Advanced Synthesis of L-Leucine-4-Nitroaniline Hydrochloride for High-Purity Biochemical Applications
Introduction to Advanced Biochemical Reagent Synthesis
The development of efficient synthetic routes for chromogenic substrates is critical for the advancement of enzymatic assays and diagnostic applications. Patent CN102936207A introduces a groundbreaking methodology for the production of L-leucine-4-nitroaniline hydrochloride, a vital biochemical reagent used extensively as a substrate for leucine aminopeptidase and in the synthesis of diazotizing dyes. This innovation addresses the longstanding challenge of low reactivity associated with p-nitroaniline derivatives, offering a robust solution that significantly enhances both yield and operational simplicity. By leveraging a novel condensation strategy, this technology transforms the manufacturing landscape for high-purity peptide substrates, ensuring that research and industrial sectors have access to reliable materials with superior quality specifications. The comprehensive reaction pathway illustrates a streamlined three-step process that maximizes efficiency while minimizing waste.
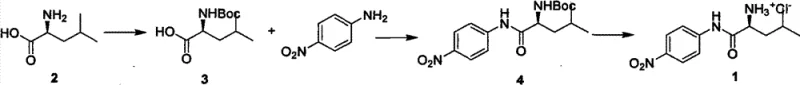
For procurement specialists and supply chain managers, the implications of this patented technology are profound, as it establishes a foundation for a more stable and cost-effective supply of essential biochemical intermediates. The ability to produce this compound with a total recovery rate exceeding 74% and purity levels above 98% represents a significant leap forward compared to historical benchmarks. As a reliable biochemical reagent supplier, understanding these technical nuances allows us to better serve the needs of pharmaceutical and diagnostic companies that demand consistency and excellence in their raw materials. This report delves deep into the mechanistic advantages and commercial viability of this synthesis, providing a clear roadmap for stakeholders looking to optimize their sourcing strategies for complex amino acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino acid p-nitroanilides has been plagued by significant technical hurdles, primarily stemming from the inherently weak nucleophilic character of the p-nitroaniline moiety. Traditional coupling protocols often rely on reagents such as DCC (dicyclohexylcarbodiimide), DCC/HOBt combinations, or acid anhydrides, which frequently result in dismal yields ranging from merely 0% to 30%. Furthermore, some prior art methods necessitate the use of hazardous and expensive reagents like vinyl chloroformate, introducing severe safety risks and escalating production costs due to the need for specialized handling and disposal procedures. These inefficiencies not only drive up the price of the final API intermediate but also create bottlenecks in the supply chain, as low yields necessitate larger batch sizes to meet demand, thereby straining manufacturing capacity. The accumulation of difficult-to-remove byproducts from these conventional condensation agents further complicates downstream purification, often requiring extensive chromatography that is impractical for large-scale commercial operations.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent utilizes phosphorus oxychloride (POCl3) as a potent activating agent within a pyridine solvent system to drive the condensation reaction. This method effectively overcomes the kinetic barrier posed by the weak nucleophile, facilitating the formation of the amide bond with remarkable efficiency and selectivity. The reaction between Boc-L-Leu-OH and p-nitroaniline under these conditions generates the key intermediate Boc-L-Leu-pNA in high yield, bypassing the need for costly coupling additives or toxic chloroformates. This strategic shift in reagent selection not only simplifies the operational workflow but also drastically reduces the environmental footprint of the synthesis by eliminating heavy metal catalysts or complex organic byproducts. The visual representation of this critical coupling step highlights the elegance of the chemistry, where simple reagents combine to form a complex molecular architecture with high fidelity.

From a commercial perspective, this novel approach translates directly into cost reduction in biochemical reagent manufacturing, as the raw materials are commodity chemicals that are easily sourced from global suppliers. The elimination of expensive coupling agents like HOBt or DCC removes a significant line item from the bill of materials, while the simplified workup procedure—often involving simple filtration or extraction—reduces labor and solvent consumption. For supply chain heads, this means a more resilient production model that is less susceptible to fluctuations in the availability of niche reagents, ensuring continuous delivery of high-purity OLED material precursors or pharmaceutical intermediates without interruption.
Mechanistic Insights into POCl3-Mediated Amidation
The core of this technological breakthrough lies in the activation mechanism facilitated by phosphorus oxychloride in a basic pyridine environment. In this system, POCl3 reacts with the carboxylic acid group of the Boc-protected leucine to form a highly reactive mixed anhydride or acyl chloride species in situ. This activated intermediate possesses a much higher electrophilic character than the parent acid, making it susceptible to nucleophilic attack even by the electron-deficient nitrogen of the p-nitroaniline ring. The pyridine serves a dual role as both a solvent and a base, scavenging the hydrogen chloride generated during the reaction and driving the equilibrium towards product formation. This mechanistic pathway avoids the formation of stable urea byproducts typical of carbodiimide couplings, thereby simplifying the impurity profile and enhancing the overall purity of the crude product before any purification steps are undertaken.
