Advanced Palladium-Catalyzed Carbonylation for Scalable Homopiperazinone Derivative Synthesis
Advanced Palladium-Catalyzed Carbonylation for Scalable Homopiperazinone Derivative Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex nitrogen-containing heterocycles, which serve as critical scaffolds in numerous bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112062769B, which discloses a highly efficient process for preparing penta-and hepta-homopiperazinone derivatives via catalytic carbonylation of aminoalkyleneamine oxides. This technology represents a paradigm shift from traditional cyclization methods, offering a streamlined pathway that utilizes carbon monoxide gas as a key building block under palladium catalysis. The ability to construct these seven-membered rings with high atom economy and selectivity addresses long-standing challenges in medicinal chemistry, particularly for targets requiring specific spatial arrangements of nitrogen atoms. As a leading manufacturer, we recognize the immense potential of this synthetic route to enhance the supply chain reliability for high-purity pharmaceutical intermediates.
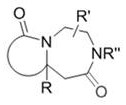
The core innovation lies in the transformation of readily available aminoalkenyl amide compounds into complex bicyclic systems. Unlike conventional approaches that may require harsh conditions or multi-step sequences, this patented method leverages the power of transition metal catalysis to achieve direct ring closure. The general structure of the resulting products, as illustrated in the patent, allows for significant diversification through various R groups, enabling the synthesis of a broad library of analogues for drug discovery programs. This flexibility is crucial for R&D directors who need to rapidly iterate on lead compounds while maintaining strict control over impurity profiles. The process operates under relatively mild thermal conditions, typically ranging from 50°C to 150°C, which minimizes thermal degradation of sensitive functional groups often present in advanced intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of five-and seven-membered cyclic compounds containing nitrogen has relied heavily on Wacker-type cyclization reactions. However, these traditional methods face substantial hurdles when applied to substrates containing diamine functionalities. The presence of multiple nitrogen atoms creates an extremely strong coordination effect with metal centers, particularly copper, which is commonly used in standard Wacker oxidation systems. This strong coordination effectively poisons the catalyst or prevents the necessary substrate activation, leading to poor conversion rates and low yields. Furthermore, conventional routes often necessitate the use of stoichiometric amounts of toxic heavy metal oxidants, generating significant quantities of hazardous waste that complicate downstream processing and environmental compliance. The purification of products from these messy reaction mixtures often requires extensive chromatographic separation, driving up both the cost of goods sold and the lead time for material delivery.
The Novel Approach
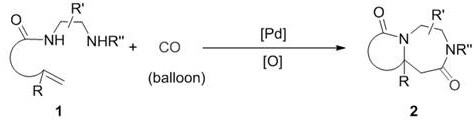
The novel approach described in the patent circumvents these limitations by employing a palladium-catalyzed carbonylation strategy that is specifically tuned to tolerate diamine coordination. By introducing carbon monoxide gas into the reaction system, the process facilitates a unique cyclization mechanism that constructs the homopiperazinone core with exceptional efficiency. The use of modulated oxidants, such as benzoquinone or specific copper salts, in conjunction with the palladium catalyst ensures smooth substrate conversion without the inhibition seen in traditional Wacker systems. This method not only improves the chemical yield, with examples demonstrating yields as high as 96%, but also simplifies the operational workflow. The reaction proceeds in common organic solvents like 1,4-dioxane or toluene, and the gaseous nature of the carbonyl source means that excess reagent can be easily vented, leaving behind a cleaner crude product that is easier to purify.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for scaling the process effectively. The reaction initiates with the coordination of the palladium catalyst to the olefinic moiety of the aminoalkenyl amide substrate. In the presence of carbon monoxide, a migratory insertion occurs, forming an acyl-palladium species. This intermediate is key to the formation of the new carbon-carbon bond that closes the ring. The presence of the oxidant is critical for regenerating the active palladium species from its reduced state, thereby sustaining the catalytic cycle. Unlike standard oxidative carbonylations where copper chloride is the sole oxidant, this system benefits from the use of organic oxidants like benzoquinone or specific inorganic salts that do not strongly sequester the palladium center. This delicate balance allows the reaction to proceed with high turnover numbers, ensuring that even at low catalyst loadings of 0.5-5 mol%, the conversion remains high.
From an impurity control perspective, the mechanism offers distinct advantages. The high selectivity of the palladium catalyst towards the specific intramolecular cyclization minimizes the formation of oligomeric by-products or intermolecular coupling products that often plague amine chemistry. The reaction conditions are sufficiently mild to prevent the epimerization of chiral centers adjacent to the nitrogen atoms, preserving the stereochemical integrity of the molecule. Additionally, the use of carbon monoxide gas as a reagent means that there are no residual liquid carbonyl sources to remove during workup, reducing the burden on the purification team. The resulting crude products typically show clean NMR spectra with minimal side peaks, indicating that the reaction pathway is highly directed towards the desired thermodynamic product. This level of control is essential for meeting the stringent purity specifications required for clinical trial materials.
