Revolutionizing Cyclic Amide Production: A Deep Dive into High-Yield Laurolactam Manufacturing
Revolutionizing Cyclic Amide Production: A Deep Dive into High-Yield Laurolactam Manufacturing
The chemical manufacturing landscape for high-performance polyamides is undergoing a significant transformation driven by the innovations detailed in patent CN107438594B. This intellectual property discloses a highly efficient, economical, and industrially advantageous straight-through process for synthesizing cyclic amides, specifically targeting the production of laurolactam, a critical monomer for Nylon-12. Traditional methods for producing these valuable intermediates have long been plagued by complex multi-step procedures, excessive solvent usage, and the generation of hazardous by-products. The disclosed technology offers a paradigm shift by enabling the conversion of corresponding cyclic ketones and hydroxylammonium salts into substantially pure cyclic amides in a single, telescoped operation. For R&D directors and procurement strategists alike, this represents a pivotal opportunity to enhance supply chain resilience while drastically improving the purity profile of essential polymer precursors. By leveraging amphoteric metal oxides in combination with bases, this method achieves high yields without the need for intermediate isolation, setting a new benchmark for sustainable chemical synthesis in the specialty chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing lactams, such as those described in US4054562 and US8309714B2, rely heavily on cumbersome unit operations that inflate both capital expenditure and operational costs. Conventional techniques often necessitate the use of concentrated sulfuric acid at extreme temperatures ranging from 240°C to 1250°C to decompose ammonium sulfate by-products, creating severe safety hazards and energy inefficiencies. Furthermore, prior art methods frequently require the isolation and purification of the oxime intermediate before it can undergo the Beckmann rearrangement. This multi-stage approach not only increases the risk of product loss during transfer and filtration but also demands significant volumes of solvents for extraction and washing. The reliance on strong mineral acids often leads to equipment corrosion and generates substantial quantities of acidic waste, complicating environmental compliance and waste treatment protocols. Additionally, processes utilizing stoichiometric amounts of metal oxides without pH control have been observed to cause polymerization of the final lactam product due to excess metal salt accumulation, resulting in unacceptable purity levels and reduced overall yield.
The Novel Approach
In stark contrast to these legacy methodologies, the process defined in CN107438594B introduces a streamlined straight-through chemistry that minimizes solvent usage and eliminates intermediate purification steps entirely. This novel approach utilizes a synergistic combination of amphoteric metal oxides, such as zinc oxide, and alkali bases, like sodium carbonate, to facilitate the reaction in situ. By carefully balancing the ratio of the metal oxide to the base, the process generates the necessary catalytic metal salt species internally while simultaneously neutralizing excess acidity that would otherwise trigger polymerization. This allows for the direct conversion of cyclododecanone to laurolactam with exceptional efficiency. The elimination of isolation steps means that the reaction mass proceeds directly from oxime formation to rearrangement after simple azeotropic water removal. This continuity not only accelerates the production timeline but also ensures that the final product is obtained in substantially pure form with yields reaching up to 96.1%. Such a reduction in unit operations translates directly to lower processing losses and a significantly smaller environmental footprint, making it an ideal solution for modern, green chemical manufacturing facilities.
