Advanced Synthesis of Polyphenyl Pyridinium Salts for High-Performance Material Applications
Advanced Synthesis of Polyphenyl Pyridinium Salts for High-Performance Material Applications
The landscape of functional organic materials is constantly evolving, driven by the demand for compounds with superior electrochemical and optical properties. A pivotal advancement in this sector is detailed in patent CN101619037A, which introduces a robust and scalable method for preparing polyphenyl substituted alpha active pyridinium compounds. These specialized heterocyclic salts represent a significant leap forward from traditional pyridinium derivatives, offering unique structural characteristics that enable applications ranging from nonlinear optical materials to charge transport layers in electronic devices. The innovation lies not just in the final molecular architecture, but in the streamlined synthetic pathway that transforms readily available polyphenyl-substituted cyclopentadiene precursors into high-value pyridinium salts through a pyrylium intermediate. This technical breakthrough addresses long-standing challenges in heterocyclic synthesis, providing a reliable foundation for the commercial production of advanced fine chemicals.
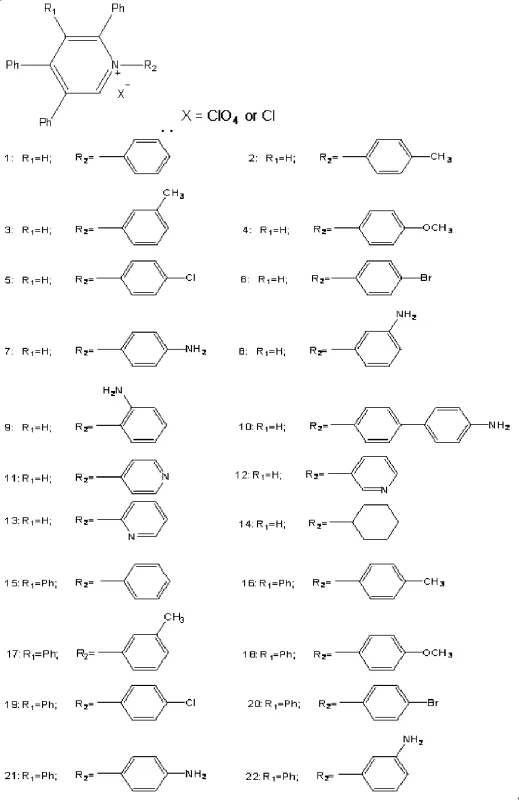
For R&D directors and procurement specialists seeking a reliable fine chemical supplier, understanding the structural versatility of these compounds is crucial. The patent delineates two primary classes of products, with the first class featuring a monomeric pyridinium core adorned with multiple phenyl groups. The ability to tune the substituents at the nitrogen position (R2) and the ring positions (R1) allows for precise modulation of the compound's solubility, melting point, and electronic band gap. This tunability is essential for optimizing performance in specific applications such as phase transfer catalysis or as cationic surfactants. Furthermore, the synthetic route described ensures that these complex architectures can be accessed without the prohibitive costs often associated with multi-step heterocyclic functionalization, making them viable candidates for large-scale industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridinium salts has been fraught with inefficiencies that hinder commercial viability. Traditional methodologies often rely on the Menschutkin reaction between pyridine derivatives and halohydrocarbons, which can suffer from slow kinetics and the formation of difficult-to-remove quaternary ammonium byproducts. Alternatively, ring-closing condensations or cycloaddition reactions frequently require severe reaction conditions, including high temperatures and strong Lewis or Brønsted acids, which can degrade sensitive functional groups. These conventional pathways are typically multi-step processes with cumulative yield losses, leading to increased waste generation and higher overall manufacturing costs. For supply chain managers, the reliance on harsh reagents and complex purification protocols translates to longer lead times and greater operational risk, particularly when scaling up to metric ton quantities required for material science applications.
