Advanced One-Step Synthesis of Chiral Nitrogenous Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex chiral architectures, particularly nitrogenous compounds that serve as pivotal scaffolds for bioactive molecules. A significant advancement in this domain is detailed in patent CN102757364B, which discloses a novel preparation and synthetic method for a specific chiral nitrogenous compound, identified chemically as N,N-bis-[(R)-(2-methoxy-phenyl) cyanomethyl]-(S)-1-phenylethylamine. This innovation addresses the critical need for efficient access to ethylamino phenyl-phenylacetonitrile derivatives, which are recognized as important medicinal intermediates capable of facilitating the synthesis of heterocyclic drugs such as arylpyrazines. The disclosed methodology leverages a copper-based chiral catalyst system to achieve stereocontrol, offering a streamlined alternative to traditional multi-step sequences that often suffer from poor atom economy and cumbersome purification requirements.
For research and development directors overseeing process chemistry, the structural integrity and purity of such intermediates are paramount. The target molecule features a sophisticated arrangement where a central nitrogen atom is bonded to a chiral phenylethyl group and two cyanomethyl-substituted aromatic rings, creating a rigid and sterically defined environment. ![Chemical structure of N,N-bis-[(R)-(2-methoxy-phenyl) cyanomethyl]-(S)-1-phenylethylamine showing chiral centers](/insights/img/chiral-nitrogenous-compound-synthesis-pharma-supplier-20260312002143-01.png) This specific configuration is not merely academic; it dictates the compound's efficacy when utilized as a ligand or catalyst in downstream asymmetric transformations. The ability to access this specific stereoisomer through a direct synthetic route represents a tangible value add for supply chains dependent on high-purity chiral building blocks for API manufacturing.
This specific configuration is not merely academic; it dictates the compound's efficacy when utilized as a ligand or catalyst in downstream asymmetric transformations. The ability to access this specific stereoisomer through a direct synthetic route represents a tangible value add for supply chains dependent on high-purity chiral building blocks for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex chiral amines and nitrile-containing intermediates has been plagued by operational inefficiencies that hinder commercial viability. Conventional routes often rely on harsh reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate waste management and increase safety risks in a production setting. Furthermore, traditional methods frequently necessitate multiple protection and deprotection steps to manage the reactivity of amine and aldehyde functional groups, leading to a significant reduction in overall yield and a dramatic increase in production costs. The reliance on stoichiometric amounts of chiral auxiliaries rather than catalytic systems further exacerbates the economic burden, generating substantial quantities of chemical waste that require expensive disposal protocols. These factors collectively create bottlenecks in the supply chain, extending lead times and reducing the agility of manufacturers to respond to market demands for new therapeutic candidates.
The Novel Approach
In stark contrast to these legacy challenges, the method described in the patent introduces a remarkably concise one-step synthesis that directly constructs the target chiral framework from readily available starting materials. By employing a specific copper complex derived from (S)-1-phenylethylamine, the process achieves asymmetric induction without the need for cryogenic conditions or exotic reagents. The reaction utilizes 2-methoxybenzaldehyde and trimethylsilyl cyanide as primary substrates, which are commoditized chemicals with stable supply chains, thereby mitigating raw material sourcing risks. 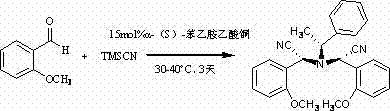 The operational simplicity is further enhanced by the use of absolute methanol as a solvent, allowing the reaction to proceed at ambient temperature over a three-day period. This elimination of energy-intensive heating or cooling cycles translates directly into lower utility costs and a reduced carbon footprint, aligning with modern green chemistry principles while maintaining high standards of chemical selectivity.
The operational simplicity is further enhanced by the use of absolute methanol as a solvent, allowing the reaction to proceed at ambient temperature over a three-day period. This elimination of energy-intensive heating or cooling cycles translates directly into lower utility costs and a reduced carbon footprint, aligning with modern green chemistry principles while maintaining high standards of chemical selectivity.
Mechanistic Insights into Copper-Catalyzed Asymmetric Cyanosilylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for larger scales. The proposed mechanism suggests a cooperative interaction between the copper center and the chiral amine ligand, which activates the trimethylsilyl cyanide towards nucleophilic attack on the aldehyde carbonyl. Initially, the reaction generates an intermediate hydroxynitrile species, specifically (R)-(1-hydroxy)-2-methoxybenzeneacetonitrile, with high stereochemical fidelity dictated by the chiral environment of the catalyst. This intermediate does not remain isolated but undergoes a subsequent condensation reaction with another equivalent of the activated nitrile species or interacts further with the catalyst system. The loss of water molecules through the interaction of hydrogen protons and hydroxyl groups facilitates the formation of the final tertiary amine structure, locking in the desired stereochemistry.
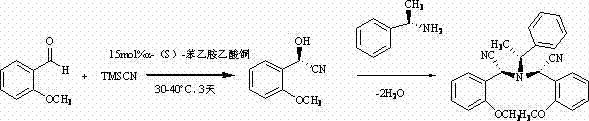 This cascade-like behavior implies that the catalyst plays a dual role: first as an activator for the cyanosilylation and subsequently as a promoter for the condensation and dehydration steps. For process chemists, this insight is valuable as it highlights the importance of water management and solvent dryness in maximizing yield. The fact that the reaction proceeds to form a stable single crystal upon natural volatilization indicates a thermodynamic driving force towards the product, which simplifies the crystallization and isolation phases. Such mechanistic clarity allows for better troubleshooting of impurities, ensuring that the final material meets the stringent specifications required for pharmaceutical applications where trace metal residues or incorrect stereoisomers can be detrimental.
