Scalable Synthesis of 5-Oxo-Tetrahydropyran-3-Carboxylic Ester for Advanced Pharmaceutical Applications
Scalable Synthesis of 5-Oxo-Tetrahydropyran-3-Carboxylic Ester for Advanced Pharmaceutical Applications
The pharmaceutical industry constantly demands robust, scalable, and environmentally sustainable pathways for constructing complex heterocyclic scaffolds essential for modern drug discovery. Patent CN108558806B introduces a groundbreaking methodology for the preparation of 5-oxo-tetrahydropyran-3-carboxylic ester, a pivotal building block in the synthesis of potent therapeutic agents. This technology addresses critical bottlenecks in the manufacturing of tetrahydropyran-based intermediates, which are widely utilized in the development of ATPase inhibitors and mTOR kinase inhibitors for oncology applications. By leveraging a strategic sequence of halogenation, palladium-catalyzed carbonylation, and selective oxidation, this process offers a superior alternative to legacy synthetic routes. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance and commercial viability of this patent is paramount for securing supply chains for next-generation antitumor medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically disclosed in patent CN102171210B, relies on a convoluted and hazardous synthetic strategy that poses significant risks for industrial implementation. The traditional route initiates with the reaction of methyl glycolate and methyl acrylate using sodium hydride, followed by reduction and nucleophilic substitution with aniline. Crucially, the final steps involve the use of diiodomethane for alkylation and tri-n-butyltin hydride for radical dehalogenation. These reagents are not only highly toxic and environmentally damaging but also necessitate complex purification protocols to remove tin residues, which is a major regulatory hurdle in API manufacturing. Furthermore, the use of benzene as a solvent presents severe occupational health risks. The cumulative effect of these inefficiencies results in a dismal total yield of merely 7.07 percent, rendering the process economically unviable for large-scale commercial production.
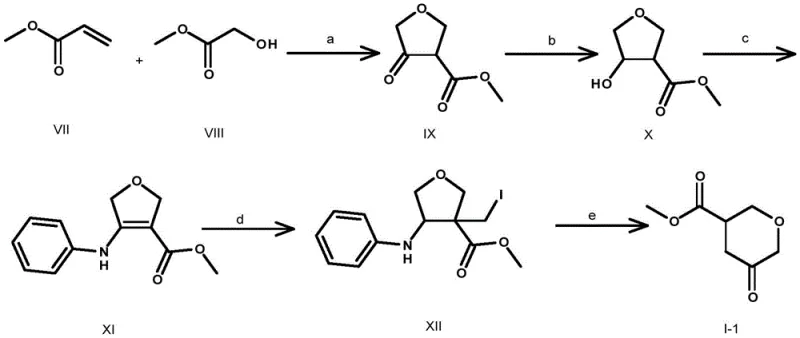
The Novel Approach
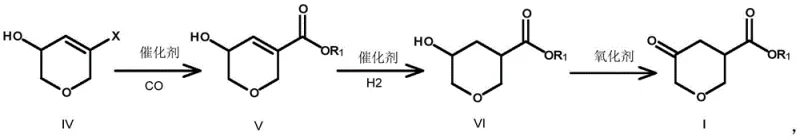
In stark contrast, the novel methodology outlined in CN108558806B utilizes 2H-pyran-3,5(4H,6H)-dione as a readily available starting material to construct the target scaffold with exceptional efficiency. This innovative route bypasses the need for toxic organotin reagents and carcinogenic solvents entirely. Instead, it employs a palladium-catalyzed carbonylation step to install the ester functionality directly onto the pyran ring, a transformation that is both atom-economical and operationally simple. The process flow involves a controlled halogenation to activate the ring, followed by selective reduction to an allylic alcohol, carbonylation to introduce the ester group, hydrogenation to saturate the double bond, and finally, mild oxidation to regenerate the ketone. This logical progression not only enhances safety profiles but also dramatically improves the overall process mass intensity, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation and Selective Oxidation
The core innovation of this synthesis lies in the palladium-catalyzed carbonylation of the halogenated intermediate (Compound IV). In this critical step, the carbon-halogen bond is activated by a Pd(0) species, such as tetrakis(triphenylphosphine)palladium or Pd(dppf)Cl2, facilitating the oxidative addition. Subsequent coordination and insertion of carbon monoxide gas, maintained at pressures between 1 atm and 10 atm, generates an acyl-palladium complex. Methanolysis or ethanolysis of this intermediate then releases the desired ester (Compound V) while regenerating the active catalyst. This mechanism allows for the precise installation of the carboxylate group at the 3-position of the tetrahydropyran ring with high regioselectivity. The reaction conditions are remarkably mild, typically proceeding at temperatures between 40°C and 78°C, which minimizes side reactions and decomposition of the sensitive heterocyclic core.
