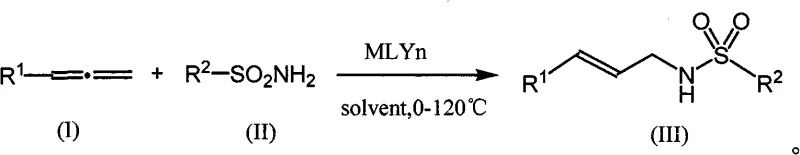
Advanced Gold-Catalyzed Synthesis of Allyl Sulfonamides for Scalable Pharmaceutical Intermediate Production
The chemical landscape for synthesizing complex nitrogen-containing scaffolds has evolved significantly with the introduction of transition metal catalysis, particularly involving gold complexes. Patent CN101585788A, filed in late 2009, introduces a groundbreaking methodology for the preparation of allyl sulfonamide compounds, specifically those defined by formula (III). This technology represents a pivotal shift from traditional harsh synthetic routes to a more refined, atom-economical process that leverages the unique pi-acidity of gold cations. For R&D directors and procurement specialists in the fine chemical sector, this patent outlines a pathway to produce high-purity intermediates that are crucial for the downstream synthesis of cinnamaldehyde derivatives, which are themselves vital building blocks in the agrochemical and pharmaceutical industries. The innovation lies not just in the product structure, but in the operational simplicity and environmental profile of the reaction, which proceeds under mild thermal conditions with exceptional selectivity.
The strategic value of this technology extends to its ability to generate diverse structural analogues by varying the R1 and R2 substituents, allowing for the customization of intermediates for specific drug discovery programs or pesticide formulations. By utilizing commercially available sulfonamides and easily accessible allene precursors, the process minimizes supply chain bottlenecks often associated with exotic starting materials. Furthermore, the reaction's tolerance for various functional groups ensures that sensitive moieties required in advanced medicinal chemistry can remain intact during the sulfonamide installation step. This robustness makes the technology highly attractive for the commercial scale-up of complex pharmaceutical intermediates where consistency and purity are paramount.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of this gold-catalyzed protocol, the synthesis of allyl sulfonamides was fraught with significant technical and economic challenges that hindered efficient large-scale manufacturing. Traditional methods often relied on palladium catalysis or multi-step condensation reactions involving unstable intermediates like sulfonyl isocyanates or formates. These legacy processes typically required rigorous exclusion of moisture and oxygen, utilized high-boiling solvents that were difficult to remove during workup, and frequently resulted in poor regioselectivity, leading to complex mixtures of branched and linear isomers. The necessity for extensive purification steps, such as repeated recrystallizations or preparative HPLC, drastically reduced the overall throughput and increased the cost of goods sold. Additionally, the use of stoichiometric amounts of activating agents or harsh bases often generated substantial chemical waste, conflicting with modern green chemistry principles and increasing the burden on waste treatment facilities.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a synergistic catalytic system comprising a gold chloride-triphenylphosphine complex and a silver salt co-catalyst to activate allenes towards nucleophilic attack by sulfonamides. This method operates effectively at temperatures ranging from 0°C to 50°C, significantly reducing energy consumption compared to refluxing conditions required by older methods. The reaction exhibits remarkable atom economy, as essentially all atoms from the starting allene and sulfonamide are incorporated into the final product without the generation of stoichiometric by-products. The use of inert organic solvents like 1,4-dioxane or dichloromethane facilitates straightforward workup procedures involving simple aqueous extraction and column chromatography. This streamlined workflow not only accelerates the production timeline but also enhances the safety profile for operators by eliminating the need for hazardous reagents and extreme reaction parameters.
Mechanistic Insights into Gold-Catalyzed Hydroamination
The core of this technological advancement lies in the activation mechanism of the allene moiety by the cationic gold species generated in situ. Upon coordination with the gold center, the electron-rich pi-system of the allene becomes highly electrophilic, facilitating a regioselective nucleophilic attack by the nitrogen atom of the sulfonamide. The choice of ligand, specifically triphenylphosphine, plays a critical role in stabilizing the gold center and modulating its electronic properties to favor the formation of the linear E-alkene product over potential branched isomers. This high degree of stereocontrol is essential for pharmaceutical applications where the geometric configuration of the double bond can profoundly influence the biological activity of the final active pharmaceutical ingredient. The presence of the silver co-catalyst, such as silver trifluoromethanesulfonate, serves to abstract the chloride ligand from the gold precursor, generating the active cationic gold species necessary for catalytic turnover.
