Advanced Solvent-Free Manufacturing of DWP205190 for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, a trend vividly exemplified by the innovative synthesis method detailed in patent CN110526856B. This pivotal intellectual property introduces a novel, solvent-free melting synthesis route for DWP205190, a potent anti-inflammatory inhibitor that combines the structural motifs of rolipram and thalidomide. Unlike traditional multi-step protocols that rely heavily on volatile organic compounds and transition metal catalysts, this breakthrough methodology utilizes a direct condensation reaction under thermal conditions, effectively bypassing the need for external catalytic agents. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, offering a streamlined pathway to access this high-value isoindolinone derivative. The strategic implementation of this technology not only aligns with stringent global environmental regulations but also promises substantial improvements in final product quality and process safety. By eliminating solvent residues at the source, manufacturers can achieve higher purity profiles essential for downstream API synthesis, thereby reducing the burden on quality control laboratories.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic intermediates like DWP205190 has been plagued by inefficient, multi-step sequences that impose heavy logistical and financial burdens on production facilities. Conventional literature describes routes involving the reduction of substituted N-phenylphthalimide or intermolecular coupling reactions that necessitate the use of large volumes of organic solvents and expensive catalysts. These traditional approaches often suffer from low overall yields due to material losses during multiple isolation and purification stages, leading to increased waste generation and higher disposal costs. Furthermore, the reliance on transition metal catalysts introduces the risk of heavy metal contamination, requiring rigorous and costly purification steps to meet pharmaceutical grade specifications. The environmental footprint of these legacy methods is significant, characterized by high E-factors and the emission of volatile organic compounds that complicate regulatory compliance. From a supply chain perspective, the dependency on specialized reagents and complex solvent recovery infrastructure creates bottlenecks that can delay production timelines and increase vulnerability to raw material price fluctuations.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent leverages a solvent-free melting synthesis technique that fundamentally redefines the efficiency of producing this critical pharmaceutical intermediate. By heating the reactants, specifically methyl 2-bromomethyl-benzoate and 3-(cyclopentyloxy)-4-methoxyaniline, directly together without any solvent medium, the reaction proceeds through a clean thermal activation mechanism. This methodology drastically simplifies the work-up procedure, as the absence of solvent eliminates the need for energy-intensive distillation or evaporation steps typically required to isolate the crude product. The process is remarkably robust, operating effectively within a temperature range of 80°C to 180°C, with preferred embodiments showing exceptional performance between 130°C and 160°C. Experimental data from the patent indicates that this streamlined route can achieve yields as high as 97%, a substantial improvement over the modest outputs often seen in solvent-based counterparts. This direct transformation not only accelerates the reaction kinetics but also minimizes the formation of side products associated with solvent participation, resulting in a cleaner reaction profile.
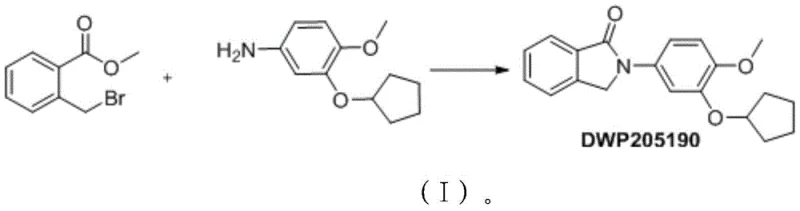
Mechanistic Insights into Thermal Condensation and Cyclization
The core of this innovative process lies in the thermal activation of the nucleophilic substitution and subsequent intramolecular cyclization, driven purely by the kinetic energy provided through heating. In the absence of a solvent, the reactants exist in a molten state where the local concentration is maximized, significantly enhancing the frequency of effective collisions between the amine nucleophile and the benzylic bromide electrophile. The primary amine group of the 3-(cyclopentyloxy)-4-methoxyaniline attacks the methylene carbon of the methyl 2-bromomethyl-benzoate, displacing the bromide ion to form an initial secondary amine intermediate. Following this substitution, the proximity of the ester carbonyl group facilitates an intramolecular nucleophilic attack by the newly formed amine nitrogen, leading to the closure of the five-membered isoindolinone ring. This cascade occurs seamlessly in the melt phase, where the thermal energy overcomes the activation barrier for both the substitution and the cyclization steps without requiring Lewis acid or base catalysts. The lack of solvent molecules means there is no solvation shell to stabilize the transition states differently, often leading to a more direct reaction coordinate that favors the desired product formation.
From an impurity control perspective, this solvent-free mechanism offers distinct advantages by limiting the pathways for solvent-mediated side reactions that often plague traditional syntheses. In conventional solution-phase chemistry, solvents can sometimes participate in transesterification or act as nucleophiles themselves, generating difficult-to-remove byproducts that compromise the purity of the final intermediate. By removing the solvent variable entirely, the impurity profile of DWP205190 becomes much more predictable and manageable, primarily consisting of unreacted starting materials which are easily removed during the recrystallization step. The patent specifies recrystallization using common solvents like ethanol, dichloromethane, or ethyl acetate, which effectively purify the crude melt product to high standards. This controlled environment ensures that the final material meets the stringent purity specifications required for reliable pharmaceutical intermediate supplier applications. Furthermore, the thermal stability of the reactants allows for a wide operating window, providing process engineers with flexibility to optimize for either speed or yield without compromising the structural integrity of the molecule.