Following carbonylation, the process requires careful management of oxidation states to achieve the final 5-oxo structure. The double bond in Compound V is first reduced via catalytic hydrogenation using Pd/C or Raney Nickel under mild hydrogen pressure (1 atm to 10 atm) to yield the saturated alcohol (Compound VI). The final transformation involves the oxidation of the secondary hydroxyl group to a ketone. The patent demonstrates versatility here, allowing for oxidants such as 2-iodoxybenzoic acid (IBX), pyridinium chlorochromate (PCC), or Dess-Martin periodinane. For instance, using IBX in acetonitrile at 70°C provides a clean conversion without over-oxidation or ring degradation. This control over the oxidation step is vital for maintaining high purity specifications, ensuring the intermediate meets the stringent quality standards required for downstream coupling reactions in drug synthesis.
How to Synthesize 5-Oxo-Tetrahydropyran-3-Carboxylic Ester Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the halogenation of 2H-pyran-3,5-dione using reagents like carbon tetrabromide or oxalyl chloride at temperatures ranging from -60°C to 60°C. Following isolation, the halo-enone is reduced to the allylic alcohol using hydride sources such as lithium aluminum hydride or sodium borohydride. The subsequent carbonylation step is the technological centerpiece, requiring an inert atmosphere and controlled CO pressure. Detailed operational procedures, including specific workup protocols and purification methods like column chromatography or crystallization, are essential for reproducibility. The standardized synthesis steps below outline the critical parameters derived from the patent examples to ensure successful replication in a pilot or production setting.
- Halogenation of 2H-pyran-3,5-dione using agents like CBr4 or oxalyl chloride to form the halo-enone intermediate.
- Selective reduction of the ketone to an allylic alcohol followed by Palladium-catalyzed carbonylation with CO to introduce the ester group.
- Catalytic hydrogenation of the double bond and final oxidation of the hydroxyl group to yield the target 5-oxo-tetrahydropyran-3-carboxylic ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical superiority. The elimination of tri-n-butyltin hydride and benzene removes significant liability and waste disposal costs associated with hazardous material handling. By replacing these dangerous reagents with catalytic systems and safer solvents like methanol, ethanol, and THF, the process aligns perfectly with modern green chemistry principles and environmental compliance regulations. This shift drastically simplifies the waste treatment workflow, reducing the burden on environmental health and safety departments. Furthermore, the use of commodity chemicals like carbon monoxide and standard palladium catalysts ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that often plague specialty reagent-dependent processes.
- Cost Reduction in Manufacturing: The new route achieves a substantial increase in overall yield compared to the legacy 7.07 percent benchmark, directly translating to lower cost of goods sold (COGS). By avoiding expensive and toxic radical mediators like diiodomethane and tin hydrides, the raw material costs are significantly optimized. Additionally, the simplified purification requirements resulting from cleaner reaction profiles reduce solvent consumption and processing time. These factors collectively drive down the manufacturing cost per kilogram, providing a competitive pricing advantage for bulk purchasers of this key pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on robust, well-established chemical transformations such as hydrogenation and carbonylation ensures high process reliability and consistency. Unlike the sensitive radical reactions in the old method, which are prone to variability, this catalytic approach offers predictable kinetics and scalability. This stability allows for more accurate production planning and shorter lead times for high-purity pharmaceutical intermediates. Suppliers can maintain consistent inventory levels, ensuring that downstream drug manufacturers face no interruptions in their own production schedules due to intermediate shortages.
- Scalability and Environmental Compliance: The mild reaction conditions, particularly the moderate temperatures and pressures used in the carbonylation and hydrogenation steps, make this process highly amenable to commercial scale-up from 100 kgs to 100 MT annual production. The absence of highly exothermic or explosive reagents reduces the engineering controls required for large reactors, lowering capital expenditure for facility upgrades. Moreover, the reduced toxicity profile facilitates easier regulatory approval for manufacturing sites, ensuring long-term operational continuity without the threat of environmental shutdowns or strict emission limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-oxo-tetrahydropyran-3-carboxylic ester. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope and product utility. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific drug development pipelines.
Q: What are the key advantages of this new synthesis route over prior art?
A: The new route eliminates highly toxic reagents like tri-n-butyltin hydride and benzene, replacing them with safer catalytic systems. It significantly improves total yield compared to the 7.07% yield of previous methods and uses milder reaction conditions suitable for industrial scale-up.
Q: Which catalysts are preferred for the carbonylation step?
A: The patent specifies effective catalysts including tetrakis(triphenylphosphine)palladium, bis(triphenylphosphine)palladium dichloride, and [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride, operating under CO pressures of 1 to 10 atm.
Q: Is this intermediate suitable for oncology drug development?
A: Yes, the resulting 5-oxo-tetrahydropyran-3-carboxylic ester is a verified key intermediate for synthesizing mTOR kinase inhibitors and microsomal prostaglandin E2 synthetase-1 inhibitors, which are critical in antitumor research.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Oxo-Tetrahydropyran-3-Carboxylic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering 5-oxo-tetrahydropyran-3-carboxylic ester with stringent purity specifications, utilizing advanced analytical techniques in our rigorous QC labs to guarantee batch-to-batch consistency. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, including high-pressure carbonylation and controlled hydrogenation, safely and efficiently.
We invite you to collaborate with us to optimize your supply chain for oncology and anti-inflammatory drug projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D goals and commercial timelines effectively.