The Novel Approach
In stark contrast, the methodology outlined in CN101619037A offers a paradigm shift towards efficiency and simplicity. By utilizing a polyphenyl-substituted cyclopentadiene as the starting material, the process bypasses the need for pre-functionalized pyridine rings. The key innovation involves an oxidative transformation to generate a pyrylium salt intermediate, which is highly reactive towards nucleophilic attack by amines. This two-stage sequence—oxidation followed by aminolysis—proceeds under relatively mild conditions, often at room temperature or with gentle heating, significantly reducing energy consumption. The use of common organic solvents and commercially available oxidants further simplifies the supply chain logistics. This novel approach not only enhances the productive rate, with experimental examples demonstrating yields around 74% to 80%, but also facilitates easier product isolation through simple precipitation and recrystallization techniques, thereby drastically reducing downstream processing costs.
Mechanistic Insights into Oxidative Cyclization and Aminolysis
To fully appreciate the value proposition for cost reduction in electronic chemical manufacturing, one must delve into the mechanistic underpinnings of this synthesis. The process initiates with the oxidation of the cyclopentadiene derivative. In the presence of an oxidant such as perchlorate or hypochlorite, the five-membered ring undergoes a transformation that introduces oxygen, forming the aromatic pyrylium cation. This intermediate is electronically activated, possessing a positive charge that makes the ring carbons highly susceptible to nucleophilic attack. When an organic amine is introduced, it attacks the alpha-position of the pyrylium ring. This nucleophilic addition is followed by a ring-opening and subsequent recyclization or direct substitution mechanism that incorporates the nitrogen atom into the six-membered ring, ultimately yielding the stable pyridinium cation. The presence of base and acid assistants in the reaction mixture plays a critical role in buffering the system and driving the equilibrium towards the desired pyridinium product, ensuring high conversion rates.
Impurity control is another critical aspect where this mechanism excels, directly impacting the purity profiles required by pharmaceutical and electronic grade customers. The specificity of the amine attack on the pyrylium intermediate minimizes the formation of regio-isomers that often plague traditional electrophilic aromatic substitutions. Furthermore, the byproduct of the transformation is typically water or simple salts, which are easily removed during the aqueous workup or washing steps described in the patent embodiments. The structural integrity of the polyphenyl groups is maintained throughout the oxidative and aminolytic steps, preserving the conjugated system necessary for the material's optical and charge-transport properties. This mechanistic clarity allows process chemists to predict and control the outcome with high precision, ensuring batch-to-b consistency which is paramount for high-purity OLED material or intermediate production.
How to Synthesize Polyphenyl Pyridinium Salts Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometry to maximize yield and purity. The patent specifies a range of compatible organic solvents including methylene dichloride, acetonitrile, and toluene, allowing flexibility based on solubility parameters of the specific substrates. The oxidation step is typically conducted under air atmosphere with dropwise addition of the oxidant to control exotherms, followed by concentration to isolate the pyrylium intermediate. In the second stage, the intermediate is redissolved, and the amine component is added along with a base agent such as carbonate or hydroxide.
- Oxidize polyphenyl-substituted cyclopentadiene in an organic solvent using an oxidant to generate the pyrylium salt intermediate.
- Dissolve the pyrylium intermediate and react with an organic amine, base agent, and acid assistant under stirring.
- Isolate the crude product via precipitation, followed by recrystallization and washing to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for exotic catalysts or extreme reaction conditions, the process lowers the barrier to entry for production and reduces the dependency on specialized equipment. This accessibility fosters a more resilient supply chain, as the raw materials—polyphenyl cyclopentadienes and common amines—are widely available from global chemical distributors. The robustness of the reaction conditions also implies a lower risk of batch failures due to minor process deviations, enhancing overall supply reliability for downstream customers who depend on consistent material flow for their own production lines.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the high atom economy and simplified purification steps. Unlike traditional methods that may require chromatography or extensive distillation to remove complex byproducts, this process relies on precipitation and recrystallization. This shift from energy-intensive separation techniques to simple phase separations results in substantial cost savings in utilities and labor. Additionally, the high yields reported in the patent embodiments suggest that less raw material is wasted per unit of product, further optimizing the cost of goods sold (COGS) and allowing for more competitive pricing in the global market for specialty intermediates.
