Scalable Synthesis and Commercial Supply of 3-Fluoro-5-Nitropyridine-2-Formaldehyde for Advanced Drug Discovery
The pharmaceutical industry continuously demands high-purity heterocyclic intermediates to accelerate the development of novel therapeutic agents, particularly those incorporating fluorinated pyridine scaffolds which are known to enhance metabolic stability and binding affinity in drug candidates. Patent CN112679361B discloses a robust and highly efficient synthetic methodology for the preparation of 3-fluoro-5-nitropyridine-2-formaldehyde, a critical building block that addresses the longstanding challenges associated with regioselective functionalization of pyridine rings. This technical insight report analyzes the proprietary process detailed in the patent, highlighting its superiority in terms of yield optimization, impurity control, and operational simplicity compared to legacy methods. By leveraging a strategic sequence of nitration, halogenation, cross-coupling, and oxidation, the described route ensures consistent quality essential for regulatory compliance in active pharmaceutical ingredient (API) manufacturing. As a leading partner in fine chemical synthesis, understanding the nuances of this patent allows us to offer reliable supply chain solutions for complex pharmaceutical intermediates that meet the rigorous standards of global R&D teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for fluorinated nitro-pyridine aldehydes often suffer from significant drawbacks including poor regioselectivity, harsh reaction conditions that compromise safety, and the generation of difficult-to-remove impurities that lower overall yield. Conventional methods frequently rely on direct oxidation of methyl groups using non-selective oxidants or multi-step sequences that require protecting group strategies, thereby increasing material costs and processing time substantially. Furthermore, older methodologies may utilize expensive or hazardous reagents that pose environmental risks and complicate waste disposal protocols, creating bottlenecks in the supply chain for procurement managers focused on sustainability and cost efficiency. The lack of precise temperature control in historical nitration processes often leads to the formation of isomeric by-products, necessitating costly purification steps such as repeated recrystallization or preparative chromatography which are not ideal for large-scale production. These inefficiencies collectively result in higher production costs and longer lead times, making it challenging for manufacturers to secure a stable supply of high-quality intermediates for time-sensitive drug development projects.
The Novel Approach
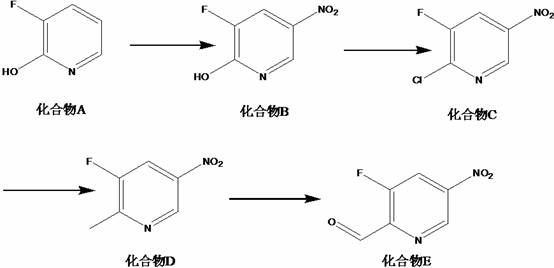
The innovative process outlined in the patent overcomes these historical limitations by introducing a streamlined four-step sequence that maximizes atom economy and minimizes waste generation through careful selection of reagents and reaction parameters. By initiating the synthesis with a controlled nitration of 3-fluoro-2-hydroxypyridine under mild acidic conditions, the method ensures high conversion rates while suppressing the formation of unwanted isomers that typically plague conventional nitration reactions. The subsequent chlorination step utilizes a specific combination of phosphorus oxychloride and phosphorus pentachloride at low temperatures to activate the ring for further functionalization without degrading the sensitive nitro group. This strategic activation enables a highly efficient palladium-catalyzed methylation using trimethylboroxine, which introduces the carbon framework necessary for the final aldehyde functionality with exceptional precision. Finally, the use of selenium dioxide for the oxidation step provides superior selectivity for converting the methyl group to an aldehyde, avoiding over-oxidation to carboxylic acids and ensuring the final product meets stringent purity specifications required for downstream pharmaceutical applications.
Mechanistic Insights into the Multi-Step Catalytic Sequence
The core of this synthetic breakthrough lies in the precise orchestration of electrophilic aromatic substitution and transition metal catalysis to construct the target molecule with high fidelity. The initial nitration step involves the generation of the nitronium ion in a concentrated sulfuric acid medium, which attacks the electron-rich positions of the pyridine ring; maintaining the temperature between 20-25°C is critical to control the exothermic nature of this reaction and prevent thermal runaway or decomposition of the starting material. Following isolation, the hydroxyl group is converted to a chloro substituent via an Appel-type reaction mechanism facilitated by phosphorus halides, creating a superior leaving group for the subsequent cross-coupling reaction. The third step employs a Suzuki-Miyaura type coupling mechanism where the palladium catalyst, specifically Pd(dppf)Cl2, facilitates the transmetallation of the methyl group from the boron species to the pyridine ring in the presence of a cesium carbonate base. This step is pivotal as it establishes the carbon-carbon bond under relatively mild heating conditions of 80-90°C, demonstrating the robustness of the catalytic system even in the presence of electron-withdrawing nitro groups.
Impurity control is rigorously managed throughout the process by optimizing stoichiometric ratios and reaction times to minimize side reactions such as over-chlorination or incomplete oxidation. In the final oxidation stage, selenium dioxide acts as a specific oxidant that targets the benzylic-like methyl position adjacent to the nitrogen heteroatom, converting it to the aldehyde functionality while leaving the nitro and fluoro substituents intact. The use of dioxane as a solvent in both the coupling and oxidation steps ensures solubility of the intermediates and facilitates heat transfer, which is essential for maintaining reaction homogeneity and reproducibility. Workup procedures involving extraction with ethyl acetate and pH adjustment with saturated sodium bicarbonate effectively remove acidic by-products and residual metal catalysts, contributing to the high purity profiles observed in the experimental examples. This comprehensive mechanistic understanding allows process chemists to troubleshoot potential deviations and ensures that the commercial scale-up of complex pharmaceutical intermediates can be executed with confidence and consistency.
