Advanced Synthesis of Vinyl Tris(2,2,2-trifluoro) Ethoxyl Silane for Industrial Scale-up
The chemical industry is constantly seeking robust methodologies to synthesize high-performance fluorinated materials that combine the thermal stability of silicones with the surface properties of fluorocarbons. Patent CN103012462A introduces a significant advancement in this domain by detailing a preparation method for vinyl tris(2,2,2-trifluoro) ethoxyl silane, a critical monomer for next-generation fluorosilicone polymers. This synthesis leverages a direct alcoholysis reaction between vinyl trichlorosilane and trifluoroethanol, utilizing dichloromethane as a solvent under strictly controlled inert atmospheric conditions. The innovation lies not merely in the chemical transformation itself, but in the precise optimization of reaction parameters—such as temperature, gas flow rates, and reagent addition speeds—that collectively enable a scalable, high-yield process suitable for industrial manufacturing. For R&D directors and procurement specialists alike, this patent represents a viable pathway to producing specialized intermediates that drive performance in coatings, agrochemicals, and advanced material science applications without the complexity of multi-step catalytic cycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for alkoxy silanes often suffer from significant drawbacks that hinder their economic viability and operational safety on a large scale. Many conventional methods rely on harsh reaction conditions that can degrade sensitive functional groups, such as the vinyl moiety, leading to polymerization side reactions or the formation of complex impurity profiles that are difficult to separate. Furthermore, older protocols frequently necessitate the use of expensive transition metal catalysts or Lewis acids, which not only inflate the raw material costs but also introduce stringent purification requirements to meet regulatory standards for heavy metal residues in final products. The management of exothermic heat during the alcoholysis of chlorosilanes is another critical challenge; without precise control, rapid heat generation can lead to runaway reactions, compromising both safety and product consistency. Additionally, the volatility and moisture sensitivity of chlorosilane precursors demand sophisticated equipment setups that many standard facilities may lack, creating a barrier to entry for reliable production of these high-value intermediates.
The Novel Approach
The methodology outlined in the patent data offers a transformative solution by employing a mild, catalyst-free alcoholysis strategy that directly addresses the inefficiencies of legacy processes. By utilizing dichloromethane as a solvent medium, the process effectively moderates the reaction kinetics, allowing for a controlled interaction between the vinyl trichlorosilane and trifluoroethanol at relatively low temperatures ranging from 20°C to 40°C. This approach minimizes thermal stress on the vinyl group, preserving its integrity for subsequent polymerization applications while ensuring a clean reaction profile with minimal side products. The implementation of a constant pressure funnel for the slow, dropwise addition of trifluoroethanol, combined with continuous inert gas protection (nitrogen or argon), provides an exceptional level of control over the reaction environment, effectively suppressing hydrolysis and oxidation risks. This streamlined process not only simplifies the equipment requirements to standard three-necked flasks and condensers but also facilitates a straightforward workup procedure involving simple distillation, thereby drastically reducing the operational complexity and energy consumption associated with product isolation.
Mechanistic Insights into Alcoholysis of Chlorosilane
The core chemical transformation driving this synthesis is a nucleophilic substitution at the silicon center, where the oxygen atom of the trifluoroethanol attacks the electrophilic silicon atom of the vinyl trichlorosilane. This mechanism proceeds through a pentacoordinate silicon intermediate, eventually displacing the chloride ions which are scavenged as hydrogen chloride gas or trapped within the solvent matrix depending on the specific stoichiometry and gas flow dynamics. The presence of the electron-withdrawing trifluoromethyl group on the ethanol enhances the acidity of the hydroxyl proton, potentially facilitating the deprotonation step required to form the stable Si-O-C bond, although the reaction is primarily driven by the thermodynamic stability of the resulting silyl ether linkage compared to the silyl chloride precursor. The inert atmosphere plays a dual mechanistic role: it prevents the ingress of atmospheric moisture which would otherwise hydrolyze the reactive chlorosilane into silanols and eventually siloxanes, and it aids in the continuous removal of the generated HCl byproduct, shifting the equilibrium towards the desired product according to Le Chatelier's principle. Understanding this delicate balance of nucleophilicity, steric hindrance, and byproduct management is crucial for scaling the reaction while maintaining the high purity required for electronic or pharmaceutical grade applications.
Controlling the impurity profile in this synthesis is paramount, particularly given the sensitivity of the vinyl group to radical polymerization or acid-catalyzed rearrangement. The patent specifies a rigorous control of temperature and addition rates to mitigate these risks, ensuring that the reaction mixture remains homogeneous and that local hotspots of high acidity do not trigger degradation pathways. The use of dichloromethane, a solvent with excellent solvating power for both organic and organometallic species, helps to stabilize the transition states and keeps the growing oligomeric species in solution, preventing premature precipitation or gelation. Furthermore, the final purification step via reduced pressure distillation is designed to separate the target monomer from unreacted starting materials and higher boiling point oligomers based on their distinct volatility differences. This mechanistic understanding allows process chemists to fine-tune the distillation parameters to achieve the reported yields of 63% to 75%, ensuring that the final product meets the stringent specifications necessary for downstream polymerization into high-performance fluorosilicones.
