Scalable Synthesis of Semi-Sandwich Organometallic Macrocycles for Advanced Catalysis
Scalable Synthesis of Semi-Sandwich Organometallic Macrocycles for Advanced Catalysis
The landscape of supramolecular chemistry and nanotechnology has been significantly advanced by the innovations detailed in patent CN101693727A, which introduces a robust methodology for constructing tetranuclear and hexanuclear macrocyclic complexes. This technology leverages the unique coordination geometry of half-sandwich organometallic units containing Iridium, Rhodium, or Ruthenium to create ordered, nano-aperture structures through spontaneous self-assembly. For industrial partners seeking reliable organometallic macrocycle suppliers, this patent represents a pivotal shift from laborious stepwise synthesis to efficient, one-pot assembly processes that maintain high structural integrity. The ability to tune the nuclearity of the complex (n=4 or n=6) simply by altering the pyridine-substituted beta-diketone ligand offers unprecedented flexibility for designing materials with specific electronic or magnetic properties. Furthermore, the compatibility of this method with standard laboratory equipment and ambient reaction conditions suggests a clear pathway for commercial scale-up of complex organometallic intermediates without requiring exotic high-pressure or high-temperature infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing high-nuclearity organometallic cages often suffer from low thermodynamic stability and poor selectivity, frequently resulting in oligomeric mixtures that are difficult to separate and purify. Conventional routes typically rely on rigid, pre-organized ligands that require extensive synthetic effort to prepare, driving up the cost reduction in advanced material manufacturing and limiting the diversity of accessible structures. Moreover, the use of labile metal-ligand bonds in older methodologies often necessitates strict exclusion of moisture and oxygen throughout the entire process, creating significant bottlenecks for supply chain reliability and batch-to-batch consistency. The purification of these conventional products often involves challenging chromatographic separations due to the similar polarity of linear oligomers and the desired cyclic products, leading to substantial material loss and extended production lead times. Additionally, many historical methods utilize coordinating anions that can compete with the ligand for metal binding sites, thereby disrupting the formation of the desired nano-aperture architecture and compromising the functional performance of the final material.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a dynamic self-assembly strategy driven by the thermodynamic stability of the final macrocyclic product, effectively correcting errors during the formation process to ensure high purity. By employing chloro-bridged dimers of pentamethylcyclopentadienyl metal dichlorides as precursors, the method capitalizes on the lability of the chloride bridge which is easily displaced by silver salts to generate reactive cationic species in situ. This activation step allows for the rapid and directional coordination of pyridine-substituted beta-diketone ligands, which act as linear spacers to bridge the metal centers into defined ring structures. The steric bulk of the pentamethylcyclopentadienyl (Cp*) group plays a critical role by shielding one face of the metal center, thereby enforcing a specific coordination geometry that favors cyclization over polymerization. This strategic design results in a synthesis protocol that is not only operationally simple but also highly scalable, making it an ideal candidate for reducing lead time for high-purity organometallic complexes in industrial settings.
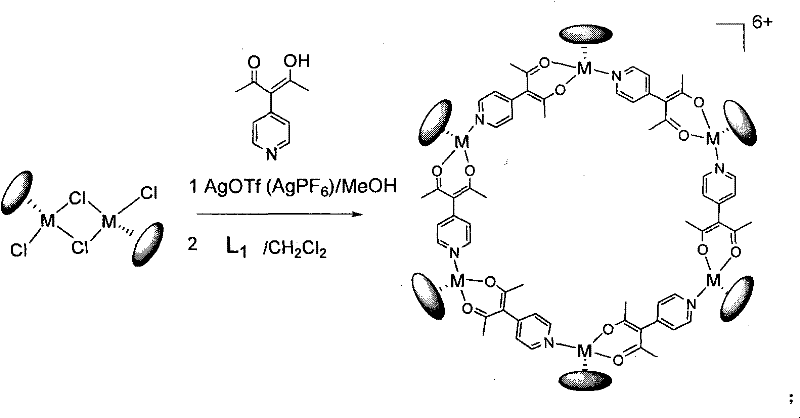
The visual representation of the hexanuclear assembly process highlights the elegance of this coordination-driven synthesis, where six metal centers are seamlessly linked by six organic ligands to form a large, stable ring. The reaction conditions are remarkably mild, proceeding efficiently at room temperature in methanol, which eliminates the energy costs associated with heating or cooling large reactor vessels. The precipitation of silver chloride serves as a visual indicator of reaction progress and drives the equilibrium forward by removing chloride ions from the system, ensuring near-quantitative conversion of the starting materials. This level of control over the supramolecular architecture is essential for applications requiring precise molecular recognition or gas storage capabilities, as even minor structural defects can drastically alter the pore size and chemical environment within the cavity.