Impurity control is paramount in the production of enzyme substrates, as trace contaminants can interfere with kinetic assays and lead to erroneous diagnostic results. The specificity of the POCl3-mediated reaction ensures that side reactions such as racemization of the chiral center in leucine are minimized, preserving the stereochemical integrity required for biological activity. The patent data indicates that the intermediate Boc-L-Leu-pNA can be obtained with purity exceeding 97%, which sets a strong foundation for the final deprotection step. By maintaining strict control over reaction temperatures, specifically utilizing ice-water baths during the addition of reagents, the process prevents thermal degradation and exothermic runaways that could otherwise generate unknown impurities. This rigorous attention to reaction conditions underscores the commitment to producing high-purity biochemical reagents that meet the stringent specifications demanded by the life sciences industry.
How to Synthesize L-Leucine-4-Nitroaniline Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for manufacturing this critical reagent, beginning with the protection of L-leucine and culminating in the formation of the hydrochloride salt. The process is designed to be scalable, utilizing standard laboratory equipment such as four-neck flasks and mechanical stirrers, which facilitates easy translation from pilot scale to full commercial production. Each step has been optimized to maximize yield while minimizing the need for intermediate purification, a key factor in reducing overall processing time and solvent usage. For technical teams looking to implement this route, the following guide summarizes the critical operational parameters and stoichiometric ratios required to achieve the reported success rates.
- Protect L-Leucine with Boc anhydride in aqueous NaOH/dioxane to form Boc-L-Leu-OH.
- Condense Boc-L-Leu-OH with p-Nitroaniline using phosphorus oxychloride (POCl3) in pyridine to form the key intermediate Boc-L-Leu-pNA.
- Deprotect the Boc group using HCl gas in glacial acetic acid to obtain the final L-Leucine-4-Nitroaniline Hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel synthesis method offers substantial strategic benefits for organizations focused on optimizing their supply chain resilience and reducing manufacturing overheads. By shifting away from low-yielding, toxic processes to a high-efficiency condensation route, companies can secure a more stable supply of critical intermediates while simultaneously lowering their cost basis. The use of commodity chemicals like phosphorus oxychloride and pyridine, rather than specialized coupling reagents, insulates the production process from supply shocks associated with niche fine chemicals. Furthermore, the simplified purification requirements mean that manufacturing cycles are shorter, allowing for increased throughput and faster response times to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive coupling agents such as DCC and HOBt, which are traditionally required for difficult amidations, results in a direct and significant decrease in raw material costs. Additionally, the high yield of the reaction reduces the amount of starting material wasted, effectively lowering the cost per kilogram of the final active pharmaceutical ingredient intermediate. The avoidance of toxic reagents like vinyl chloroformate also reduces expenditures related to hazardous waste disposal and regulatory compliance, contributing to a leaner and more economically viable production model.
- Enhanced Supply Chain Reliability: Since the synthesis relies on widely available bulk chemicals rather than proprietary or scarce reagents, the risk of supply disruption is markedly reduced. This accessibility ensures that production schedules can be maintained consistently, preventing delays that could impact downstream customers in the diagnostic and pharmaceutical sectors. The robustness of the reaction conditions further enhances reliability, as the process is less sensitive to minor variations in reagent quality, making it easier to qualify multiple suppliers for raw materials and build a diversified supply base.
- Scalability and Environmental Compliance: The process is inherently scalable, with workup procedures involving simple extractions and filtrations that are easily adapted to large-scale reactor systems. The reduction in toxic waste generation aligns with modern green chemistry principles, facilitating easier permitting and environmental compliance in various jurisdictions. This sustainability advantage not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturer, appealing to end-users who prioritize eco-friendly supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of L-leucine-4-nitroaniline hydrochloride, based on the detailed specifications provided in the patent literature. Understanding these aspects is crucial for R&D directors and procurement managers who are evaluating the feasibility of integrating this reagent into their workflows or supply networks. The answers reflect the specific advantages of the new synthesis method over traditional approaches, highlighting improvements in purity, yield, and operational safety.
Q: Why is the conventional synthesis of L-Leu-pNA inefficient?
A: Conventional methods using DCC or vinyl chloroformate often suffer from extremely low yields (0-30%) due to the weak nucleophilicity of p-nitroaniline, alongside issues with toxic reagents and high costs.
Q: What purity levels can be achieved with this new POCl3 method?
A: The novel synthesis route described in patent CN102936207A consistently achieves a final product purity exceeding 98%, with a total recovery rate greater than 74%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes readily available starting materials and avoids complex purification steps for intermediates, making it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Leucine-4-Nitroaniline Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality biochemical reagents play in advancing medical diagnostics and pharmaceutical research. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global enterprises without sacrificing quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of L-Leucine-4-Nitroaniline Hydrochloride meets the highest industry standards for performance and consistency. Our commitment to technical excellence allows us to deliver products that empower your research and development initiatives with reliable and precise data.
We invite you to collaborate with us to explore how this advanced synthesis technology can benefit your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the biochemical and pharmaceutical sectors.
