Advanced Synthetic Strategy for Lubaibailong: Enhancing Yield and Commercial Viability
Advanced Synthetic Strategy for Lubaibailong: Enhancing Yield and Commercial Viability
The pharmaceutical landscape for metabolic disorder treatments is constantly evolving, driven by the need for more efficient and cost-effective manufacturing processes for active pharmaceutical ingredients (APIs). Patent CN114380811A introduces a groundbreaking synthetic methodology for Lubaibailong (CAS: 391926-19-5), a potent β3-adrenoceptor agonist utilized in the management of type II diabetes and obesity. This patent details a novel six-step sequence that strategically bypasses traditional bottlenecks associated with noble metal catalysis and complex purification regimes. By leveraging a nickel-catalyzed reduction system and optimizing Suzuki coupling conditions, the disclosed route offers a compelling value proposition for global supply chains seeking to secure reliable sources of high-purity pharmaceutical intermediates. The technical innovations presented here not only promise substantial improvements in overall yield but also align with modern green chemistry principles by reducing reliance on precious metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for complex heterocyclic compounds like Lubaibailong often rely heavily on palladium-catalyzed hydrogenation or other noble metal-mediated reductions to convert nitro groups into amines. These conventional methods frequently necessitate high-pressure equipment, specialized safety protocols, and expensive catalyst recovery systems, which collectively inflate the cost of goods sold (COGS). Furthermore, the removal of trace heavy metal residues from the final API is a rigorous and costly regulatory requirement, often involving multiple purification cycles that erode overall process efficiency. In many legacy pathways, the use of harsh reaction conditions can lead to the formation of difficult-to-separate impurities, complicating the downstream processing and potentially compromising the quality of the final drug substance. These factors create significant friction in the supply chain, leading to longer lead times and reduced flexibility for manufacturers responding to market demand fluctuations.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift by introducing a mild, atmospheric pressure reduction strategy using nickel chloride hexahydrate and sodium borohydride. This approach effectively circumvents the need for high-pressure hydrogenation, thereby drastically simplifying the operational infrastructure required for production. The substitution of expensive palladium catalysts with abundant nickel salts results in a direct and significant reduction in raw material costs, while the mild reaction conditions minimize thermal degradation and side reactions. Additionally, the optimized workup procedures, which utilize simple solvent extractions and filtrations rather than complex chromatographic separations at every stage, streamline the manufacturing workflow. This novel pathway not only enhances the economic viability of Lubaibailong production but also ensures a more robust and consistent supply of critical intermediates for downstream formulation.
Mechanistic Insights into Nickel-Catalyzed Reduction and Suzuki Coupling
The core chemical innovation lies in the reduction of the nitro-intermediate (Intermediate 1) to the corresponding amine (Intermediate 2) using a NiCl2·6H2O and NaBH4 system. Mechanistically, this generates a reactive nickel boride species in situ, which acts as a highly effective hydrogenation catalyst under ambient conditions. This catalytic cycle facilitates the transfer of hydride equivalents to the nitro group with high chemoselectivity, preserving other sensitive functional groups within the molecule that might be susceptible to reduction under harsher conditions. The precise control of stoichiometry, specifically maintaining a molar ratio of intermediate to reducing agent around 1:10, ensures complete conversion while preventing over-reduction or decomposition. This mechanistic elegance allows for the generation of high-purity amine intermediates without the contamination risks associated with heterogeneous noble metal catalysts, thereby simplifying the subsequent purification logic.
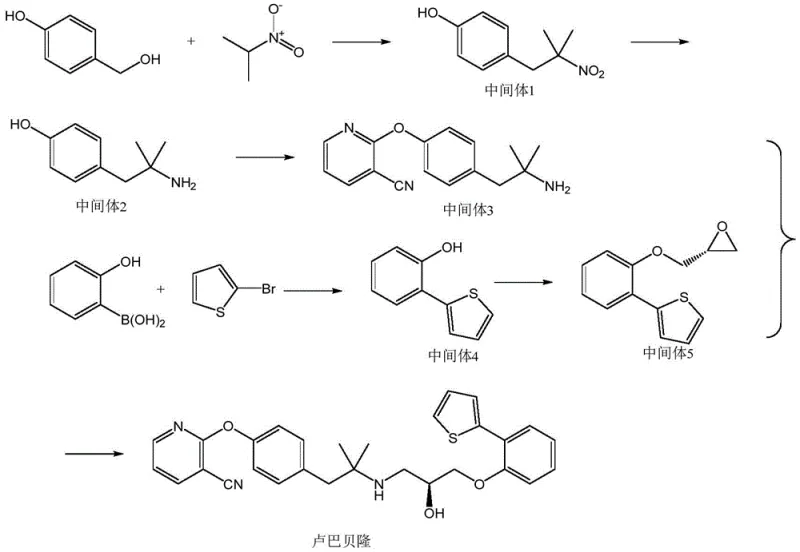
Furthermore, the integration of a Suzuki-Miyaura cross-coupling reaction to construct the biaryl ether linkage demonstrates a sophisticated approach to building molecular complexity with high fidelity. The reaction between 2-hydroxyphenylboronic acid and 2-bromothiophene, catalyzed by tetrakis(triphenylphosphine)palladium, proceeds efficiently in a mixed solvent system of toluene, ethanol, and water. This specific solvent combination optimizes the solubility of both organic and inorganic reagents, facilitating the transmetallation step crucial for the catalytic cycle. The careful regulation of base concentration and temperature (75-85°C) minimizes the formation of homocoupling byproducts and protodeboronation side reactions. By securing this key carbon-carbon bond early in the synthesis with high yield and selectivity, the overall impurity profile of the final API is significantly improved, reducing the burden on final crystallization steps and ensuring compliance with stringent pharmacopeial standards.