How to Synthesize DWP205190 Efficiently
Implementing this solvent-free protocol requires precise control over stoichiometry and thermal parameters to ensure consistent batch-to-batch reproducibility on a commercial scale. The process begins with the accurate weighing of methyl 2-bromomethyl-benzoate and 3-(cyclopentyloxy)-4-methoxyaniline, typically in a molar ratio ranging from 0.5 to 2.0, though equimolar amounts are often preferred for optimal atom economy. Once charged into a suitable reaction vessel equipped with efficient stirring and heating capabilities, the mixture is heated to the target condensation temperature, where it transitions into a homogeneous melt. Maintaining this temperature for a duration of 0.5 to 4 hours allows the reaction to reach completion, after which the mixture is cooled to ambient temperature to solidify the crude product. The detailed standardized synthesis steps, including specific equipment recommendations and safety precautions for handling the molten reactants, are outlined in the guide below.
- Charge a reaction vessel with methyl 2-bromomethyl-benzoate and 3-(cyclopentyloxy)-4-methoxyaniline in a molar ratio ranging from 0.5 to 2.0.
- Heat the mixture to a condensation temperature between 80°C and 180°C, preferably 130°C to 160°C, and stir for 0.5 to 4 hours to facilitate the melting synthesis.
- Cool the reaction mixture to room temperature and recrystallize the crude product using a solvent such as ethanol, dichloromethane, or ethyl acetate to obtain high-purity DWP205190.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of bulk solvents from the reaction phase removes a major cost center associated with purchasing, storing, and disposing of hazardous chemicals, thereby significantly reducing the overall cost of goods sold. Additionally, the simplified workflow reduces the demand for specialized solvent recovery infrastructure, allowing facilities to reallocate capital towards capacity expansion or other value-added activities. The robustness of the melting synthesis also enhances supply chain reliability by reducing the number of critical raw materials required, minimizing the risk of disruptions caused by solvent shortages or price volatility. This streamlined approach supports the commercial scale-up of complex pharmaceutical intermediates by lowering the barrier to entry for large-volume production.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of reaction solvents, which traditionally account for a substantial portion of material costs in fine chemical synthesis. Without the need to purchase, recover, or treat tons of organic solvents per kilogram of product, the variable costs associated with production are drastically lowered. Furthermore, the absence of catalysts eliminates the expense of precious metals and the downstream costs associated with scavenging residual metals to meet regulatory limits. The high yields reported, reaching up to 97%, mean that less raw material is wasted, further driving down the unit cost of the final intermediate. These cumulative savings create a competitive pricing structure that allows suppliers to offer more attractive terms to downstream API manufacturers while maintaining healthy margins.
- Enhanced Supply Chain Reliability: By simplifying the bill of materials to essentially two key organic building blocks, the supply chain becomes far more resilient to external shocks. Traditional methods relying on specific catalysts or exotic solvents are vulnerable to geopolitical supply disruptions, whereas the commoditized nature of the reactants in this new process ensures steady availability. The shorter processing time, ranging from just 0.5 to 4 hours, significantly increases throughput capacity, allowing manufacturers to respond more rapidly to fluctuating market demands. This agility is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug development programs are not delayed by material shortages. The simplified logistics also reduce the complexity of inventory management, freeing up warehouse space and reducing administrative overhead.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the solvent-free melt process make it exceptionally well-suited for scaling from pilot plant to full commercial production. Without the risks associated with large volumes of flammable solvents, the process safety profile is improved, facilitating easier regulatory approval for new manufacturing lines. The reduction in waste generation aligns perfectly with global sustainability goals, helping companies meet their carbon reduction targets and avoid potential environmental fines. This green chemistry approach not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner. The ease of scale-up ensures that supply continuity can be maintained even as demand for DWP205190 grows in the therapeutic market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of the solvent-free melting synthesis for DWP205190 compared to traditional methods?
A: The solvent-free melting synthesis described in patent CN110526856B eliminates the need for organic solvents and catalysts, significantly reducing environmental pollution and production costs while simplifying the purification process and improving overall yield up to 97%.
Q: What are the optimal reaction conditions for maximizing the yield of DWP205190?
A: Optimal results are achieved by maintaining a condensation temperature between 130°C and 160°C for a duration of 0.5 to 4 hours, using a molar ratio of reactants between 0.5 and 2.0, followed by recrystallization from solvents like dichloromethane or ethyl acetate.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the method is highly scalable due to its simple operation, short reaction route, and absence of complex solvent recovery systems, making it ideal for large-scale commercial production of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DWP205190 Supplier
As the global demand for advanced anti-inflammatory therapeutics continues to rise, securing a stable supply of high-quality intermediates like DWP205190 is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging cutting-edge technologies such as the solvent-free melting synthesis to deliver superior value to our partners. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of DWP205190 meets the exacting standards required for clinical and commercial applications. Our commitment to green chemistry not only reduces environmental impact but also translates into cost efficiencies that we pass on to our clients.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how this new synthetic route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for DWP205190. Let us be your trusted partner in navigating the complexities of modern pharmaceutical manufacturing with innovation and reliability.
