Scalable Green Synthesis of 3-Methoxy-N-phenylpropionamide via Selectfluor Oxidation
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for greener, safer, and more cost-effective synthetic routes for high-value intermediates. A recent breakthrough detailed in patent CN110563599B introduces a highly efficient methodology for the preparation of 3-methoxy-N-phenylpropionamide, a crucial scaffold found in various bioactive molecules and pharmaceutical candidates. This innovative approach leverages the unique reactivity of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate)) to facilitate a direct oxidative substitution, bypassing the hazardous multi-step sequences traditionally associated with this chemical class. By utilizing 3-methylthio-N-phenylpropionamide as a stable and readily available starting material, the process achieves remarkable conversion rates under relatively mild thermal conditions. For R&D directors and process chemists, this represents a significant paradigm shift away from transition-metal catalysis toward organo-mediated oxidation, offering a cleaner impurity profile and simplified downstream processing. The strategic implementation of this technology not only enhances laboratory safety but also lays a robust foundation for industrial scale-up, addressing key pain points in the supply chain for complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this development, the synthesis of 3-methoxy-N-phenylpropionamide relied heavily on methodologies that posed substantial environmental and operational challenges. The most prominent existing route, documented in literature such as Chemical Communications (2013), necessitates the initial preparation of N-phenylacrylamide using acryloyl chloride. Acryloyl chloride is notoriously difficult to handle due to its high volatility, severe toxicity, and potential as a lachrymator, requiring specialized containment equipment and rigorous safety protocols that inflate operational expenditures. Furthermore, the subsequent methoxylation step typically employs copper chloride as a catalyst alongside cesium carbonate in dichloromethane. The reliance on transition metal catalysts introduces a critical bottleneck: the absolute necessity for extensive purification steps to reduce residual metal content to ppm levels, which is mandatory for pharmaceutical applications. These metal scavenging processes not only increase material costs but also generate significant volumes of hazardous solid waste, complicating environmental compliance and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast, the novel protocol disclosed in the patent utilizes a direct, one-pot transformation that elegantly circumvents these historical hurdles. By employing 3-methylthio-N-phenylpropionamide as the precursor, the synthesis avoids the generation of the unstable acrylamide intermediate entirely. The reaction proceeds efficiently in methanol, a green and inexpensive solvent, using potassium carbonate as a benign base and Selectfluor as the key oxidant. This streamlined approach consolidates what was previously a multi-step sequence into a single operational unit, drastically reducing solvent consumption and processing time. The efficacy of this method is demonstrated by the high isolated yields achieved under optimized conditions, providing a compelling alternative for manufacturers seeking to modernize their production lines. The elimination of toxic reagents and transition metals simplifies the workup procedure, allowing for a more direct isolation of the target compound with high purity, which is essential for maintaining the integrity of downstream drug synthesis.
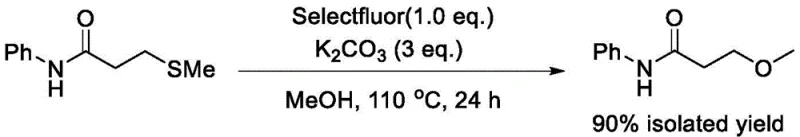
Mechanistic Insights into Selectfluor-Mediated Oxidative Substitution
The mechanistic pathway of this transformation offers fascinating insights into the reactivity of sulfides under electrophilic fluorination conditions. The patent proposes two plausible mechanisms that may operate concurrently or sequentially to drive the reaction forward. In the first scenario, Selectfluor acts as an oxidant to convert the thioether moiety into a reactive sulfonium or sulfoxide intermediate, which subsequently undergoes elimination to generate an N-phenylacrylamide species in situ. This transient acrylamide is then intercepted by methanol in a conjugate addition facilitated by the basic environment provided by potassium carbonate. Alternatively, the mechanism may involve the direct activation of the sulfur atom followed by a nucleophilic displacement where the methoxide ion attacks the beta-carbon, displacing the activated sulfur group. Understanding these nuances is vital for process optimization, as it highlights the dual role of the reagents: Selectfluor serves not merely as a fluorinating agent but as a potent oxidant that activates the C-S bond, while the base ensures the neutrality of the system and promotes the nucleophilic attack.
Crucially, control experiments underscore the indispensable nature of the oxidant in this catalytic cycle. When the reaction is attempted using only potassium carbonate in methanol at 110 °C, absolutely no conversion to the desired product is observed, resulting in a 0% isolated yield. This negative result definitively proves that thermal conditions and base alone are insufficient to cleave the robust C-S bond or facilitate the necessary rearrangement. The presence of Selectfluor is the driving force that lowers the activation energy for the substitution, likely by creating a superior leaving group on the sulfur atom. This mechanistic understanding assures quality control teams that the process is robust; without the oxidant, the starting material remains inert, minimizing the formation of complex degradation byproducts that often arise from uncontrolled thermal decomposition in metal-free systems.
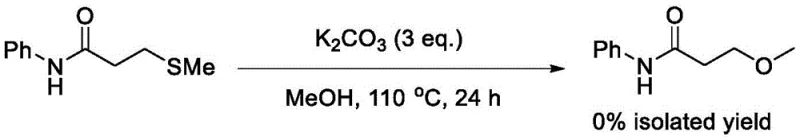
How to Synthesize 3-Methoxy-N-phenylpropionamide Efficiently
Implementing this synthesis on a pilot or production scale requires strict adherence to the optimized parameters established in the patent data to ensure maximum efficiency and reproducibility. The process is designed to be operationally simple, requiring standard glass-lined or stainless steel reactors capable of withstanding moderate pressure and temperature. The key to success lies in the precise stoichiometric balance of the reagents, particularly the ratio of the base to the oxidant, which dictates the reaction kinetics and final purity. Operators should be trained to monitor the reaction progress carefully, as the extended reaction time of 24 hours is necessary to drive the conversion to completion given the stability of the starting thioether. Following the reaction, the workup involves straightforward concentration and chromatographic purification, although on a large scale, crystallization methods may be developed to replace chromatography for further cost reduction. Detailed standardized operating procedures for this synthesis are outlined below to guide technical teams in replication.