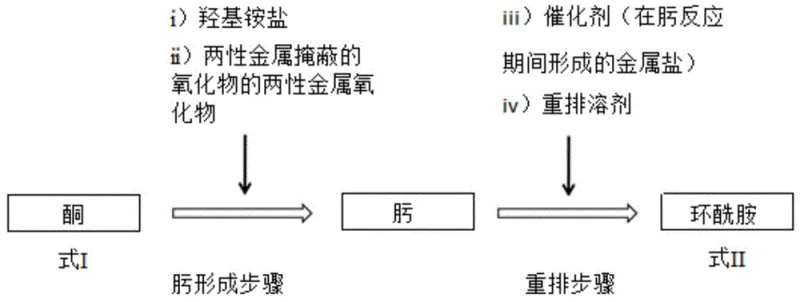
Mechanistic Insights into Amphoteric Oxide-Catalyzed Beckmann Rearrangement
The core mechanistic breakthrough of this technology lies in the precise control of the reaction environment during the Beckmann rearrangement phase. In traditional zinc chloride-catalyzed systems, the presence of excess Lewis acid often promotes unwanted side reactions, including the polymerization of the lactam ring. The inventors discovered that by reacting hydroxylamine hydrochloride with zinc oxide in the presence of a base like sodium carbonate, the system generates zinc chloride in situ as a by-product. However, unlike conventional methods where this salt accumulates unchecked, the added base neutralizes the hydrochloric acid component of the hydroxylamine salt, preventing the medium from becoming overly acidic. This delicate balance ensures that there is sufficient catalytic activity to drive the rearrangement of the oxime to the amide without creating conditions favorable for polymerization. The amphoteric nature of the metal oxide allows it to react with the acid to form the active catalyst while the base modulates the pH to protect the product integrity. This dual-function mechanism effectively transforms a potential impurity source into a controlled catalytic driver, ensuring that the reaction proceeds with high selectivity towards the desired cyclic amide structure.
Furthermore, the impurity control mechanism is reinforced by the avoidance of high-temperature decomposition steps associated with sulfate-based processes. By maintaining reaction temperatures around 80°C to 112°C during the azeotropic distillation and rearrangement phases, thermal degradation of the sensitive oxime intermediate is prevented. The use of toluene for water removal ensures that the reaction environment remains anhydrous prior to the addition of the rearrangement solvent, acetonitrile, which further stabilizes the transition state. This meticulous control over reaction parameters results in a product with GC purity exceeding 99.7%, significantly reducing the burden on downstream purification units. For pharmaceutical and high-performance polymer applications, where trace impurities can compromise material properties or regulatory compliance, this level of intrinsic purity is a decisive advantage. The ability to generate high-quality laurolactam without extensive recrystallization or chromatography underscores the robustness of this catalytic system for industrial scale-up.
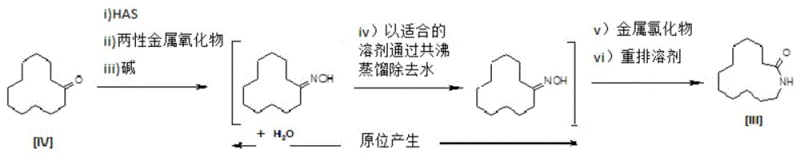
How to Synthesize Laurolactam Efficiently
The implementation of this straight-through synthesis route requires careful attention to reagent stoichiometry and temperature profiling to maximize the benefits of the amphoteric catalytic system. The process begins with the melting of the cyclic ketone substrate, followed by the sequential addition of hydroxylamine hydrochloride, zinc oxide, and sodium carbonate. Maintaining the initial reaction temperature at approximately 85°C is critical to ensure complete conversion to the oxime intermediate without premature degradation. Once the ketone conversion reaches the desired threshold, typically around 90%, the process transitions immediately to water removal via azeotropic distillation using toluene. This step is vital as residual water can inhibit the subsequent rearrangement reaction. Following dehydration, the introduction of acetonitrile and heating to 80°C initiates the rearrangement, driven by the in situ generated zinc chloride catalyst. The detailed standardized synthesis steps for replicating this high-efficiency protocol are outlined below.
- Contact the cyclic ketone with hydroxylamine hydrochloride, zinc oxide, and sodium carbonate at approximately 85°C to form the oxime intermediate in situ.
- Remove water formed during oximation via azeotropic distillation using toluene without isolating the intermediate.
