Scalable Synthesis Of Indolinobenzodiazepine Derivatives For Commercial Drug Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those serving as critical payloads or linkers in advanced therapeutic modalities like antibody-drug conjugates (ADCs). Patent CN115448926A introduces a highly efficient methodology for the preparation of indolinobenzodiazepine derivatives, addressing the longstanding challenges associated with constructing this privileged structural motif. The disclosed process leverages a strategic combination of iron-mediated reductive cyclization and palladium-catalyzed transfer hydrogenation, offering a distinct advantage over traditional methods that often rely on costly reagents or harsh reaction conditions. By optimizing solvent systems and catalyst loading, this invention provides a pathway that is not only chemically elegant but also commercially viable for the production of high-purity pharmaceutical intermediates. For R&D teams and procurement specialists alike, understanding the nuances of this patented technology is essential for securing a reliable supply chain for next-generation oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzodiazepine-fused indoline systems has been plagued by inefficiencies that hinder large-scale adoption. Conventional routes frequently employ stoichiometric amounts of expensive transition metal catalysts or require high-pressure hydrogenation equipment, which introduces significant safety risks and capital expenditure barriers for manufacturing facilities. Furthermore, traditional nitro-reduction steps often generate substantial quantities of metal waste, complicating downstream purification and environmental compliance. The reliance on sensitive reagents can also lead to inconsistent batch-to-batch quality, with impurities arising from over-reduction or incomplete cyclization proving difficult to remove without extensive chromatography. These factors collectively contribute to prolonged lead times and inflated production costs, making it challenging for suppliers to meet the rigorous demands of global pharmaceutical clients who require consistent quality and competitive pricing for their active pharmaceutical ingredient (API) precursors.
The Novel Approach
In stark contrast, the methodology outlined in CN115448926A presents a streamlined solution that mitigates these operational bottlenecks through the use of earth-abundant iron powder for the critical cyclization step. This approach eliminates the need for precious metal catalysts in the initial ring-closing phase, drastically reducing raw material expenses while maintaining high conversion rates. The subsequent debenzylation step utilizes a transfer hydrogenation strategy with 1,4-cyclohexadiene and a supported palladium catalyst, which allows for mild reaction conditions and simplified workup procedures. This dual-strategy ensures that the process remains robust even when scaled to multi-kilogram batches, as evidenced by the detailed experimental examples provided in the patent documentation. By prioritizing reagent availability and process safety, this novel approach establishes a new standard for the commercial scale-up of complex heterocyclic intermediates, enabling manufacturers to deliver products with superior purity profiles and reduced environmental impact.
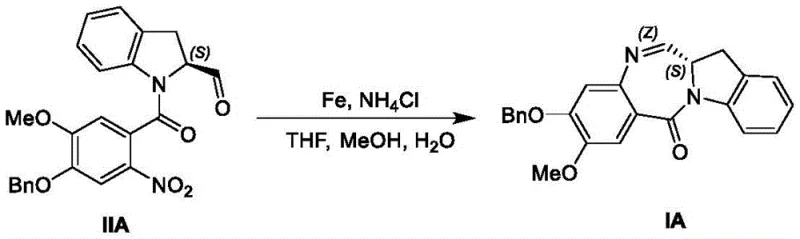
Mechanistic Insights into Iron-Mediated Reductive Cyclization
The core innovation of this synthetic route lies in the mechanistic efficiency of the iron-mediated reduction of the nitro group, which triggers an intramolecular cyclization to form the seven-membered diazepine ring. In the presence of ammonium chloride within a mixed solvent system of THF, methanol, and water, iron acts as a single-electron donor, facilitating the stepwise reduction of the nitro functionality to a hydroxylamine and subsequently to an amine. This newly formed amine immediately attacks the adjacent aldehyde or ketone moiety, driving the equilibrium towards the formation of the stable imine bond characteristic of the indolinobenzodiazepine core. The choice of solvent mixture is critical, as it ensures the solubility of the organic substrate while allowing the inorganic salts to function effectively at the interface. This mechanism avoids the formation of toxic byproducts often associated with other reduction methods, thereby simplifying the isolation of the intermediate compound (Formula I) and ensuring that the stereochemical integrity of the molecule is preserved throughout the transformation.
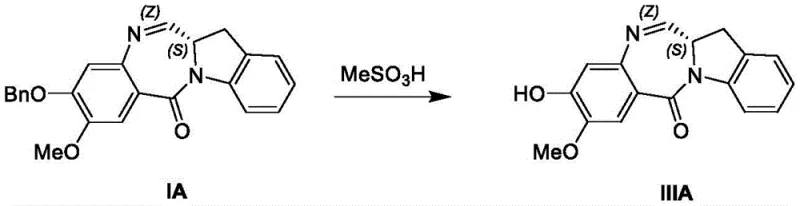
Following the cyclization, the process employs a sophisticated purification strategy to manage impurity profiles, which is vital for meeting regulatory standards. The patent details specific recrystallization protocols using binary solvent systems such as dichloromethane and ethanol or toluene and acetonitrile. These systems are designed to selectively precipitate the desired product while keeping soluble impurities in the mother liquor. The thermal parameters, such as heating to 80°C in toluene followed by controlled cooling, are optimized to promote the growth of well-defined crystals, which inherently possess higher purity than amorphous solids. This focus on crystallization engineering demonstrates a deep understanding of solid-state chemistry, ensuring that the final high-purity indolinobenzodiazepine derivatives are suitable for direct use in subsequent conjugation reactions without the need for further chromatographic purification, thus saving both time and resources in the overall manufacturing workflow.