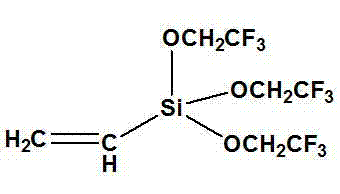
How to Synthesize Vinyl Tris(2,2,2-trifluoro) Ethoxyl Silane Efficiently
To successfully replicate this synthesis in a laboratory or pilot plant setting, operators must adhere to a strict protocol that emphasizes moisture exclusion and thermal regulation throughout the entire process. The procedure begins with the assembly of a dry, inert-gas-purged reaction vessel equipped with precise addition capabilities, followed by the careful introduction of the solvent and the chlorosilane precursor under a protective blanket of nitrogen or argon. The subsequent slow addition of trifluoroethanol is the critical rate-determining step that dictates the overall success of the batch, requiring constant monitoring of internal temperature and gas evolution to prevent exothermic spikes. Detailed standardized synthetic steps, including specific molar ratios, flow rates, and distillation cut points, are essential for reproducibility and are outlined in the technical guide below for immediate implementation by process development teams.
- Prepare a three-necked flask equipped with an airway, constant pressure funnel, and ball-type condenser. Add dichloromethane solvent and introduce vinyl trichlorosilane under the liquid level while protecting with nitrogen or argon gas.
- Slowly drip trifluoroethanol from the constant pressure funnel at a controlled rate (0.5-1 mL/min) while maintaining magnetic stirring and heating to initiate reflux.
- After the addition is complete, maintain the reaction temperature between 20-40°C for 4-7 hours, then remove solvent and excess alcohol via distillation to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this patented synthesis route offers substantial benefits that extend far beyond simple chemical yield improvements. By eliminating the dependency on scarce or expensive transition metal catalysts, manufacturers can significantly reduce the raw material cost base and insulate their supply chains from the volatility associated with precious metal markets. The simplicity of the equipment setup—requiring only standard glass-lined or stainless steel reactors capable of handling mild corrosives—means that existing manufacturing infrastructure can often be repurposed for this production without the need for massive capital expenditure on specialized high-pressure or high-temperature units. This flexibility translates directly into enhanced supply chain reliability, as production can be rapidly scaled up or shifted between facilities with minimal downtime or requalification efforts, ensuring a consistent flow of critical intermediates to downstream customers in the coatings and electronics sectors.
- Cost Reduction in Manufacturing: The absence of noble metal catalysts removes the need for costly recovery and purification systems, such as activated carbon beds or ion-exchange columns, which are typically required to meet trace metal specifications in high-end applications. Furthermore, the mild reaction conditions (20-40°C) result in drastically lower energy consumption for heating and cooling compared to traditional high-temperature pyrolysis or catalytic cracking processes. The use of dichloromethane, a common and recyclable solvent, further optimizes the operational expenditure by allowing for efficient solvent recovery loops that minimize waste disposal costs and environmental compliance burdens. These cumulative efficiencies create a leaner manufacturing model that delivers substantial cost savings per kilogram of finished product, enhancing the overall margin profile for suppliers operating in the competitive fluorine materials market.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically vinyl trichlorosilane and trifluoroethanol, are commodity chemicals produced by multiple global suppliers, reducing the risk of single-source bottlenecks that often plague specialized fine chemical supply chains. The robustness of the reaction against minor fluctuations in ambient conditions ensures high batch-to-batch consistency, which is critical for maintaining long-term contracts with quality-sensitive clients in the automotive and aerospace industries. Additionally, the relatively short reaction time of 4 to 7 hours allows for high throughput and rapid turnover of production assets, enabling suppliers to respond quickly to surges in demand or urgent orders without compromising on product quality or delivery timelines. This agility is a key differentiator in a market where lead times for custom fluorinated intermediates can often stretch into months.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of recoverable solvent and manageable amounts of hydrogen chloride which can be neutralized or scrubbed using standard industrial abatement systems. The lack of heavy metal contaminants simplifies the wastewater treatment process and reduces the regulatory burden associated with discharging effluent containing toxic metals. Moreover, the scalability of the alcoholysis reaction is well-proven, as the heat transfer characteristics of the system remain favorable even when moving from liter-scale laboratory flasks to cubic-meter industrial reactors, provided that the addition rates are adjusted proportionally. This ease of scale-up ensures that the technology can seamlessly transition from pilot validation to full commercial production, supporting the growing global demand for durable, weather-resistant fluorosilicone materials in a sustainable manner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of vinyl tris(2,2,2-trifluoro) ethoxyl silane, derived directly from the experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of this synthesis route for potential partners and technical stakeholders evaluating the technology for integration into their own supply chains. Understanding these nuances is essential for making informed decisions about process adoption and long-term sourcing strategies in the specialized field of fluorinated organosilicon chemistry.
Q: What are the critical reaction conditions for synthesizing vinyl tris(2,2,2-trifluoro) ethoxyl silane?
A: The synthesis requires strict moisture exclusion using inert gases like nitrogen or argon. The reaction is conducted in dichloromethane at mild temperatures ranging from 20°C to 40°C, with a controlled addition rate of trifluoroethanol to manage exothermicity and ensure high conversion.
Q: Why is this alcoholysis method preferred over traditional catalytic routes?
A: This method eliminates the need for expensive transition metal catalysts, thereby reducing raw material costs and simplifying downstream purification by avoiding heavy metal removal steps. It also utilizes readily available raw materials like vinyl trichlorosilane and trifluoroethanol.
Q: What are the primary industrial applications of this fluorosilane monomer?
A: The resulting monomer is a key building block for fluorosilicone polymers, offering superior weather resistance, oil repellency, and thermal stability. It is widely used in high-performance coatings, release agents, textile finishing, and lubricant additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Tris(2,2,2-trifluoro) Ethoxyl Silane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity fluorosilicone intermediates play in the development of advanced materials for demanding industrial applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market-ready product is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Whether you require this specific vinyl silane for polymer modification or as a cross-linking agent, our technical expertise ensures that your supply chain remains robust, compliant, and cost-effective.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this catalyst-free methodology for your manufacturing needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the performance of our materials in your own formulations with confidence and speed.
