Scalable Iron-Mediated Synthesis of Beta-Hydroxycarbonyl Compounds for Advanced Pharmaceutical Manufacturing
Introduction to Advanced Iron-Mediated Synthetic Methodologies
The landscape of organic synthesis for pharmaceutical intermediates is undergoing a significant transformation driven by the need for sustainable, cost-effective, and robust manufacturing processes. A pivotal development in this field is detailed in Chinese Patent CN110105212B, which discloses a novel method for preparing beta-hydroxycarbonyl compounds using inexpensive commercial iron powder. This technology represents a paradigm shift from traditional organometallic reagents, offering a pathway that combines economic efficiency with exceptional chemical versatility. For R&D directors and procurement strategists in the global fine chemical sector, this innovation addresses critical pain points regarding raw material costs and environmental compliance. The patent outlines a protocol where metallic iron, mediated by iodine or ferrous iodide, facilitates the coupling of alpha-halo carbonyl compounds with aldehydes or ketones. This approach not only simplifies the supply chain by utilizing ubiquitous base metals but also ensures high purity profiles essential for downstream API synthesis. As we delve deeper into the technical specifics, it becomes evident that this methodology is not merely an academic curiosity but a viable industrial solution for producing complex molecular scaffolds found in medicines, agrochemicals, and fragrances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-hydroxycarbonyl structures has relied heavily on the classical Reformatsky reaction, which typically employs activated zinc dust or magnesium turnings. While effective, these traditional methods suffer from inherent logistical and chemical drawbacks that complicate large-scale manufacturing. Zinc dust, for instance, often requires rigorous activation procedures to remove surface oxides, leading to batch-to-batch variability and inconsistent reaction initiation. Furthermore, organozinc and organomagnesium reagents are notoriously sensitive to moisture and oxygen, necessitating strictly anhydrous conditions and inert atmosphere handling that drive up operational expenditures. From a safety and environmental perspective, the disposal of heavy metal waste streams associated with zinc and magnesium poses significant regulatory challenges. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming to scale production efficiently. The sensitivity of these reagents also limits the scope of compatible functional groups, often restricting the synthesis of molecules containing sensitive moieties like nitriles or certain halogens.
The Novel Approach
In stark contrast, the methodology presented in CN110105212B leverages the unique reactivity of zero-valent iron in the presence of a catalytic amount of iodine. This system operates under much milder conditions, typically at 60°C in acetonitrile, eliminating the need for cryogenic temperatures or ultra-dry solvents. The use of commercial iron powder, which is orders of magnitude cheaper than activated zinc or specialized organometallics, fundamentally alters the cost structure of the synthesis. More importantly, the iron-iodine system exhibits remarkable functional group tolerance, successfully coupling substrates with electron-withdrawing groups such as chloro, bromo, cyano, and trifluoromethyl substituents without side reactions. This broad compatibility allows for the direct synthesis of highly functionalized intermediates that would otherwise require multi-step protection-deprotection sequences. The reaction mechanism likely involves the in situ generation of an organoiron species that is less basic and more nucleophilic than its zinc counterpart, thereby minimizing elimination byproducts. This robustness makes the process ideal for the commercial scale-up of complex pharmaceutical additives and agrochemical intermediates where structural integrity is paramount.
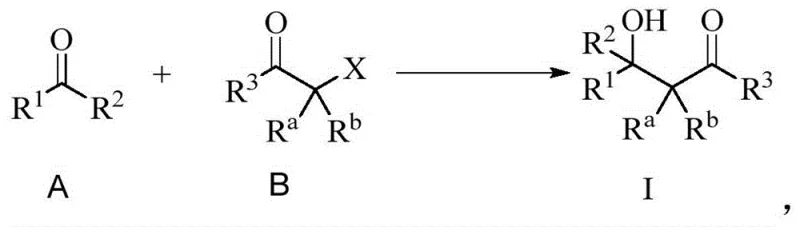
Mechanistic Insights into Fe/I2-Catalyzed Reformatsky-Type Reaction
The core of this technological breakthrough lies in the synergistic interaction between metallic iron and the iodine promoter. Mechanistically, the iodine serves to activate the surface of the iron powder, facilitating the oxidative insertion of iron into the carbon-halogen bond of the alpha-halo carbonyl substrate. This generates a reactive organoiron intermediate, which subsequently undergoes nucleophilic addition to the carbonyl group of the aldehyde or ketone. Unlike zinc-mediated processes where the formation of the enolate can be sluggish or incomplete, the iron-iodine system ensures rapid and quantitative generation of the nucleophile. The presence of FeI2 as a potential catalytic species further enhances the Lewis acidity of the system, activating the electrophilic carbonyl partner and lowering the activation energy of the addition step. This dual activation mode explains the high yields observed even with sterically hindered or electronically deactivated substrates. For process chemists, understanding this mechanism is crucial for optimizing reaction parameters such as stoichiometry and temperature to maximize throughput while minimizing impurity formation.
Furthermore, the impurity profile of reactions conducted under these conditions is exceptionally clean, a critical factor for GMP manufacturing environments. The mild nature of the organoiron species prevents common side reactions such as aldol condensation of the starting aldehyde or self-coupling of the alpha-halo ester. The patent data demonstrates that even sensitive functionalities like vinyl groups or heterocycles remain intact throughout the transformation. This selectivity is particularly valuable when synthesizing fluorine-containing beta-hydroxycarbonyl compounds, which are key motifs in modern medicinal chemistry for enhancing metabolic stability and binding affinity. The ability to tolerate diverse substituents without compromising yield suggests that this methodology can be universally applied across a wide library of building blocks. For quality control teams, this translates to simplified purification protocols, often requiring only standard silica gel chromatography to achieve high-purity specifications suitable for clinical trial materials.
