Advanced Synthesis of Deuterated Ibrutinostat Racemate for Commercial Pharmaceutical Production
The pharmaceutical industry is constantly seeking robust methodologies to enhance the metabolic stability of therapeutic agents, and the synthesis process detailed in patent CN109096243B represents a significant advancement in this domain. This intellectual property discloses a novel, metal-free pathway for the production of deuterated ibrutinostat racemate, a critical intermediate for treating Gaucher disease type I. By strategically replacing hydrogen atoms with deuterium on metabolically vulnerable sites, the resulting compound exhibits improved pharmacokinetic profiles compared to its non-deuterated counterparts. The core innovation lies in the construction of the key intermediate 2,3-dihydro-1,4-benzodioxan-2,2,3,3-d4-6-aldehyde without relying on toxic transition metals. This approach not only aligns with green chemistry principles but also addresses the stringent purity requirements demanded by global regulatory bodies for orphan drug applications. 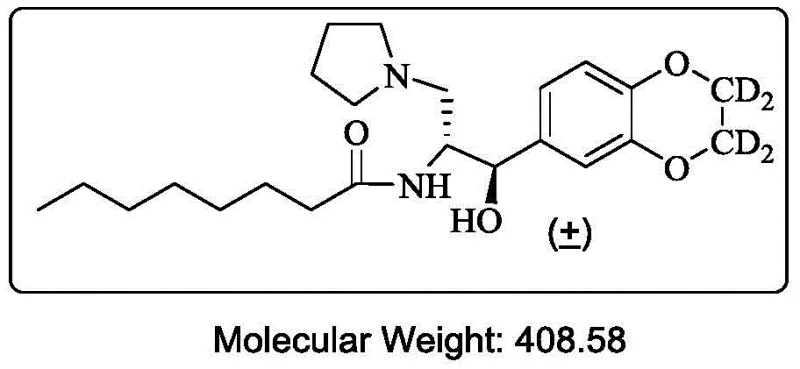
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar amino-alcohol intermediates has heavily relied on the Henry reaction, which typically necessitates the use of copper catalysts to facilitate the nitroaldol condensation. While effective in laboratory settings, the presence of copper introduces substantial downstream processing challenges, particularly regarding the removal of residual heavy metals to parts-per-million levels. Furthermore, the nucleophilic substrates required for traditional Henry reactions are often not readily available on a commercial scale, leading to supply chain bottlenecks and increased raw material costs. The multi-step purification processes needed to eliminate metal traces often result in significant yield losses and generate hazardous waste streams that complicate environmental compliance. These factors collectively hinder the economic viability and scalability of conventional routes when applied to the industrial manufacturing of high-value pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN109096243B circumvents these issues by employing isocyanamide as a nucleophilic reagent condensed with a key aldehyde intermediate under mild alkaline conditions. This strategic shift eliminates the requirement for any transition metal catalysis, thereby simplifying the workup procedure and drastically reducing the burden on quality control laboratories. The process utilizes easily obtainable starting materials, such as m-nitrobenzenesulfonyl chloride and pyrrolidine derivatives, which ensures a consistent and reliable supply chain for continuous production. Additionally, the reaction conditions are optimized for ambient or near-ambient temperatures in several steps, lowering energy consumption and enhancing operational safety. This streamlined workflow offers a clear pathway for cost reduction in pharmaceutical intermediates manufacturing while maintaining high stereochemical integrity. 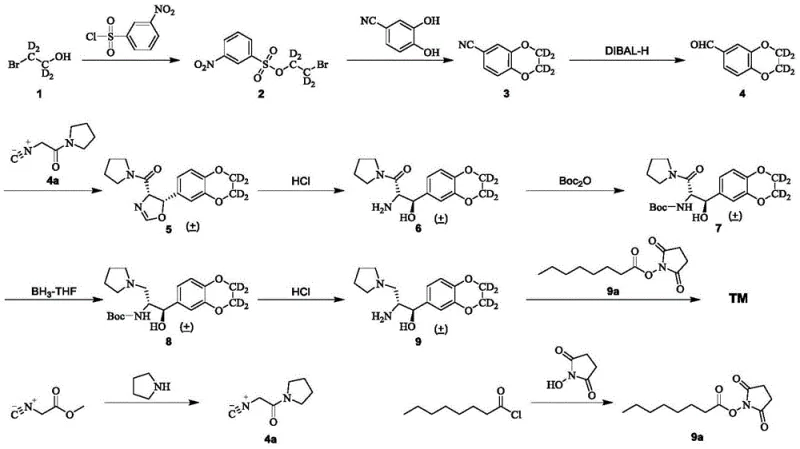
Mechanistic Insights into Isocyanamide Condensation
The heart of this synthetic innovation is the condensation reaction between the deuterated aldehyde intermediate and the isocyanamide derivative, which proceeds through a unique cyclization mechanism to form the oxazoline ring system. Under alkaline conditions, the isocyanamide acts as a carbon nucleophile, attacking the carbonyl carbon of the aldehyde to initiate the bond formation without the need for Lewis acid activation. This mechanism is particularly advantageous because it avoids the formation of metal-complexed by-products that are notoriously difficult to separate from the desired chiral alcohol. The use of potassium hydroxide in methanol provides a homogeneous reaction environment that promotes high conversion rates while minimizing side reactions such as aldol self-condensation. Detailed analysis of the reaction kinetics suggests that the steric environment created by the deuterated benzodioxan ring further directs the stereoselectivity of the addition. 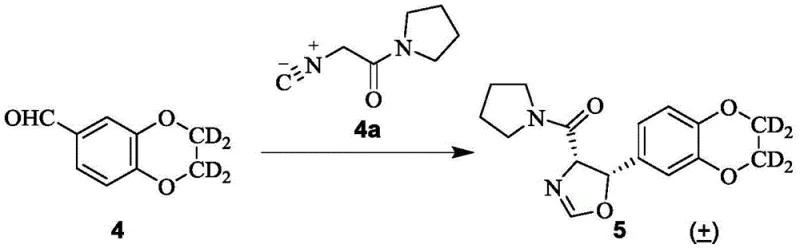
Impurity control is inherently built into this mechanistic design, as the absence of transition metals removes an entire class of potential inorganic contaminants from the process stream. The subsequent hydrolysis steps, utilizing concentrated hydrochloric acid, are designed to open the oxazoline ring selectively without affecting the sensitive deuterium labels on the benzodioxan moiety. This selectivity is crucial for maintaining the isotopic enrichment of the final product, which is directly correlated with its metabolic stability in vivo. Furthermore, the protection and deprotection strategies employed, such as the use of Boc groups, are orthogonal to the reaction conditions, allowing for precise manipulation of the amine functionality. The cumulative effect of these mechanistic choices is a process that delivers high-purity intermediates with a significantly simplified impurity profile compared to metal-catalyzed alternatives.
