Advanced Copper Catalysis for High-Purity Benzofuran-Pyrrole Pharmaceutical Intermediates Manufacturing
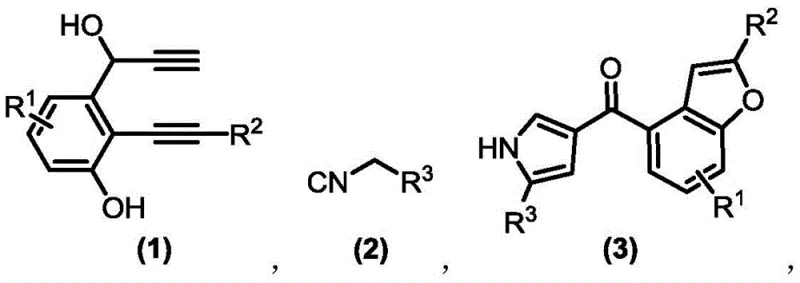
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. A significant breakthrough in this domain is detailed in patent CN109180653B, which discloses a novel method for preparing benzofuran-pyrrole compounds via copper catalysis. This technology addresses the critical need for scalable and environmentally friendly synthetic routes for bioactive molecular frameworks. By utilizing 1,6-diyne-3-alcohol compounds and isocyanides as raw materials in the presence of a metal copper salt catalyst, this process enables the formation of highly functionalized benzofuran-pyrrole structures under remarkably mild conditions. The innovation lies not only in the chemical transformation itself but also in its practical applicability for large-scale industrial production, offering a pathway to high-purity intermediates essential for drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrrole and benzofuran rings has relied on classical methodologies such as Knorr synthesis, Hantzsch reaction, Paal-Knorr condensation, and various multi-component coupling strategies. While these methods have served organic chemistry for decades, they present substantial drawbacks when applied to modern industrial manufacturing. Conventional catalytic systems often suffer from a narrow substrate range, requiring specific starting materials that limit structural diversity. Furthermore, these traditional routes frequently necessitate harsh reaction conditions, including extreme temperatures or strong acidic/basic environments, which can degrade sensitive functional groups and lead to complex impurity profiles. The low product yields associated with these older methods result in poor atom economy, generating excessive waste and driving up the cost of goods sold. Additionally, the multi-step manipulations often required to achieve the final target molecules increase the overall processing time and operational complexity, making them less attractive for rapid scale-up in a commercial setting.
The Novel Approach
In stark contrast to these legacy techniques, the copper-catalyzed method described in the patent offers a streamlined and efficient solution. This novel approach utilizes a cascade reaction strategy where 1,6-diyne-3-alcohol compounds react with isocyanides to directly form the benzofuran-pyrrole skeleton. The reaction proceeds smoothly in common organic solvents like 1,2-dichloroethane at a moderate temperature of 80 °C. The use of readily available metal copper salts, particularly cuprous iodide, as catalysts ensures that the process remains cost-effective while maintaining high catalytic activity. This methodology is particularly distinguished by its ability to synthesize various highly functionalized derivatives with high efficiency and yield, often exceeding 80% and reaching up to 94% in optimized examples. The simplicity of the operation, combined with the ease of product separation and purification, makes this route exceptionally suitable for large-scale industrial production, effectively overcoming the bottlenecks of traditional synthesis.
Mechanistic Insights into Copper-Catalyzed Cascade Cyclization
The success of this synthetic route relies on the unique ability of copper(I) species to activate alkyne functionalities towards nucleophilic attack. In the proposed mechanism, the copper catalyst coordinates with the alkyne moiety of the 1,6-diyne-3-alcohol substrate, increasing its electrophilicity. This activation facilitates an intramolecular cyclization involving the phenolic hydroxyl group to form the benzofuran intermediate. Subsequently, the remaining alkyne group undergoes further activation and reacts with the isocyanide component. This step involves the insertion of the isocyanide carbon into the copper-carbon bond, followed by cyclization and proton transfer to establish the pyrrole ring fused to the benzofuran system. The mild nature of the copper catalysis prevents side reactions such as polymerization or decomposition of the sensitive isocyanide reagents, ensuring a clean reaction profile. This mechanistic pathway highlights the elegance of transition metal catalysis in constructing complex polycyclic systems from simple precursors in a single pot.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring product quality. Harsh conditions often lead to the formation of regioisomers or over-reacted byproducts that are difficult to separate. However, the specificity of the copper-catalyzed cycle minimizes these side pathways. The use of TLC tracking allows for precise monitoring of substrate disappearance, preventing over-reaction. Furthermore, the workup procedure involving extraction with dichloromethane and washing with saturated saline effectively removes inorganic copper residues and polar byproducts. Final purification via silica gel column chromatography using a petroleum ether and ethyl acetate system yields the target benzofuran-pyrrole compounds with high purity, as confirmed by NMR and HRMS data. This rigorous control over the reaction environment and purification process is critical for meeting the stringent quality standards required for pharmaceutical intermediates.
