heated to 50°C in a 1,4-dioxane and water solvent blend. Monitoring via thin-layer chromatography (TLC) typically indicates complete conversion within one hour, after which standard workup procedures involving column chromatography yield the pure product. Detailed standardized synthesis steps are provided in the guide below.
- Charge a pressure-resistant tube with benzofuran derivatives, allenes, cuprous oxide catalyst, and 1,10-phenanthroline ligand.
- Add the solvent system consisting of 1,4-dioxane and water, then heat the mixture to 50°C for approximately 1 hour.
- Monitor reaction completion via TLC, then purify the crude product using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this copper-catalyzed synthesis route offers profound strategic advantages for procurement and supply chain operations, primarily driven by the simplification of the manufacturing process and the accessibility of raw materials. The shift away from complex multi-step sequences towards a direct cyclization reaction drastically reduces the number of unit operations required, thereby lowering capital expenditure and operational overheads. Furthermore, the elimination of harsh reagents and extreme conditions minimizes the need for specialized corrosion-resistant equipment, allowing for production in standard stainless steel reactors. This flexibility enhances supply chain reliability by enabling manufacturing across a broader range of facilities, reducing the risk of bottlenecks associated with specialized infrastructure.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, stemming largely from the use of base metal catalysis instead of precious metals like palladium or rhodium. Copper salts are orders of magnitude cheaper and more abundant, leading to significant savings in catalyst costs. Additionally, the high yields reported (ranging from 43% to 84% across various substrates) and the short reaction time of one hour maximize throughput and reactor utilization rates. The simplified purification process, facilitated by the clean reaction profile, reduces solvent consumption and waste disposal costs, contributing to a lower overall cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including various allenes and isonitriles, are commodity chemicals with robust global supply chains. Unlike exotic reagents that may be subject to geopolitical restrictions or limited availability, these precursors can be sourced from multiple vendors, ensuring continuity of supply. The mild reaction conditions also reduce the risk of unplanned shutdowns due to safety incidents or equipment failure, providing a more predictable production schedule. For procurement managers, this reliability translates into the ability to negotiate better terms and secure long-term contracts with confidence, knowing that the underlying technology is stable and scalable.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is inherently safer due to the absence of high-pressure or high-temperature hazards. The exothermic nature of the reaction is manageable at 50°C, reducing the cooling load and energy requirements for large-scale batches. Moreover, the use of water as a co-solvent aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with increasingly stringent environmental regulations mitigates regulatory risks and enhances the sustainability profile of the supply chain, a key consideration for modern pharmaceutical companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new drug development programs.
Q: What are the key advantages of this copper-catalyzed method over traditional pyrrole synthesis?
A: Unlike traditional methods such as Paal-Knorr or Hantzsch synthesis which often require harsh conditions, this novel approach utilizes mild temperatures (50°C) and avoids high pressure, significantly improving safety and scalability for industrial production.
Q: Does this synthesis method support diverse substituent groups for library generation?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing a series of compounds with varying alkyl and aryl groups at the R1, R2, and R3 positions, making it ideal for establishing diverse compound libraries.
Q: What is the biological potential of the synthesized pyrrole derivatives?
A: Specific derivatives, such as Compound 16 containing a 4-fluorophenylsulfonyl group, have demonstrated inhibitory activity against human leukemia cells (HL-60), indicating strong potential for development into antitumor therapeutic agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4-Trisubstituted Pyrroles Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this innovative patent technology into commercial reality. Our team of experienced chemists is adept at optimizing copper-catalyzed pathways, ensuring that the transition from laboratory scale to industrial production is seamless and efficient. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our state-of-the-art rigorous QC labs enforce stringent purity specifications, ensuring that every batch of 2,3,4-trisubstituted pyrroles meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis method for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial viability.