- Charge a sealed reactor with methanol solvent, 3-methylthio-N-phenylpropionamide substrate, Selectfluor oxidant (1.0 equivalent), and potassium carbonate base (3.0 equivalents).
- Heat the reaction mixture to 110 °C and maintain vigorous stirring for 24 hours to ensure complete conversion of the thioether precursor.
- Upon completion, concentrate the reaction solution under reduced pressure and purify the crude residue via column chromatography to isolate the target amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Selectfluor-mediated route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for acryloyl chloride, companies can remove a high-hazard material from their inventory, thereby reducing insurance premiums, storage costs, and regulatory reporting burdens associated with toxic substances. Furthermore, the shift away from transition metal catalysts like copper chloride removes the dependency on fluctuating metal markets and eliminates the costly downstream processing steps required for metal scavenging. This translates directly into a leaner manufacturing process with fewer unit operations, lower energy consumption, and reduced waste disposal fees. The use of methanol as the sole solvent further aligns with green chemistry principles, facilitating easier solvent recovery and recycling, which contributes to a more sustainable and economically viable supply chain model for fine chemical production.
- Cost Reduction in Manufacturing: The economic impact of this new method is profound, primarily driven by the elimination of expensive and hazardous reagents. Traditional routes require the purchase of acryloyl chloride, which commands a premium price due to its handling difficulties, and copper salts, which add to the raw material bill. By replacing these with Selectfluor and potassium carbonate, the direct material cost profile is significantly improved. Moreover, the removal of the metal catalyst means that the expensive and time-consuming step of metal scavenging—often involving specialized resins or additional filtration stages—is no longer necessary. This reduction in processing steps leads to lower labor costs, decreased utility usage for heating and cooling multiple stages, and a substantial decrease in the volume of hazardous waste generated, all of which contribute to a lower overall cost of goods sold (COGS) without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: From a logistics perspective, this process enhances supply security by relying on stable, commercially abundant starting materials. 3-Methylthio-N-phenylpropionamide is a robust solid that is easier to store and transport than volatile liquids like acryloyl chloride, reducing the risk of supply disruptions caused by transportation restrictions on hazardous goods. Additionally, the avoidance of transition metals mitigates the risk of supply chain bottlenecks associated with the mining and refining of scarce metals like copper or palladium. The robustness of the reaction conditions—using common solvents and bases—means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring specialized equipment modifications. This flexibility ensures a continuous and reliable flow of intermediates to downstream customers, safeguarding production schedules against external market volatilities.
- Scalability and Environmental Compliance: Scaling this process to multi-ton quantities is inherently safer and more compliant with increasingly stringent environmental regulations. The absence of toxic heavy metals in the effluent stream simplifies wastewater treatment, allowing facilities to meet discharge standards with less intensive remediation efforts. The use of methanol, a solvent with well-established recovery infrastructure, further supports circular economy initiatives within the plant. Because the reaction does not generate complex metal-laden sludge, the disposal costs are markedly lower, and the environmental footprint is minimized. This 'green' credential is increasingly valuable for pharmaceutical companies aiming to improve their sustainability ratings, making suppliers who adopt this technology more attractive partners for long-term contracts focused on responsible sourcing and environmental stewardship.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process details and scalability. These responses are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding the nuances of reagent roles and optimization parameters is critical for successful technology transfer. The following section addresses key concerns regarding catalyst necessity, safety improvements, and stoichiometric sensitivity to provide a comprehensive overview of the method's capabilities and limitations in a commercial setting.
Q: Why is Selectfluor critical for this transformation compared to traditional base-only conditions?
A: Control experiments demonstrate that using potassium carbonate alone results in 0% yield, indicating that the electrophilic fluorinating agent Selectfluor is essential for activating the sulfur moiety, likely through an oxidative mechanism that facilitates the subsequent nucleophilic attack by methanol.
Q: How does this method improve safety profiles compared to the prior art involving acryloyl chloride?
A: This protocol eliminates the need for acryloyl chloride, a highly toxic and volatile lachrymator used in previous synthetic routes to generate the acrylamide intermediate, thereby significantly reducing occupational health hazards and simplifying waste gas treatment requirements.
Q: What is the impact of base stoichiometry on the reaction efficiency?
A: Optimization studies reveal that reducing the potassium carbonate loading from 3.0 equivalents to 2.0 equivalents causes a significant drop in isolated yield from 90% to 55%, suggesting that a sufficient excess of base is required to neutralize acidic byproducts and drive the equilibrium toward the methoxy-substituted product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methoxy-N-phenylpropionamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-methoxy-N-phenylpropionamide meets the highest industry standards. We are committed to leveraging advanced synthetic technologies, such as the Selectfluor-mediated oxidation described here, to deliver high-quality intermediates that accelerate your drug development timelines while maintaining full regulatory compliance.
We invite you to collaborate with us to explore how this optimized synthetic route can benefit your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the economic advantages of switching to this greener methodology. Please contact our technical procurement team today to request specific COA data for our reference batches and to discuss detailed route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your critical pharmaceutical intermediates.