From an impurity control perspective, the mild reaction conditions inherently suppress side reactions such as allene polymerization or over-oxidation, which are common pitfalls in transition metal catalysis. The kinetic profile of the reaction allows for precise monitoring via thin-layer chromatography, enabling operators to quench the reaction at optimal conversion to minimize the formation of degradation products. Furthermore, the stability of the sulfonamide group under these neutral to slightly acidic catalytic conditions ensures that acid-sensitive protecting groups elsewhere in the molecule remain unaffected. This chemoselectivity reduces the need for protective group strategies, thereby shortening the overall synthetic sequence and improving the cumulative yield of the manufacturing process. The result is a cleaner crude product profile that simplifies downstream purification and ensures consistent quality across different production batches.
How to Synthesize Allyl Sulfonamide Efficiently
The practical implementation of this synthesis route is designed for scalability and reproducibility in a standard chemical manufacturing environment. The process begins with the precise weighing of the allene substrate and the sulfonamide nucleophile, followed by their dissolution in a dry, inert solvent to prevent catalyst deactivation. The addition of the gold catalyst and silver co-catalyst initiates the reaction immediately, requiring careful temperature control to maintain the optimal range of 0°C to 50°C depending on the specific substrate reactivity. Detailed standardized operating procedures for mixing ratios, addition rates, and quenching protocols are essential to maximize yield and safety. The following guide outlines the critical steps derived from the patent examples to ensure successful execution of this transformation.
- Mix allene compound and sulfonamide compound in an inert organic solvent such as 1,4-dioxane.
- Add gold chloride-triphenylphosphine complex and silver trifluoromethanesulfonate as co-catalyst to the mixture.
- React at mild temperatures between 0°C and 50°C for 4 to 20 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this gold-catalyzed technology offers tangible benefits in terms of cost structure and logistical reliability. The shift towards a high-yield, single-step coupling reaction eliminates the need for multiple intermediate isolation steps, which traditionally consume significant time, solvent, and labor resources. By reducing the number of unit operations, the overall manufacturing footprint is minimized, leading to substantial cost savings in facility usage and utility consumption. Moreover, the high atom economy means that raw material costs are optimized, as less feedstock is wasted in the form of by-products. This efficiency translates directly into a more competitive pricing model for the final intermediate, allowing downstream customers to better manage their own cost of goods.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the reduction in reaction times contribute to a leaner cost base. While gold is a precious metal, it is used in catalytic quantities, and the simplified purification process reduces the expenditure on chromatography media and solvents. The mild conditions also lower energy costs associated with heating and cooling, further enhancing the economic viability of the process. Additionally, the high selectivity reduces the loss of valuable starting materials, ensuring that every kilogram of input contributes maximally to the output.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as substituted sulfonamides and allenes mitigates the risk of supply disruptions associated with custom-synthesized reagents. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuating market demands. The simplified workup procedure also shortens the lead time from reaction completion to finished product release, improving inventory turnover rates. This agility is crucial for maintaining continuous supply lines to pharmaceutical and agrochemical clients who operate on tight production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable from gram to multi-ton quantities without significant re-engineering of the reaction parameters. The use of common organic solvents and the absence of highly toxic reagents simplify waste management and regulatory compliance. The high purity of the crude product reduces the environmental load associated with extensive purification steps, aligning with increasingly stringent global environmental standards. This sustainability profile enhances the long-term viability of the supply chain by future-proofing the manufacturing process against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. The responses cover catalyst specifics, comparative advantages, and downstream utility to provide a comprehensive overview.
Q: What are the key advantages of the gold-catalyzed method over traditional palladium methods?
A: The gold-catalyzed method operates under significantly milder conditions (0-50°C vs higher temps), offers superior atom economy with no by-products, and achieves higher selectivity and yields compared to conventional palladium-catalyzed routes which often suffer from cumbersome steps and difficult purification.
Q: What is the primary industrial application of these allyl sulfonamide compounds?
A: These compounds serve as critical intermediates that can be converted into cinnamaldehyde derivatives through desulfonylation and oxidation, which are essential raw materials for synthesizing various pesticides and pharmaceutical agents.
Q: Is the catalyst system cost-effective for large-scale production?
A: Yes, the process uses catalytic amounts of gold complexes (0.02-0.1 molar ratio) combined with readily available sulfonamides and allenes, ensuring high raw material utilization and reducing overall processing costs despite the use of precious metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Sulfonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development and agricultural chemical programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of allyl sulfonamide meets the exacting standards required for GMP manufacturing. Our commitment to technical excellence ensures that you receive a product that is not only chemically pure but also consistent in its physical properties, facilitating smooth downstream processing.
We invite you to collaborate with us to leverage this advanced gold-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the fine chemical industry.