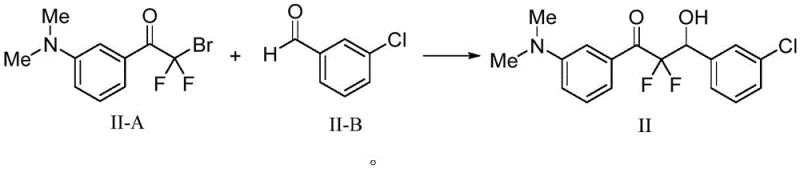
How to Synthesize Beta-Hydroxycarbonyl Compounds Efficiently
Implementing this iron-mediated protocol in a laboratory or pilot plant setting is straightforward and aligns well with existing infrastructure. The process begins with the simple mixing of the alpha-halo carbonyl compound, the aldehyde or ketone substrate, commercial iron powder, and a catalytic amount of iodine in acetonitrile. The reaction mixture is then heated to a moderate temperature of 60°C and stirred for approximately 24 hours. This operational simplicity eliminates the need for specialized equipment required for handling pyrophoric reagents or maintaining cryogenic conditions. Workup involves a standard aqueous quench with ammonium chloride, followed by extraction and purification, making it accessible for both small-scale discovery and large-scale production. The detailed standardized synthetic steps for specific derivatives are outlined below to guide process optimization.
- Combine the alpha-halo carbonyl compound and aldehyde/ketone substrate with commercial iron powder (3 equivalents) and iodine catalyst (0.2 equivalents) in acetonitrile solvent.
- Heat the reaction mixture to 60°C and stir continuously for 24 hours to ensure complete conversion under inert atmosphere conditions.
- Quench the reaction with ammonium chloride solution, extract with ethyl acetate, wash with brine, dry over anhydrous sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology offers transformative benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic reduction of raw material costs. By replacing expensive, activated zinc dust or specialized organometallic reagents with commodity-grade iron powder, manufacturers can significantly lower the bill of materials for key intermediates. Iron is one of the most abundant and affordable metals on earth, ensuring long-term price stability and shielding production budgets from the volatility often seen in the markets for specialty reagents. Moreover, the elimination of rigorous activation steps reduces labor costs and processing time, allowing facilities to increase throughput without capital investment in new reactors. This cost structure improvement is vital for maintaining competitiveness in the generic pharmaceutical and agrochemical sectors where margin pressure is intense.
- Cost Reduction in Manufacturing: The substitution of traditional reagents with commercial iron powder results in substantial cost savings across the entire production lifecycle. Since iron is non-toxic and inexpensive, the expense associated with purchasing, storing, and disposing of hazardous metal waste is virtually eliminated. This qualitative shift in material usage directly impacts the bottom line by reducing waste treatment fees and lowering the overall cost of goods sold. Additionally, the high yields reported in the patent minimize the loss of valuable starting materials, further enhancing the economic viability of the process for high-volume manufacturing.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like iron powder and iodine significantly de-risks the supply chain compared to sourcing specialized activated metals which may have limited suppliers. The widespread availability of these raw materials ensures continuity of supply even during global market disruptions. This reliability is crucial for meeting strict delivery schedules for downstream API manufacturers. Furthermore, the robustness of the reaction reduces the likelihood of batch failures due to reagent quality variations, ensuring a consistent flow of high-quality intermediates to customers.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional methods, aligning with increasingly stringent global regulations on heavy metal discharge. Iron is biologically benign, simplifying the effluent treatment process and reducing the regulatory burden on manufacturing sites. The scalability of the reaction is proven by its tolerance to various substrates and mild conditions, making it suitable for scaling from kilogram to multi-ton production without significant re-engineering. This ease of scale-up supports rapid commercialization of new drug candidates and ensures that supply can meet demand as products move through clinical phases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-mediated synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of using iron powder over zinc in Reformatsky reactions?
A: Iron powder is significantly cheaper and less toxic than zinc or magnesium. Furthermore, the iron-mediated process described in CN110105212B demonstrates superior functional group tolerance, particularly for substrates containing sensitive halogens and nitriles, without requiring rigorous activation steps often needed for zinc dust.
Q: Can this methodology be applied to fluorine-containing pharmaceutical intermediates?
A: Yes, the patent explicitly highlights the synthesis of fluorine-containing beta-hydroxycarbonyl compounds, such as precursors for potent GABA agonists. The mild reaction conditions preserve sensitive fluoro-substituents that might degrade under harsher organometallic conditions.
Q: Is the iodine catalyst essential for the reaction yield?
A: Experimental data within the patent indicates that while iron alone can mediate the reaction, the addition of catalytic iodine (or FeI2) drastically improves yields, often pushing them above 90% for standard substrates like 4-chlorobenzaldehyde, by facilitating the formation of the active organoiron species.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxycarbonyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic methodologies like the iron-catalyzed Reformatsky reaction to enhance our service offerings. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging this cost-effective and environmentally friendly technology, we can offer our partners a competitive edge in terms of both pricing and supply security.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how this methodology can reduce your overall manufacturing expenses. Please contact us to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis needs. Let us be your partner in driving efficiency and innovation in your supply chain.
