Industrial Scale-Up of Carbon-Losing Cholestanal: A Breakthrough in Bile Acid Derivative Manufacturing
The pharmaceutical industry is constantly seeking efficient routes to synthesize complex bile acid derivatives, which serve as pivotal scaffolds for treating metabolic and infectious diseases. Patent CN113135971A introduces a groundbreaking methodology for the preparation of carbon-losing cholestanal, a specialized nor-bile acid derivative, utilizing duck cholic acid as a readily available starting material. This innovation addresses long-standing challenges in side-chain degradation, offering a pathway that significantly enhances yield and purity while leveraging low-cost industrial waste streams. For R&D directors and procurement specialists, this technology represents a strategic opportunity to optimize the supply chain for high-value pharmaceutical intermediates. The process circumvents the harsh conditions and low efficiencies associated with historical methods, positioning itself as a robust solution for commercial scale-up of complex API intermediates.
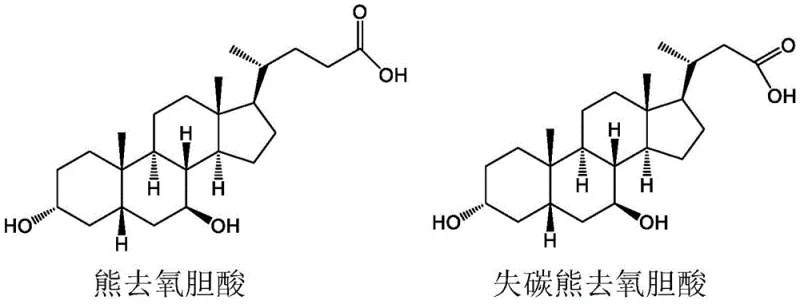
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nor-bile acids involved the degradation of natural cholic acids using strong oxidizing agents like potassium permanganate. As illustrated in the prior art reaction schemes, this traditional approach suffers from severe drawbacks regarding selectivity and operational complexity. The aggressive nature of potassium permanganate often leads to over-oxidation and the formation of complex byproduct mixtures, necessitating rigorous purification steps such as column chromatography combined with recrystallization. Consequently, the total yield for these conventional processes rarely exceeds 50%, making them economically unviable for large-scale manufacturing. Furthermore, the management of manganese-containing waste streams poses significant environmental compliance burdens, complicating the cost reduction in pharmaceutical intermediate manufacturing efforts for modern facilities.
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a controlled oxidative degradation using sodium hypochlorite or periodate under mild alkaline conditions. This novel route selectively cleaves the side chain of duck cholic acid to generate the target aldehyde functionality without compromising the integrity of the steroid nucleus. The process operates at moderate temperatures ranging from 45°C to 60°C and utilizes common solvents like butyl acetate, facilitating straightforward phase separation workups. By eliminating the need for chromatographic purification and achieving isolated yields of over 90%, this approach drastically simplifies the production workflow. This efficiency gain is critical for establishing a reliable pharmaceutical intermediate supplier network capable of meeting global demand without the bottlenecks of legacy chemistry.
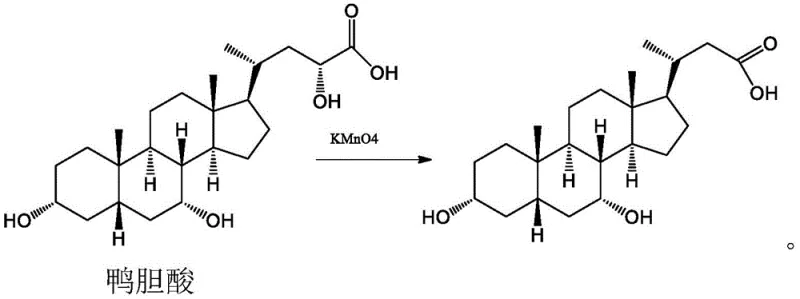
Mechanistic Insights into Oxidative Side-Chain Degradation
The core of this technological advancement lies in the precise control of the oxidative cleavage mechanism. Unlike non-selective oxidants, the sodium hypochlorite system functions through a specific interaction with the vicinal diol or activated methylene groups on the side chain of the duck cholic acid substrate. The reaction proceeds via the formation of hypochlorite esters or chloramine intermediates, which subsequently undergo beta-elimination or hydrolysis to sever the carbon-carbon bond. This mechanism is highly dependent on the pH and temperature parameters, which are tightly regulated between 45°C and 60°C to prevent over-oxidation to the carboxylic acid or degradation of the steroid core. Understanding this mechanistic nuance allows process chemists to fine-tune the stoichiometry, typically maintaining a molar ratio of duck cholic acid to sodium hypochlorite around 1:2.5, ensuring complete conversion while minimizing reagent waste.
Impurity control is another critical aspect where this new method excels. In traditional syntheses, the presence of multiple hydroxyl groups on the bile acid scaffold often leads to competing oxidation reactions, generating ketones or lactones that are difficult to separate. The novel protocol mitigates this by leveraging the specific reactivity of the side chain in the presence of the chosen solvent system, such as butyl acetate. The biphasic nature of the reaction mixture helps in extracting the product as it forms, shielding it from further degradation in the aqueous oxidative phase. This inherent selectivity results in a crude product with HPLC purity exceeding 95%, significantly reducing the burden on downstream purification units and ensuring a consistent quality profile essential for high-purity pharmaceutical intermediates.