- Add acetonitrile as a rearrangement solvent and heat to approximately 80°C to catalyze the Beckmann rearrangement into the final lactam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers profound strategic advantages that extend beyond simple yield improvements. The fundamental redesign of the synthesis pathway addresses several critical pain points associated with traditional lactam manufacturing, particularly regarding cost structure and operational reliability. By collapsing multiple discrete reaction and isolation stages into a single continuous workflow, the technology drastically reduces the requirement for intermediate storage tanks, filtration equipment, and solvent recovery units. This simplification of the plant infrastructure lowers capital investment barriers and reduces the complexity of daily operations, allowing for more agile response to market demand fluctuations. Moreover, the significant reduction in solvent consumption directly correlates with lower raw material procurement costs and diminished expenses related to hazardous waste disposal. The ability to produce high-purity material with minimal downstream processing further enhances the economic viability of the operation, ensuring that every kilogram of input material is converted into saleable product with maximum efficiency.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps removes the need for expensive filtration and drying operations, which are traditionally resource-intensive. By avoiding the use of concentrated sulfuric acid and the subsequent high-temperature decomposition of ammonium sulfate, the process sidesteps the heavy energy costs and specialized corrosion-resistant equipment required by legacy methods. The in situ generation of the catalyst from inexpensive zinc oxide and hydroxylamine salts further optimizes the bill of materials, removing the dependency on costly pre-formed Lewis acid catalysts. These cumulative efficiencies result in a substantially leaner cost structure, allowing manufacturers to maintain competitive pricing even in volatile raw material markets while preserving healthy profit margins through improved process economics.
- Enhanced Supply Chain Reliability: Simplifying the manufacturing process inherently reduces the number of potential failure points within the supply chain. Fewer unit operations mean less handling of hazardous intermediates, which decreases the risk of accidents, spills, or containment breaches that could halt production. The robustness of the amphoteric oxide system ensures consistent batch-to-batch quality, reducing the likelihood of off-spec material that would otherwise require reprocessing or scrapping. This reliability is crucial for maintaining uninterrupted supply to downstream polymer producers who depend on just-in-time delivery schedules. Furthermore, the use of widely available and stable reagents like zinc oxide and sodium carbonate mitigates the risk of supply disruptions associated with more exotic or regulated catalysts, ensuring long-term continuity of supply for critical nylon-12 monomers.
- Scalability and Environmental Compliance: The straight-through nature of this chemistry is inherently scalable, as it avoids the bottlenecks typically associated with solid-liquid separations and multi-stage extractions. This facilitates a smoother transition from pilot plant trials to full commercial production volumes without the need for disproportionate increases in facility footprint. From an environmental perspective, the drastic reduction in solvent usage and the absence of sulfuric acid waste streams significantly lower the ecological impact of the manufacturing process. This alignment with green chemistry principles simplifies regulatory compliance and reduces the burden on wastewater treatment facilities. Companies adopting this technology can leverage these sustainability credentials to meet increasingly stringent corporate social responsibility goals and satisfy the growing demand for eco-friendly polymer supply chains from end-users in the automotive and consumer goods sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced lactam synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios. The answers highlight the specific mechanisms by which the invention overcomes historical limitations related to yield, purity, and operational safety.
Q: How does this process prevent polymerization during lactam formation?
A: The process utilizes a precise combination of amphoteric metal oxides and bases to control the acidity level. This prevents the excess metal salt formation that typically causes polymerization in traditional zinc chloride-catalyzed methods.
Q: What are the yield advantages over conventional sulfuric acid methods?
A: Unlike conventional methods requiring high temperatures and strong acids which degrade product quality, this straight-through process achieves yields exceeding 96% with purity greater than 99% by avoiding harsh decomposition conditions.
Q: Is intermediate purification required for the oxime?
A: No, the key innovation is the elimination of isolation steps. The oxime intermediate undergoes in situ rearrangement immediately after water removal, significantly reducing unit operations and processing losses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Laurolactam Supplier
As the global demand for high-performance polyamides continues to surge, securing a dependable source of high-purity monomers like laurolactam has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry evolution, leveraging cutting-edge synthetic methodologies to deliver superior chemical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for nylon-12 polymerization and pharmaceutical applications. Our commitment to quality assurance means that you receive material that is ready for immediate use, minimizing your internal testing burdens and accelerating your own production cycles.
We invite you to collaborate with us to explore how this innovative straight-through synthesis can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible benefits of partnering with a leader in advanced chemical manufacturing. Let us help you achieve greater efficiency and reliability in your lactam sourcing strategy.