How to Synthesize 3-Fluoro-5-Nitropyridine-2-Formaldehyde Efficiently
Implementing this synthesis requires strict adherence to the specified reaction conditions and reagent grades to replicate the high yields reported in the patent literature. The process begins with the careful mixing of starting materials under inert atmosphere where applicable, followed by precise temperature ramping and monitoring to ensure each transformation proceeds to completion before moving to the next stage. Operators must be trained in handling corrosive acids and toxic oxidants safely, utilizing appropriate personal protective equipment and engineering controls to mitigate occupational hazards associated with chemical manufacturing. Detailed standard operating procedures should be established for the workup and purification phases to guarantee that the final isolate meets the required specifications for water content, residual solvents, and heavy metals. The standardized synthesis steps provided below serve as a foundational guide for laboratory validation and subsequent technology transfer to pilot or production scales.
- Nitration of 3-fluoro-2-hydroxypyridine with concentrated sulfuric acid and 65% nitric acid at 20-25°C to form Compound B.
- Chlorination of Compound B using phosphorus oxychloride and phosphorus pentachloride at 0-3°C followed by heating to 60°C to yield Compound C.
- Palladium-catalyzed methylation of Compound C with trimethylboroxine and cesium carbonate in dioxane at 80-90°C to generate Compound D.
- Selective oxidation of Compound D using selenium dioxide in dioxane at 100-110°C to obtain the final 3-fluoro-5-nitropyridine-2-formaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial value to procurement managers and supply chain directors by significantly reducing the complexity and cost associated with sourcing high-value fluorinated intermediates. The elimination of cumbersome protecting group manipulations and the use of commercially available starting materials streamline the manufacturing process, leading to drastic simplification of the supply chain and reduced dependency on exotic reagents that may face availability constraints. By achieving high yields at each step, the overall material throughput is maximized, which directly translates to substantial cost savings in raw material consumption and waste disposal fees for the manufacturing facility. Furthermore, the robustness of the reaction conditions allows for flexible production scheduling and easier scale-up, ensuring that supply continuity can be maintained even during periods of high market demand or logistical disruptions. These operational efficiencies enable suppliers to offer more competitive pricing structures while maintaining healthy margins, creating a win-win scenario for both manufacturers and their downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive and readily available reagents such as concentrated sulfuric acid and selenium dioxide instead of costly proprietary catalysts or oxidants. By avoiding the need for cryogenic conditions or ultra-high pressure equipment, the capital expenditure required for setting up production lines is drastically simplified, allowing for faster return on investment. The high selectivity of the reactions minimizes the formation of by-products, thereby reducing the volume of solvent and energy required for purification processes like chromatography or distillation. Consequently, the overall cost of goods sold (COGS) is lowered, enabling more aggressive pricing strategies in the competitive landscape of fine chemical manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the synthetic route relies on commodity chemicals that are widely produced and stocked by multiple global vendors, mitigating the risk of single-source supply failures. The moderate reaction temperatures and pressures reduce the strain on production equipment, leading to less frequent maintenance downtime and higher asset utilization rates within the manufacturing plant. This operational stability ensures that delivery schedules can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing drug developers to adhere to their clinical trial timelines without delay. Additionally, the simplified logistics of handling fewer hazardous specialty chemicals streamline the transportation and storage requirements, further enhancing the resilience of the supply network against external shocks.
- Scalability and Environmental Compliance: The method is inherently designed for scalability, with reaction parameters that can be easily translated from laboratory glassware to industrial reactors without significant loss of efficiency or safety. The reduction in waste generation through high atom economy aligns with increasingly stringent environmental regulations, lowering the burden of effluent treatment and helping companies meet their sustainability goals. By minimizing the use of heavy metal catalysts in the final steps and employing efficient workup procedures, the environmental footprint of the manufacturing process is substantially reduced. This commitment to green chemistry principles not only ensures regulatory compliance but also enhances the corporate reputation of the manufacturer as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific pyridine derivative, based on the detailed disclosures within the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding process adoption and material sourcing strategies for their specific project needs. The answers provided reflect the consensus of technical expertise derived from the experimental data and process descriptions found in the intellectual property records.
Q: What are the critical temperature controls in the nitration step of this synthesis?
A: The nitration reaction requires strict temperature maintenance between 20-25°C during the dropwise addition of 65% nitric acid to prevent over-nitration and ensure safety, followed by a heat preservation period to complete the conversion to Compound B.
Q: How does this method improve purity compared to traditional routes?
A: By utilizing a specific sequence of chlorination followed by palladium-catalyzed methylation and selective selenium dioxide oxidation, the process minimizes side reactions and impurities, achieving final product purity levels exceeding 98%.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method employs readily available raw materials and standard reaction conditions such as reflux in dioxane and controlled acid additions, making it highly adaptable for commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Fluoro-5-Nitropyridine-2-Formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability regardless of the volume required. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications for every batch of 3-fluoro-5-nitropyridine-2-formaldehyde we produce. We understand the critical nature of supply chain continuity in the pharmaceutical sector and have implemented robust quality management systems to guarantee consistency and compliance with international regulatory standards. Our team of expert chemists is dedicated to continuous process improvement, ensuring that we remain at the forefront of synthetic technology to deliver the highest quality intermediates to our global partners.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique development requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain and reduce overall project expenditures. Let us be your trusted partner in bringing innovative therapies to market faster and more efficiently through our superior chemical manufacturing capabilities and unwavering commitment to excellence.
