Revolutionizing Doxycycline Intermediate Production with Green Ionic Liquid Catalysis Technology
The pharmaceutical industry is currently witnessing a significant paradigm shift in the manufacturing of tetracycline antibiotics, driven by the urgent need for greener and safer production methodologies. Patent CN114315627B, titled "Method for synthesizing doxycycline dehydrate by catalyzing with normal-temperature ionic liquid and zeolite," presents a groundbreaking technical solution that addresses the longstanding safety and environmental hazards associated with traditional doxycycline intermediate synthesis. This innovation replaces the highly corrosive and dangerous hydrogen fluoride (HF) or thionyl chloride reagents with a sophisticated dual-catalyst system comprising a normal-temperature ionic liquid and a modified zeolite. For R&D directors and process engineers, this patent represents a critical evolution in antibiotic intermediate manufacturing, offering a pathway to high-purity doxycycline anhydrite while drastically mitigating the risks associated with handling hazardous fluorinating agents. The technical robustness of this approach lies in its ability to maintain mild reaction conditions, typically between 20°C and 25°C, which stands in stark contrast to the harsh environments required by legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of doxycycline has been plagued by the reliance on hydrogen fluoride falling-film evaporators for the crucial dehydration step of oxytetracycline chloride. This conventional methodology poses severe challenges for procurement managers and supply chain heads, primarily due to the extreme toxicity and corrosivity of hydrogen fluoride, which necessitates specialized, expensive equipment and rigorous safety protocols. The generation of strong-acid waste mother liquor is another critical bottleneck, creating substantial environmental compliance burdens and increasing the cost of waste treatment significantly. Furthermore, equipment maintenance in HF-based facilities is notoriously difficult and costly, as the aggressive nature of the reagent leads to rapid degradation of reactor linings and piping. These factors collectively contribute to supply chain fragility, where any disruption in hazardous chemical supply or waste disposal capacity can halt production entirely, making the search for alternative, safer synthetic routes a top priority for forward-thinking pharmaceutical manufacturers.
The Novel Approach
The novel approach detailed in the patent utilizes a synergistic catalytic system that fundamentally alters the reaction landscape for doxycycline synthesis. By employing a normal-temperature ionic liquid synthesized from imidazole inner salts and protonic acids, combined with a modified H-beta zeolite, the process achieves efficient dehydration without the need for hazardous fluorinating agents. This method operates under remarkably mild conditions, with the reaction temperature strictly controlled between 20°C and 25°C, thereby eliminating the thermal stress on equipment and reducing energy consumption. The use of a solid acid zeolite catalyst, which can be easily filtered and recovered, alongside a liquid ionic medium that can be distilled and reused, creates a closed-loop system that minimizes waste generation. This transition not only enhances the safety profile of the manufacturing facility but also simplifies the downstream purification process, resulting in a final product with fewer by-products and a consistently high yield, positioning this technology as a superior alternative for reliable doxycycline intermediate supplier networks.
Mechanistic Insights into Ionic Liquid-Zeolite Dual Catalysis
The core of this technological breakthrough lies in the unique interaction between the Brønsted acidic ionic liquid and the solid acid sites of the modified zeolite. The ionic liquid, formed by the reaction of an imidazole inner salt with a strong protonic acid such as trifluoromethanesulfonic acid or p-toluenesulfonic acid, serves as both the solvent and a homogeneous catalyst. This dual functionality ensures excellent solubility for the oxytetracycline chloride substrate while providing the necessary acidic protons to facilitate the dehydration mechanism. The structural integrity of the ionic liquid precursor is critical, as seen in the specific zwitterionic architecture that allows for stable salt formation and subsequent protonation.
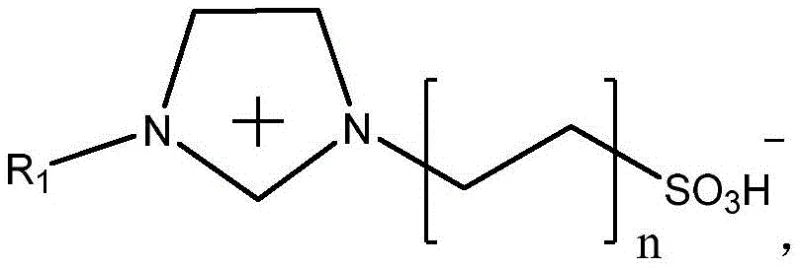
Simultaneously, the modified H-beta zeolite acts as a heterogeneous co-catalyst, providing additional acidic sites and a porous framework that may help in stabilizing transition states or adsorbing specific by-products. The modification of the zeolite with agents like citric acid or phosphoric acid further tunes its acidity and surface properties, optimizing it for this specific transformation. This dual-catalyst strategy ensures that the reaction proceeds with high selectivity, minimizing the formation of degradation products that are common in harsher acidic environments. The result is a reaction mixture that is not only efficient but also clean, with the main impurities kept to a relative content of approximately 1.2% to 1.5%, and unknown impurities maintained below 0.1%, demonstrating the precision of this mechanistic approach in controlling the impurity profile of high-purity pharmaceutical intermediates.