How to Synthesize Benzofuran-Pyrrole Derivatives Efficiently
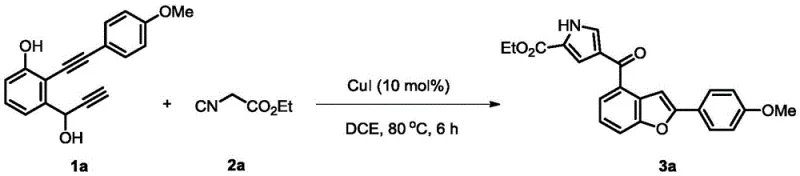
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls. The process begins with the preparation of the reaction mixture in a pressure-resistant tube equipped with magnetic stirring. The molar ratio of the 1,6-diyne-3-alcohol compound to the isocyanide compound is maintained at 1.5:1 to ensure complete consumption of the limiting reagent and maximize yield. The choice of solvent is critical, with 1,2-dichloroethane proving optimal for solubility and reaction kinetics. Detailed standardized synthesis steps see the guide below.
- Combine 1,6-diyne-3-ol compounds and isocyanide compounds in an organic solvent system such as 1,2-dichloroethane with a molar ratio of 1.5: 1.
- Add cuprous iodide (CuI) as the metal copper salt catalyst at a loading of 10 mol% relative to the diyne-alcohol substrate.
- Stir the mixture and carry out a reflux reaction at 80 °C for approximately 6 hours, monitoring progress via TLC until completion.
- Perform post-treatment by pouring into saturated saline, extracting with dichloromethane, drying, and purifying via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed technology translates into tangible strategic benefits. The primary advantage lies in the significant simplification of the manufacturing process, which directly impacts the cost structure. By eliminating the need for expensive precious metal catalysts like palladium or rhodium, and replacing them with abundant copper salts, the raw material costs are drastically reduced. Furthermore, the high atom economy and excellent yields mean that less starting material is wasted, optimizing the utilization of resources. The mild reaction conditions also reduce energy consumption compared to processes requiring high temperatures or cryogenic cooling, contributing to a lower carbon footprint and reduced utility costs. These factors combined create a compelling economic case for switching to this novel synthetic route.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like cuprous iodide significantly lowers the direct material costs. The high yields observed across a broad substrate scope minimize the need for recycling unreacted starting materials, thereby reducing waste disposal costs and improving overall process efficiency. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phases during purification, leading to substantial operational savings.
- Enhanced Supply Chain Reliability: The starting materials, specifically 1,6-diyne-3-alcohol compounds and isocyanides, are readily available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The reaction operates in standard organic solvents and does not require specialized high-pressure equipment, making it easily scalable from laboratory to pilot and commercial plant scales. The green and environment-friendly nature of the reaction system, characterized by mild conditions and easy separation, simplifies compliance with increasingly stringent environmental regulations. The ability to produce high-purity products with minimal byproducts reduces the burden on wastewater treatment facilities and aligns with sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this copper-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the optimal reaction conditions for this copper-catalyzed synthesis?
A: The optimal conditions involve using cuprous iodide (CuI) at 10 mol% loading in 1,2-dichloroethane (DCE) solvent at a temperature of 80 °C for 6 hours, yielding high purity products.
Q: Does this method support a broad range of substrate functional groups?
A: Yes, the method demonstrates excellent substrate tolerance, successfully accommodating aryl, heteroaryl, alkenyl, and substituted aryl groups with various electron-donating or withdrawing substituents like halogens and alkoxy groups.
Q: How does this copper catalysis method compare to traditional pyrrole synthesis routes?
A: Unlike traditional methods such as Knorr or Hantzsch syntheses which often suffer from harsh conditions and low yields, this copper-catalyzed cascade reaction operates under mild conditions with significantly improved atom economy and product yields up to 94%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran-Pyrrole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed methodology for producing high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including precise temperature control and efficient solvent recovery systems. We are committed to delivering benzofuran-pyrrole compounds that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify identity and potency.
We invite potential partners to engage with our technical team to explore how this innovative route can optimize your supply chain. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us collaborate to bring your next generation of pharmaceutical or material science products to market faster and more efficiently.