This cascade-like behavior implies that the catalyst plays a dual role: first as an activator for the cyanosilylation and subsequently as a promoter for the condensation and dehydration steps. For process chemists, this insight is valuable as it highlights the importance of water management and solvent dryness in maximizing yield. The fact that the reaction proceeds to form a stable single crystal upon natural volatilization indicates a thermodynamic driving force towards the product, which simplifies the crystallization and isolation phases. Such mechanistic clarity allows for better troubleshooting of impurities, ensuring that the final material meets the stringent specifications required for pharmaceutical applications where trace metal residues or incorrect stereoisomers can be detrimental.
How to Synthesize N,N-bis-[(R)-(2-methoxy-phenyl) cyanomethyl]-(S)-1-phenylethylamine Efficiently
Executing this synthesis requires strict adherence to anhydrous conditions to prevent premature hydrolysis of the silyl cyanide reagent and to ensure the stability of the copper catalyst. The protocol involves dissolving the catalyst in absolute methanol before introducing the aldehyde and cyanide sources, ensuring a homogeneous reaction medium that promotes consistent kinetics throughout the vessel. Following the reaction period, the workup involves standard column chromatography techniques, utilizing a specific ratio of petroleum ether to methylene chloride to effectively separate the target compound from unreacted starting materials and minor byproducts. The detailed standardized synthesis steps for this procedure are outlined in the guide below.
- Prepare the reaction mixture by combining 15mol% (S)-1-phenylethylamine cupric acetate catalyst, 2-methoxybenzaldehyde, and trimethylsilyl cyanide in absolute methanol solvent.
- Stir the mixture at room temperature for approximately 3 days to allow the asymmetric induction and condensation reactions to proceed to completion.
- Purify the crude product using column chromatography with a petroleum ether and methylene chloride eluent system to isolate the target single crystal.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers distinct strategic advantages that extend beyond mere chemical novelty. The simplification of the process flow from multi-step to one-step significantly reduces the number of unit operations required, which in turn lowers the capital expenditure needed for reactor occupancy and labor hours. By eliminating the need for complex transition metal removal steps often associated with palladium or other precious metal catalysts, the downstream purification burden is drastically lightened. This reduction in processing complexity directly correlates to a shorter manufacturing cycle time, enabling faster turnaround for pilot batches and accelerating the timeline from laboratory discovery to commercial production. Furthermore, the use of common solvents like methanol and standard eluents ensures that the process can be easily transferred between different manufacturing sites without requiring specialized infrastructure.
- Cost Reduction in Manufacturing: The economic benefits of this method are driven primarily by the high atom efficiency and the catalytic nature of the chiral inducer. Unlike stoichiometric chiral resolutions which inherently discard 50% of the material, this asymmetric synthesis builds chirality directly, minimizing raw material waste. The avoidance of cryogenic cooling (-78°C or lower), which is energy-intensive and requires specialized equipment, results in substantial utility savings. Additionally, the high transformation efficiency observed in the catalytic application of the product suggests that smaller quantities of the synthesized compound can drive significant downstream value, enhancing the return on investment for the initial synthesis.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 2-methoxybenzaldehyde and trimethylsilyl cyanide ensures a robust and resilient supply chain. These precursors are produced globally by numerous vendors, reducing the risk of supply disruption due to single-source dependency. The mild reaction conditions also imply that the manufacturing process is less susceptible to deviations caused by minor fluctuations in temperature control, leading to more consistent batch-to-batch quality. This reliability is critical for long-term supply agreements where consistency in impurity profiles is mandated by regulatory bodies.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the absence of exothermic hazards typically associated with cyanide additions at high concentrations. The room temperature operation allows for simpler reactor designs and safer handling protocols, reducing the engineering controls required for containment. From an environmental standpoint, the generation of waste is minimized due to the high selectivity of the reaction, and the solvents used are amenable to standard recovery and recycling distillation processes. This alignment with green chemistry metrics supports corporate sustainability goals and simplifies the permitting process for new manufacturing lines.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and characteristics of this chiral synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's fit within your specific project requirements. Understanding these details helps in assessing the feasibility of integrating this intermediate into your existing pipeline.
Q: What is the specific catalyst used in this chiral synthesis?
A: The process utilizes 15mol% of (S)-1-phenylethylamine cupric acetate as a chiral catalyst to induce stereoselectivity during the nitrile silicification reaction.
Q: What are the reaction conditions required for this synthesis?
A: The reaction proceeds under mild conditions at room temperature over a period of 3 days, using anhydrous methanol as the solvent, which simplifies thermal management.
Q: How effective is the resulting compound as a catalyst?
A: When applied to the cyanosilylation of benzaldehyde, the synthesized compound demonstrates high catalytic activity with a transformation efficiency reaching up to 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-bis-[(R)-(2-methoxy-phenyl) cyanomethyl]-(S)-1-phenylethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable industrial supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of N,N-bis-[(R)-(2-methoxy-phenyl) cyanomethyl]-(S)-1-phenylethylamine meets the exacting standards required for pharmaceutical synthesis. Our commitment to technical excellence means we can adapt the patented conditions to optimize yield and throughput specifically for your volume needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this catalytic method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development programs proceed without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