How to Synthesize Lubaibailong Efficiently
The synthesis of Lubaibailong via this optimized route requires precise adherence to reaction parameters to maximize yield and purity. The process begins with the condensation of p-hydroxybenzyl alcohol and 2-nitropropane, followed by the critical nickel-catalyzed reduction step that defines the cost-efficiency of the entire sequence. Subsequent nucleophilic substitutions and the Suzuki coupling must be monitored closely to ensure complete conversion before proceeding to the final ring-opening assembly. The detailed standardized operating procedures for each transformation, including specific solvent ratios, temperature gradients, and workup protocols, are essential for replicating the high yields reported in the patent data. For process chemists looking to implement this technology, understanding the nuances of the purification steps, such as the specific eluent ratios for column chromatography, is vital for successful technology transfer.
- Condense p-hydroxybenzyl alcohol with 2-nitropropane using potassium tert-butoxide to form Intermediate 1.
- Reduce Intermediate 1 using nickel chloride hexahydrate and sodium borohydride to obtain Intermediate 2.
- React Intermediate 2 with 2-chloro-3-cyanopyridine under alkaline conditions to generate Intermediate 3.
- Perform Suzuki coupling between 2-hydroxyphenylboronic acid and 2-bromothiophene to yield Intermediate 4.
- Substitute Intermediate 4 with dextrorotatory epichlorohydrin to form Intermediate 5.
- Conduct a ring-opening reaction between Intermediate 3 and Intermediate 5 to finalize Lubaibailong.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers transformative benefits that extend far beyond simple unit price reductions. By eliminating the dependency on noble metal catalysts and high-pressure reactors, the manufacturing process becomes inherently less capital intensive and more accessible to a broader range of qualified contract manufacturing organizations (CMOs). This diversification of potential manufacturing partners enhances supply chain resilience, reducing the risk of bottlenecks that often occur when specialized high-pressure facilities are fully booked. The simplified purification protocols further contribute to operational efficiency, shortening the batch cycle time and allowing for faster throughput without compromising on quality standards. These structural improvements in the manufacturing process translate directly into a more stable and predictable supply of Lubaibailong intermediates for global pharmaceutical clients.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with nickel-based systems fundamentally alters the cost structure of the synthesis, removing one of the most volatile and expensive input variables. This shift not only lowers the direct material costs but also reduces the downstream expenses associated with metal scavenging and waste disposal, which are significant cost drivers in traditional noble metal chemistry. The ability to perform reactions at atmospheric pressure further reduces energy consumption and equipment maintenance costs, contributing to a leaner and more competitive manufacturing model. Consequently, this enables the delivery of high-purity pharmaceutical intermediates at a price point that supports broader market accessibility for the final therapeutic.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as nickel chloride, sodium borohydride, and common organic solvents ensures that the supply chain is not vulnerable to the geopolitical or logistical constraints often associated with precious metals. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without significant yield loss, adds a layer of operational security that is highly valued in large-scale production environments. This reliability allows supply chain managers to forecast inventory needs with greater confidence and maintain optimal stock levels to meet fluctuating market demands. Ultimately, this stability fosters stronger long-term partnerships between suppliers and pharmaceutical developers.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing solvents and reagents that are readily available in bulk quantities and compatible with standard stainless steel reactor setups. The avoidance of high-pressure hydrogenation removes a major safety barrier to scale-up, facilitating a smoother transition from pilot plant to commercial tonnage production. Moreover, the reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, minimizing the ecological footprint of the manufacturing process. This commitment to sustainable chemistry not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial aspects of the Lubaibailong synthesis route, providing clarity for stakeholders evaluating this technology. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of integrating this synthetic method into existing production portfolios.
Q: How does the new synthesis route reduce production costs for Lubaibailong?
A: The novel route replaces expensive noble metal catalysts with a nickel chloride and sodium borohydride system for the reduction step. This eliminates the need for high-pressure hydrogenation equipment and costly palladium catalysts, significantly lowering raw material and operational expenses while simplifying the purification process.
Q: What are the key purity advantages of this synthetic method?
A: By optimizing reaction solvents and alkali usage in the substitution steps, the method minimizes side reactions and byproduct formation. The specific purification protocols, such as chloroform filtration and controlled column chromatography, ensure high-purity intermediates, leading to a cleaner final API with a simplified impurity profile.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the process operates under normal pressure and moderate temperatures (e.g., 55-65°C for key steps), avoiding extreme conditions that hinder scale-up. The use of common solvents like ethanol and acetone, along with robust reaction conditions, makes the transition from laboratory to commercial tonnage production highly feasible and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lubaibailong Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN114380811A for securing the future supply of critical metabolic therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality material regardless of volume. Our state-of-the-art facilities are equipped to handle the specific requirements of this nickel-catalyzed chemistry, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to translating laboratory innovations into robust industrial processes that drive value for our partners.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain objectives. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impacts of switching to this more efficient manufacturing route. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to engineer a supply solution that balances cost, quality, and reliability for your Lubaibailong requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