How to Synthesize Homopiperazinone Derivatives Efficiently
The synthesis of these valuable heterocycles can be achieved through a straightforward protocol that is amenable to both laboratory scale and pilot plant operations. The procedure involves charging a reaction vessel with the specific aminoalkenyl amide precursor, a palladium source such as PdCl2 or Pd(OAc)2, and a suitable oxidant. The choice of solvent is flexible, with options ranging from polar aprotic solvents like DMF to ethers like 1,4-dioxane, allowing process chemists to optimize for solubility and safety. Once the reagents are combined, the system is purged with carbon monoxide to establish the necessary atmosphere, and the mixture is heated to the target temperature, typically around 100°C for optimal kinetics.
- Charge the reactor with the aminoalkenyl amide substrate, palladium catalyst (e.g., PdCl2), and oxidant (e.g., benzoquinone) in a suitable solvent like 1,4-dioxane.
- Replace the atmosphere with carbon monoxide (CO) gas multiple times to ensure an inert and reactive environment, then heat the mixture to 50-150°C.
- Stir the reaction for 10-30 hours, cool to room temperature, remove solvent under reduced pressure, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant reduction of manufacturing complexity. By eliminating the need for harsh acidic conditions or exotic reagents often required in alternative cyclization methods, the process reduces the wear and tear on reactor equipment and lowers the barrier for safe operation. The use of carbon monoxide, while requiring appropriate safety protocols, is a commodity chemical that is readily available globally, ensuring that raw material supply chains remain robust and unaffected by geopolitical fluctuations affecting specialty reagents. Furthermore, the high selectivity of the reaction means that less starting material is wasted on by-products, directly improving the mass balance and reducing the overall cost of raw materials per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal scavengers is a major cost driver. Since the palladium catalyst is used in low loadings and the reaction generates fewer metal-complexed impurities, the downstream purification process is drastically simplified. This avoids the need for costly resin treatments or multiple recrystallization steps that are typical in processes using high loads of copper or other metals. Additionally, the simplicity of the workup, which often involves merely removing the solvent and running a standard column, reduces labor hours and solvent consumption, leading to substantial cost savings in the overall production budget without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates. Traditional methods that suffer from catalyst poisoning due to diamine coordination often result in stalled reactions or inconsistent yields, creating bottlenecks in the supply chain. This new method's tolerance for various functional groups and its reliable performance across a wide temperature range ensure consistent output. This predictability allows supply chain planners to forecast delivery dates with greater accuracy, reducing the risk of stockouts for critical API intermediates. The ability to source diverse substrates, including those with halogen or methoxy substituents, further diversifies the supply base and mitigates single-source risks.
- Scalability and Environmental Compliance: Scaling gas-liquid reactions can be challenging, but the moderate pressure requirements (balloon or slight positive pressure) make this process highly scalable in standard stainless steel reactors. The reduced generation of heavy metal waste aligns with increasingly strict environmental regulations, lowering the costs associated with waste disposal and treatment. The process avoids the use of chlorinated solvents in many examples, favoring greener alternatives like alcohols or ethers where possible. This environmental profile not only reduces regulatory burden but also enhances the sustainability credentials of the final product, a factor that is becoming increasingly important for downstream pharmaceutical customers aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a realistic view of the technology's capabilities. Understanding these nuances helps in setting appropriate expectations for project timelines and resource allocation.
Q: What are the optimal reaction conditions for this carbonylation process?
A: The process typically operates between 50°C and 150°C, with a preferred temperature around 100°C. The reaction duration ranges from 10 to 30 hours under a carbon monoxide atmosphere, utilizing palladium catalysts like PdCl2 at loadings of 0.5-5 mol%.
Q: Which oxidants are compatible with this palladium-catalyzed system?
A: The method supports a wide range of oxidants including organic options like benzoquinone and inorganic salts such as copper acetate, copper chloride, or silver carbonate. The molar ratio of oxidant to substrate is generally maintained between 1:1 and 2:1.
Q: How does this method improve upon traditional Wacker cyclization?
A: Traditional Wacker cyclization often struggles with diamine substrates due to strong metal coordination that inhibits conversion. This novel carbonylation approach modulates the catalyst and oxidant system to overcome these coordination issues, resulting in higher activity and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homopiperazinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we understand that translating a patented laboratory method into a commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is seamless. We are equipped with state-of-the-art high-pressure reactors capable of safely handling carbon monoxide chemistry, along with stringent purity specifications enforced by our rigorous QC labs. Our commitment to quality ensures that every batch of homopiperazinone derivatives meets the exacting standards required for pharmaceutical applications, minimizing the risk of delays in your drug development timeline.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel analogues or large-scale production of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how this efficient carbonylation process can optimize your supply chain and reduce your overall manufacturing costs.