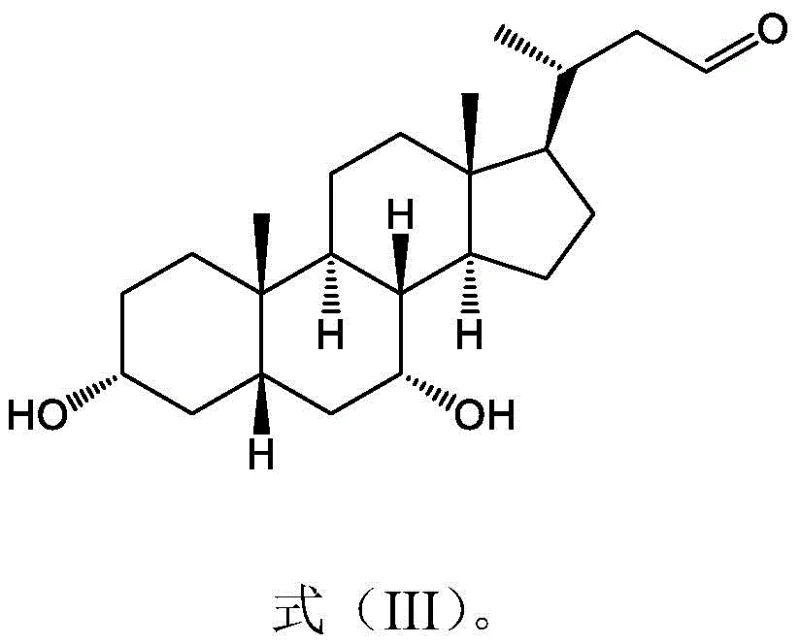
How to Synthesize Carbon-Losing Cholestanal Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing this valuable intermediate. The process begins with the dissolution of duck cholic acid in a sodium hydroxide solution, followed by the addition of an organic solvent to create a biphasic system. The degradation agent is then introduced under controlled thermal conditions to effect the side-chain cleavage. Detailed standard operating procedures regarding reagent grades, mixing speeds, and quenching protocols are essential for replicating the high yields reported in the examples. For a comprehensive guide on the exact step-by-step execution, including specific workup and purification details, please refer to the technical documentation below.
- Dissolve duck cholic acid in an alkaline solution (NaOH) and add an organic solvent such as butyl acetate.
- Introduce the degradation agent, specifically sodium hypochlorite solution, maintaining the temperature between 45-60°C for 30-40 minutes.
- Separate the organic layer, wash sequentially with sodium sulfite, sodium hydroxide, and brine, then concentrate to obtain the product with >95% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the shift to this novel synthesis route offers profound benefits for supply chain stability and cost management. The primary advantage stems from the utilization of duck cholic acid, an underutilized byproduct of the poultry industry, which serves as a far more economical feedstock compared to purified cholic acid or chenodeoxycholic acid. This raw material availability ensures a resilient supply base that is less susceptible to the fluctuations often seen in specialty botanical or fermentation-derived starting materials. Moreover, the drastic improvement in reaction yield—from roughly 50% in older methods to over 90% in this new process—directly translates to substantial material savings and reduced waste disposal costs. These factors collectively contribute to a more competitive pricing structure for the final active pharmaceutical ingredients derived from this intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like potassium permanganate, coupled with the removal of resource-intensive chromatography steps, significantly lowers the operational expenditure. The simplified workup involving basic liquid-liquid extraction and crystallization reduces solvent consumption and labor hours, driving down the overall cost of goods sold without compromising quality standards.
- Enhanced Supply Chain Reliability: By relying on duck cholic acid, which is available in large volumes as an industrial waste product, manufacturers can secure a long-term, stable supply of raw materials. This reduces the risk of production stoppages due to raw material shortages and allows for better capacity planning, ensuring reducing lead time for high-purity pharmaceutical intermediates to meet market demands efficiently.
- Scalability and Environmental Compliance: The process operates under mild conditions and generates less hazardous waste compared to heavy metal-based oxidations. This aligns with modern green chemistry principles and simplifies regulatory compliance regarding effluent treatment. The robustness of the reaction conditions also facilitates easy scale-up from pilot batches to multi-ton commercial production, supporting the growing demand for bile acid-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of carbon-losing cholestanal. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the feasibility and advantages of this synthetic route for industry stakeholders.
Q: What are the advantages of using duck cholic acid over traditional cholic acid for this synthesis?
A: Duck cholic acid is an abundant industrial waste byproduct with a lower cost profile compared to purified cholic acid. Its specific side-chain structure allows for more direct oxidative degradation pathways, significantly improving overall process economics.
Q: How does the new sodium hypochlorite method compare to the traditional potassium permanganate oxidation?
A: The traditional KMnO4 method often results in complex byproduct mixtures requiring difficult chromatographic purification, yielding only about 50%. The novel sodium hypochlorite method achieves yields exceeding 90% with simpler workup procedures involving phase separation and washing.
Q: What is the primary pharmaceutical application of carbon-losing cholestanal?
A: This compound exhibits potent bacteriostatic activity, particularly against Clostridium difficile. It serves as a critical intermediate for synthesizing nor-bile acid derivatives used in treating liver conditions and as potential antibacterial agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbon-Losing Cholestanal Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented technology in the realm of bile acid derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carbon-losing cholestanal meets the highest international standards required for pharmaceutical applications.
We invite global partners to collaborate with us to leverage this cost-effective and high-yielding synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the next generation of antibacterial and metabolic therapies.