How to Synthesize Doxycycline Dehydrate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this green chemistry solution on a commercial scale. The process begins with the preparation of the ionic liquid reaction system, where the precise stoichiometry of the imidazole inner salt and protonic acid is crucial for establishing the correct acidity. Following this, the modified zeolite is introduced, and the substrate, oxytetracycline chloride, is added under strict temperature control to prevent thermal degradation. The subsequent crystallization step involves the addition of the reaction mixture to a cooled p-toluenesulfonic acid ethanol solution, which induces the precipitation of the desired doxycycline dehydrate p-toluenesulfonate salt. Detailed standardized synthesis steps see the guide below.
- Prepare the ionic liquid reaction system by mixing imidazole inner salt with a protonic acid, then adding modified H-beta zeolite catalyst at 10-35°C.
- Add oxytetracycline chloride to the system at temperatures not exceeding 30°C, maintaining reaction temperature at 20-25°C, then filter to recover the zeolite.
- Precipitate the product by adding the reaction solution to a cooled p-toluenesulfonic acid ethanol solution, followed by filtration and ionic liquid recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-zeolite catalytic method offers profound strategic advantages that extend far beyond simple chemical efficiency. The elimination of hydrogen fluoride and thionyl chloride from the supply chain removes a major logistical and regulatory hurdle, as these substances are subject to increasingly stringent global transport and storage regulations. By shifting to non-hazardous ionic liquids and solid zeolites, companies can significantly reduce their insurance premiums, safety training costs, and the capital expenditure required for specialized corrosion-resistant infrastructure. Furthermore, the ability to recover and reuse the catalysts multiple times translates directly into a drastic reduction in raw material consumption per kilogram of finished product. This circular economy approach within the reactor vessel ensures that the cost of goods sold (COGS) is optimized over the long term, providing a buffer against volatility in raw material pricing markets.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the recyclability of the catalytic system and the reduction in waste treatment expenses. Since the ionic liquid can be recovered with efficiency exceeding 99% and reused for multiple batches, the effective cost of the solvent system is amortized over a large production volume, leading to substantial cost savings. Additionally, the absence of strong-acid waste mother liquor means that wastewater treatment facilities do not need to be oversized for neutralization capacity, further lowering operational expenditures. The mild reaction conditions also reduce energy costs associated with heating and cooling, contributing to a leaner and more cost-effective manufacturing profile for complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliance on hazardous reagents like hydrogen fluoride often creates single points of failure in the supply chain, as few suppliers are willing or able to handle such dangerous materials. By transitioning to a system based on stable ionic liquids and commercially available zeolites, manufacturers can diversify their supplier base and reduce the risk of production stoppages due to raw material shortages. The robustness of the catalyst system, which maintains high activity even after multiple reuse cycles, ensures consistent batch-to-batch quality and throughput. This stability is crucial for meeting the rigorous delivery schedules demanded by global pharmaceutical clients, thereby strengthening the position of the manufacturer as a reliable partner in the antibiotic value chain.
- Scalability and Environmental Compliance: From an environmental, social, and governance (ESG) perspective, this technology positions the manufacturing process for long-term sustainability. The near-zero discharge of hazardous waste aligns perfectly with modern environmental regulations and corporate sustainability goals, reducing the risk of fines and reputational damage. The process is inherently scalable, as demonstrated by the patent examples ranging from gram-scale to multi-hundred-kilogram batches, indicating that the heat and mass transfer characteristics remain favorable upon scale-up. This ease of commercial scale-up of complex pharmaceutical intermediates allows companies to rapidly respond to market demand without the need for extensive process re-engineering or new environmental permits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalytic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is essential for technical teams evaluating the potential for technology transfer or licensing.
Q: How does the ionic liquid-zeolite method compare to traditional hydrogen fluoride dehydration?
A: Unlike the traditional method which uses dangerous hydrogen fluoride and generates difficult-to-treat strong-acid waste, this novel method operates under mild conditions with almost no waste acid generation and allows for the recycling of both the ionic liquid and the zeolite catalyst.
Q: What represents the key advantage regarding impurity control in this process?
A: The process demonstrates superior impurity control, achieving a relative content of main impurities around 1.2% to 1.5% and unknown impurities less than 0.1%, which is significantly cleaner than conventional processes that often yield higher impurity profiles.
Q: Is the catalyst system reusable for commercial scale production?
A: Yes, the system is highly sustainable; the zeolite catalyst can be recovered with rates exceeding 99%, and the ionic liquid can be reused multiple times (up to 8 cycles in examples) with minimal loss in yield or increase in impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxycycline Anhydrite Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ionic liquid-zeolite catalytic method for the production of doxycycline intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that the transition to greener chemistry requires not just technical capability but also a deep understanding of regulatory landscapes and supply chain dynamics, areas where our team excels.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their doxycycline supply needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this green catalytic route for your operations. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your product quality and your bottom line. Let us be your trusted partner in navigating the future of sustainable antibiotic manufacturing.