How to Synthesize Indolinobenzodiazepine Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to strict control over reaction parameters, particularly temperature and reagent addition rates, to maximize yield and safety. The process begins with the suspension of the nitro-precursor in a carefully balanced solvent mixture, followed by the sequential addition of ammonium chloride and iron powder under an inert atmosphere to prevent oxidation. Maintaining the reaction temperature at approximately 65°C for an extended period, typically around 48 hours, is crucial for driving the reduction to completion without degrading the sensitive imine linkage. Once the cyclization is complete, the workup involves filtration to remove iron sludge, followed by solvent exchange and crystallization steps that are critical for achieving the target purity specifications. The subsequent debenzylation step requires precise catalyst loading and the controlled addition of the hydrogen donor to ensure selective removal of the benzyl protecting group without affecting other functional groups on the scaffold.
- Perform reductive cyclization of the nitro-precursor (Formula II) using iron powder and ammonium chloride in a THF/methanol/water solvent system at 65°C.
- Purify the resulting imine intermediate (Formula I) via recrystallization using dichloromethane and ethanol or toluene and acetonitrile mixtures.
- Execute catalytic debenzylation using 5% Pd/Alox and 1,4-cyclohexadiene in ethanol/DMSO at 50°C to yield the final phenol derivative (Formula III).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards iron-mediated chemistry represents a significant opportunity for cost reduction in API manufacturing. Iron powder is vastly cheaper and more readily available than noble metal catalysts like palladium or platinum, which are subject to volatile market prices and supply constraints. By reserving the use of palladium only for the final debenzylation step and utilizing a supported catalyst that can be easily filtered, the overall metal consumption is minimized. This reduction in precious metal usage not only lowers the direct material cost but also simplifies the recovery and recycling processes, further enhancing the economic viability of the route. Additionally, the use of common industrial solvents like THF, ethanol, and toluene ensures that sourcing is straightforward and不受 geopolitical disruptions, providing a stable foundation for long-term supply agreements with pharmaceutical partners.
- Cost Reduction in Manufacturing: The substitution of expensive hydrogenation catalysts with inexpensive iron powder for the primary cyclization step dramatically lowers the bill of materials. This qualitative shift in reagent strategy removes the dependency on fluctuating precious metal markets, allowing for more predictable budgeting and pricing models for clients. Furthermore, the simplified workup procedures reduce the labor and utility costs associated with complex purification trains, contributing to a leaner and more efficient production process that translates into tangible savings for the end customer.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as iron and ammonium chloride ensures that the supply chain is resilient against shortages that often plague specialized reagents. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites without significant re-validation, providing flexibility in production planning. This reliability is crucial for maintaining continuous supply to clinical and commercial programs, minimizing the risk of delays that could impact patient access to life-saving therapies derived from these intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are safe to operate in large reactors without the need for high-pressure equipment. The generation of iron oxide waste is manageable and less hazardous compared to heavy metal waste streams, aligning with modern green chemistry principles and easing the burden on waste treatment facilities. This environmental compatibility facilitates smoother regulatory approvals and supports the sustainability goals of major pharmaceutical companies seeking to reduce the carbon footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding these details is critical for process chemists and engineers who are evaluating the feasibility of adopting this technology for their own production lines. The answers are derived directly from the experimental data and claims presented in the patent, ensuring accuracy and relevance to real-world manufacturing scenarios. By clarifying these points, we aim to provide transparency and build confidence in the technical robustness of the described methodology.
Q: What are the key advantages of the iron-mediated reduction method described in CN115448926A?
A: The use of iron powder and ammonium chloride offers a cost-effective and scalable alternative to expensive hydrogenation catalysts or hazardous hydride reagents for the initial cyclization step, significantly reducing raw material costs.
Q: How is high purity achieved in the final indolinobenzodiazepine product?
A: High purity is ensured through specific recrystallization protocols, such as using toluene and acetonitrile mixtures at elevated temperatures, and precise control over the palladium-catalyzed debenzylation conditions to minimize side reactions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly outlines conditions optimized for large-scale production, including the use of readily available solvents like THF and ethanol, and robust catalysts like Pd/Alox that facilitate easier filtration and recovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolinobenzodiazepine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and reaction conditions required for the synthesis of indolinobenzodiazepine derivatives, guaranteeing a seamless transition from laboratory scale to full commercial manufacturing.
We invite you to collaborate with us to optimize your supply chain and accelerate your project timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us be your partner in delivering excellence, ensuring that your critical pharmaceutical projects proceed without interruption and with the highest level of quality assurance.