- Enhanced Supply Chain Reliability: The use of stable intermediates and mild reaction conditions contributes to a more predictable production schedule. The pyrylium salt intermediate can potentially be stored or transported if necessary, adding flexibility to the manufacturing timeline. Moreover, the avoidance of hazardous reagents like strong alkylating agents reduces regulatory burdens and safety risks associated with storage and handling. This compliance ease accelerates the time-to-market for new products and ensures uninterrupted supply continuity, a critical factor for clients managing just-in-time inventory systems in the fast-paced electronics and pharmaceutical sectors.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates fewer hazardous wastes compared to heavy metal-catalyzed couplings or halogen-heavy substitutions. The solvents used are standard industrial grades that can be recovered and recycled, aligning with green chemistry principles. The scalability is evidenced by the straightforward nature of the unit operations—mixing, stirring, filtering, and drying—which are easily transferred from laboratory glassware to large-scale stainless steel reactors. This seamless scale-up potential ensures that the commercial scale-up of complex polymer additives or electronic materials can be achieved rapidly to meet surging market demand without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced pyridinium salts. Understanding these details helps stakeholders make informed decisions about integrating these materials into their product portfolios.
Q: What are the primary advantages of this pyrylium salt route over traditional pyridinium synthesis?
A: This method avoids harsh Lewis acid conditions and complex multi-step ring closures found in conventional methods. It utilizes mild oxidation and ambient temperature aminolysis, resulting in simpler workup procedures and higher isolated yields suitable for industrial scaling.
Q: Can this synthesis method produce both mono- and bis-pyridinium structures?
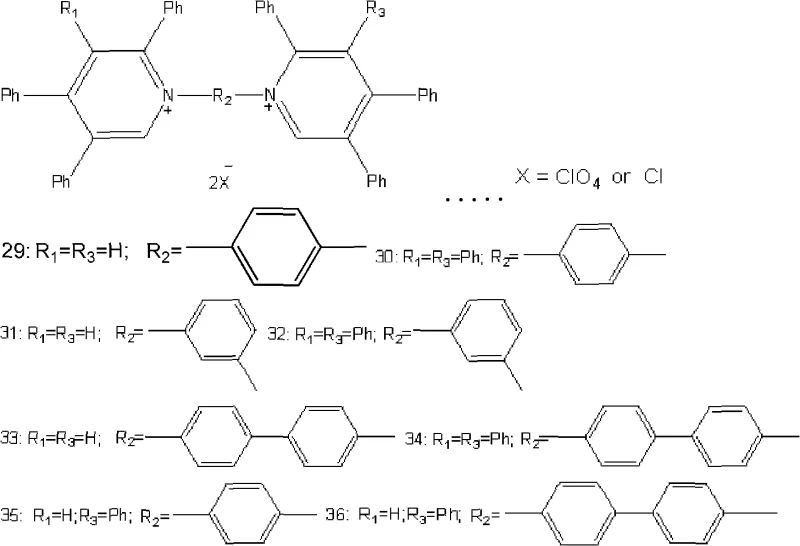
A: Yes, the patent describes two distinct classes of compounds. By selecting specific diamines or mono-amines as the nitrogen source, manufacturers can selectively produce either monomeric polyphenyl pyridinium salts or dimeric bis-pyridinium structures linked by aromatic spacers.
Q: What purification methods are recommended for these alpha-active pyridinium salts?
A: The process relies on solubility differences for purification. After the reaction, the product is precipitated using non-polar solvents like diethyl ether and subsequently purified through recrystallization from acetonitrile or similar solvents to remove residual amines and salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyphenyl Pyridinium Salt Supplier
As the global demand for high-performance organic materials continues to surge, partnering with an experienced CDMO is essential for translating patent innovations into commercial reality. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in heterocyclic chemistry to deliver exceptional results. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polyphenyl pyridinium salt performs consistently in your final applications, whether they be in advanced optics or pharmaceutical formulations.
We invite you to collaborate with us to unlock the full potential of this technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our superior manufacturing capabilities and commitment to quality excellence.
