Advanced One-Step Asymmetric Synthesis of Chiral Amino Nitriles for Commercial Pharmaceutical Applications
Advanced One-Step Asymmetric Synthesis of Chiral Amino Nitriles for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and efficient pathways to access chiral building blocks, which are essential for the development of next-generation therapeutics. Patent CN102603566B introduces a significant advancement in this domain by detailing a novel synthetic method for producing (S, R)-1-ethylamino phenyl-(2-bromine) phenyl acetonitrile, a critical nitrogenous nitrile compound. This technology leverages a specialized copper-based catalytic system to facilitate an asymmetric cyanation reaction under remarkably mild conditions. Unlike traditional methods that often demand extreme temperatures or hazardous reagents, this approach operates effectively at room temperature, utilizing trimethylsilyl cyanide as a safe cyanide source. The integration of a chiral copper complex not only drives the reaction forward but also imparts the necessary stereochemical integrity to the final product, making it a highly valuable route for generating high-purity pharmaceutical intermediates. For R&D teams and procurement strategists, understanding the nuances of this patent provides a competitive edge in sourcing reliable agrochemical intermediate supplier networks and optimizing API synthesis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alpha-amino nitriles has been fraught with significant technical and operational challenges that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical precursors. Traditional Strecker reactions often rely on the use of hydrogen cyanide (HCN) or alkali metal cyanides, which pose severe safety risks due to their high toxicity and volatility, requiring specialized containment infrastructure and rigorous safety protocols. Furthermore, achieving high enantioselectivity in these reactions typically necessitates the use of expensive noble metal catalysts or stoichiometric amounts of chiral auxiliaries, which drastically increases the cost of goods sold. Many conventional processes also require cryogenic conditions to maintain stereocontrol, leading to substantial energy consumption and limiting the throughput of manufacturing facilities. Additionally, the workup procedures for these older methods often involve complex extraction and purification steps to remove heavy metal residues and racemic byproducts, resulting in lower overall yields and extended production lead times. These factors collectively create a bottleneck for supply chain heads who are tasked with reducing lead time for high-purity pharmaceutical intermediates while maintaining cost efficiency.
The Novel Approach
In stark contrast to these legacy methodologies, the technology disclosed in CN102603566B offers a streamlined, one-step synthesis that fundamentally alters the economic and technical landscape of chiral nitrile production. By employing a catalytic amount of a chiral copper complex derived from (R)-alpha-phenylethylamine and cupric acetate, the process achieves asymmetric induction without the need for stoichiometric chiral reagents. The reaction proceeds smoothly in anhydrous methanol at ambient temperature, eliminating the capital and operational expenditures associated with cooling systems and high-pressure reactors. The use of trimethylsilyl cyanide (TMSCN) serves as a safer, liquid alternative to gaseous HCN, significantly enhancing workplace safety and simplifying handling procedures. Moreover, the reaction design facilitates the direct formation of the target chiral amino nitrile with defined stereochemistry, reducing the burden on downstream purification processes. This novel approach represents a paradigm shift towards greener chemistry, aligning with modern sustainability goals while offering a practical solution for cost reduction in electronic chemical manufacturing and broader fine chemical sectors.
Mechanistic Insights into Copper-Catalyzed Asymmetric Cyanation
The core of this technological breakthrough lies in the sophisticated interplay between the copper center and the chiral ligand, which creates a highly organized transition state for the nucleophilic addition. The catalyst, generated in situ from (R)-alpha-phenylethylamine and cupric acetate, likely forms a coordinatively unsaturated copper species that activates the aldehyde substrate through Lewis acid interactions. Simultaneously, the cyanide source, TMSCN, interacts with the catalyst system to generate a reactive copper-cyanide species or a hypervalent silicon intermediate that delivers the cyanide ion with high facial selectivity. The steric bulk of the phenylethylamine ligand shields one face of the approaching aldehyde, forcing the cyanide attack to occur from the less hindered trajectory, thereby establishing the observed (S) configuration at the newly formed stereocenter. This precise control over the reaction trajectory is critical for minimizing the formation of unwanted diastereomers and enantiomers, which are difficult and costly to separate later in the process.
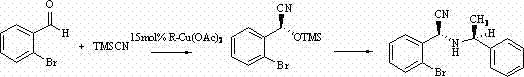
Following the initial cyanation, the mechanism involves a subsequent transformation where the silylated intermediate undergoes aminolysis or ligand exchange to incorporate the amine functionality, ultimately yielding the stable chiral amino nitrile product. The presence of the bromine substituent on the aromatic ring adds an additional layer of complexity, as it can influence the electronic properties of the aldehyde and potentially participate in secondary coordination with the metal center. Understanding these mechanistic details allows process chemists to fine-tune reaction parameters such as solvent polarity and catalyst loading to maximize efficiency. Furthermore, the mechanism suggests that impurity profiles can be tightly controlled by managing the water content and oxygen levels in the reaction vessel, as these factors can degrade the active copper species or hydrolyze the silyl intermediates prematurely. This deep mechanistic understanding is essential for R&D directors aiming to replicate and optimize this synthesis for commercial manufacturing environments.