How to Synthesize Deuterated Ibrutinostat Efficiently
The synthesis of this complex deuterated molecule involves a carefully orchestrated sequence of nine distinct chemical transformations, each optimized for yield and purity. The process begins with the protection of a deuterated ethanolamine derivative, followed by cyclization to form the core benzodioxan structure, and concludes with the coupling of the fatty acid side chain. Operators must adhere strictly to temperature controls, particularly during the reduction steps involving DIBAL-H and borane, to prevent over-reduction or decomposition of sensitive intermediates. The detailed standardized synthesis steps见下方的指南 ensure reproducibility and safety during scale-up operations.
- Preparation of deuterated sulfonate intermediate via reaction with m-nitrobenzenesulfonyl chloride.
- Cyclization to form the deuterated benzodioxan core using 3,4-dihydroxybenzonitrile.
- Reduction of nitrile to aldehyde followed by isocyanamide condensation to build the chiral center.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented route offers substantial strategic benefits by decoupling production from the volatility of transition metal markets and the regulatory scrutiny associated with heavy metal residues. The elimination of copper catalysts means that manufacturers can bypass the expensive and time-consuming steps typically required for metal scavenging, leading to a more streamlined production timeline. This efficiency translates directly into enhanced supply chain reliability, as the process is less susceptible to delays caused by specialized reagent shortages or complex purification bottlenecks. Moreover, the use of commodity chemicals as starting materials ensures that the supply base is broad and competitive, mitigating the risk of single-source dependency. These factors collectively contribute to a more resilient manufacturing ecosystem capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the synthesis pathway fundamentally alters the cost structure by eliminating the need for expensive metal scavengers and specialized filtration equipment. Without the requirement to test for and remove trace copper to ppm levels, the analytical workload is significantly reduced, freeing up quality control resources for other critical parameters. The simplified workup procedures also result in higher overall throughput, as less time is spent on purification columns and extractions designed specifically for metal removal. Consequently, the operational expenditure per kilogram of product is lowered, providing a competitive edge in pricing for bulk active pharmaceutical ingredients. This economic efficiency is achieved without compromising the quality or potency of the final deuterated therapeutic agent.
- Enhanced Supply Chain Reliability: By relying on readily available organic building blocks rather than specialized organometallic reagents, the supply chain becomes inherently more robust and less prone to disruption. The raw materials identified in this process, such as m-nitrobenzenesulfonyl chloride and standard solvents like dichloromethane and methanol, are produced by multiple global suppliers, ensuring continuity of supply even during market fluctuations. This diversification of the vendor base reduces the lead time for high-purity intermediates, allowing manufacturers to respond more agilely to changes in demand. Furthermore, the stability of the intermediates allows for safer storage and transportation, minimizing the risk of degradation during logistics. Such reliability is paramount for maintaining the production schedules of life-saving medications like those used for Gaucher disease.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to large-scale stainless steel reactors. The absence of pyrophoric reagents or extreme high-pressure conditions enhances operational safety, reducing the insurance and infrastructure costs associated with hazardous chemical handling. From an environmental standpoint, the metal-free nature of the synthesis significantly reduces the toxicity of the waste stream, simplifying effluent treatment and disposal protocols. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals. The ability to scale from pilot batches to commercial tonnage without re-optimizing the core chemistry ensures a seamless transition to full-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in a GMP environment. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines.
Q: How does this process avoid heavy metal contamination?
A: Unlike conventional methods utilizing copper-catalyzed Henry reactions, this patented route employs isocyanamide condensation under alkaline conditions, completely eliminating the need for transition metal catalysts and subsequent scavenging steps.
Q: Are the raw materials for this synthesis readily available?
A: Yes, the process utilizes commercially accessible starting materials such as m-nitrobenzenesulfonyl chloride and pyrrolidine derivatives, ensuring a stable supply chain for large-scale manufacturing.
Q: What are the purity advantages of this deuterated route?
A: The metal-free nature of the synthesis significantly reduces the risk of inorganic impurities, while the specific reaction conditions allow for effective control of organic by-products, facilitating easier purification to high-purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Ibrutinostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercially viable realities for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this metal-free route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to handle the specific analytical challenges of deuterated compounds. Our commitment to quality ensures that every batch of deuterated ibrutinostat intermediate meets the highest international standards for safety and efficacy. We are dedicated to supporting your R&D and commercialization goals with unmatched technical expertise and manufacturing capacity.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your production volume. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique requirements. Together, we can accelerate the delivery of next-generation therapies to patients who need them most, leveraging the power of advanced organic synthesis and reliable manufacturing excellence.