How to Synthesize (S, R)-1-ethylamino phenyl-(2-bromine) phenyl acetonitrile Efficiently
Executing this synthesis requires strict adherence to anhydrous and oxygen-free conditions to preserve the activity of the copper catalyst and prevent the decomposition of sensitive intermediates. The process begins with the preparation of the reaction mixture in a suitable vessel, such as a two-necked flask, where anhydrous methanol serves as the solvent medium. The substrates, 2-bromobenzaldehyde and trimethylsilyl cyanide, are introduced in specific molar ratios to ensure complete conversion while minimizing side reactions. The addition of the pre-formed or in situ generated chiral copper catalyst is the critical step that initiates the asymmetric transformation. Detailed standard operating procedures for the exact mixing sequences, stirring rates, and monitoring techniques are essential for reproducibility. For a comprehensive guide on the specific experimental steps and purification protocols, please refer to the standardized synthesis instructions below.
- Prepare the reaction mixture by combining anhydrous methanol, 2-bromobenzaldehyde, and trimethylsilyl cyanide (TMSCN) under anhydrous and oxygen-free conditions.
- Add the chiral copper catalyst complex, specifically 15mol% (R)-alpha-phenylethylamine cupric acetate, to the reaction vessel.
- Stir the mixture at room temperature for 3 days, followed by purification via column chromatography using petroleum ether and dichloromethane to isolate the monocrystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical transformation. The shift from hazardous gaseous reagents to liquid TMSCN significantly reduces the regulatory burden and insurance costs associated with handling toxic materials, leading to a safer and more compliant manufacturing environment. The ability to run the reaction at room temperature eliminates the need for specialized cryogenic equipment, allowing existing reactor trains to be utilized more flexibly and reducing capital expenditure requirements for new facility construction. This operational simplicity translates directly into enhanced supply chain reliability, as the process is less susceptible to disruptions caused by utility failures or equipment maintenance issues. Furthermore, the use of copper, an earth-abundant and inexpensive metal, compared to precious metals like palladium or rhodium, provides a inherent buffer against volatile raw material pricing, ensuring more stable long-term costing models for the final API.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction in energy consumption due to ambient temperature operation contribute to a significantly lowered cost base. By avoiding the need for complex cooling infrastructure and reducing the stoichiometric usage of chiral reagents, the overall variable costs per kilogram of product are drastically optimized. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phases required for purification, further driving down waste disposal costs and improving the overall atom economy of the process.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as 2-bromobenzaldehyde and TMSCN ensures a robust supply chain that is less prone to shortages. The mild reaction conditions reduce the risk of thermal runaways or safety incidents that could halt production lines, thereby guaranteeing consistent delivery schedules to downstream customers. This stability is crucial for maintaining continuous manufacturing flows in the highly regulated pharmaceutical sector, where interruptions can have cascading effects on drug availability.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes the number of unit operations required, making the process inherently easier to scale from laboratory to pilot and eventually to full commercial production. The reduced generation of hazardous waste and the avoidance of toxic cyanide gases align with increasingly stringent environmental regulations, facilitating smoother permitting processes and enhancing the corporate sustainability profile. This green chemistry approach not only meets current compliance standards but also future-proofs the manufacturing asset against tightening global environmental directives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for potential partners. Understanding these details is vital for making informed decisions about integrating this pathway into your existing portfolio of fine chemical intermediates.
Q: What is the primary advantage of using the copper-based catalyst in this synthesis?
A: The use of a copper-based chiral catalyst allows the reaction to proceed at room temperature, eliminating the need for energy-intensive cryogenic conditions often required in asymmetric cyanation, thereby simplifying the operational protocol.
Q: How is the stereochemistry controlled in this reaction pathway?
A: Stereocontrol is achieved through the chiral environment provided by the (R)-alpha-phenylethylamine ligand coordinated to the copper center, which directs the nucleophilic attack of the cyanide species to favor the formation of the specific (S, R) diastereomer.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes readily available starting materials and mild reaction conditions. While the patent describes laboratory-scale column chromatography, the one-pot nature of the reaction suggests significant potential for process optimization and scale-up in a commercial setting.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S, R)-1-ethylamino phenyl-(2-bromine) phenyl acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102603566B for the production of high-value chiral intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial realities. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity, assay, and impurity profiles. Our dedication to quality assurance guarantees that every batch of (S, R)-1-ethylamino phenyl-(2-bromine) phenyl acetonitrile supplied meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates the economic viability of this method against your current supply chain. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive efficiency and reliability in your manufacturing operations.