Mechanistic Insights into Self-Assembly Coordination Chemistry
The mechanistic foundation of this synthesis relies on the hard-soft acid-base theory, where the soft silver cation preferentially binds to the soft chloride ligands on the metal dimer, liberating the hard metal center for coordination with the nitrogen donor of the pyridine ligand. Once the chloride bridges are cleaved by the silver salt (AgOTf or AgPF6), the resulting cationic metal species [(Cp*M)(MeOH)x]n+ becomes highly electrophilic and ready to accept electron density from the incoming ligand. The pyridine-substituted beta-diketone ligand acts as a bifunctional connector, utilizing its beta-diketone moiety to chelate one metal center while the pyridine nitrogen coordinates to a neighboring metal center, thus propagating the growth of the ring. The stoichiometry of the reaction is critical, with a 1:1 molar ratio between the metal precursor and the ligand favoring the formation of discrete cyclic oligomers rather than infinite coordination polymers. The choice of counter-anion, either triflate or hexafluorophosphate, is also mechanistically significant as these weakly coordinating anions remain in the outer sphere, preventing them from blocking the coordination sites needed for ring closure.
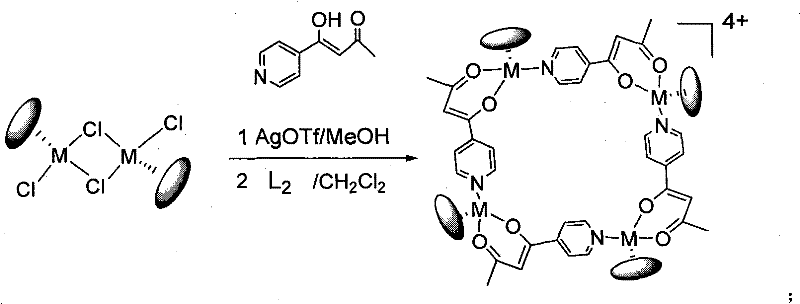
Impurity control in this system is inherently managed by the reversibility of the coordination bonds and the insolubility of the silver chloride byproduct, which can be easily removed via simple filtration. Any misaligned intermediates formed during the early stages of the reaction can dissociate and re-associate correctly due to the dynamic nature of the metal-ligand bonds under the reaction conditions, a phenomenon known as error-checking in self-assembly. This thermodynamic control ensures that the final isolated product is predominantly the desired tetranuclear or hexanuclear species, minimizing the presence of linear or irregular oligomeric impurities that often plague kinetically controlled syntheses. The structural rigidity imparted by the chelating beta-diketone group further stabilizes the complex against ligand exchange or decomposition during workup and storage, ensuring that the high-purity organometallic complexes retain their functionality upon delivery to the end user.
How to Synthesize Tetranuclear Hexanuclear Macrocyclic Complexes Efficiently
The synthesis protocol outlined in the patent provides a straightforward roadmap for producing these sophisticated materials, beginning with the preparation of anhydrous and oxygen-free solvents to prevent premature oxidation of the sensitive metal centers. Operators must carefully weigh the chloro-bridged metal dimer and the silver salt to achieve the precise 1:4 molar ratio required for complete halide abstraction, followed by stirring in the dark to prevent photo-induced degradation of the silver species. After the initial activation period of 8-12 hours, the reaction mixture is filtered to remove the silver chloride precipitate, yielding a clear solution of the activated metal cation ready for the addition of the ligand. The subsequent addition of the pyridine-substituted beta-diketone ligand initiates the self-assembly process, which proceeds to completion over another 8-12 hour period at ambient temperature without the need for external heating sources. Detailed standardized synthesis steps see the guide below.
- Dissolve the chloro-bridged dimer (Ir, Rh, or Ru) in anhydrous methanol and react with AgOTf or AgPF6 at a 1: 4 molar ratio to remove chloride ions.
- Filter the resulting silver chloride precipitate under inert atmosphere to obtain the activated cationic metal species.
- Add the pyridine-substituted beta-diketone ligand to the filtrate and stir at room temperature for 8-12 hours to induce self-assembly into the macrocyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial cost savings by simplifying the synthetic route and reducing the number of unit operations required to isolate the final product. The elimination of complex purification steps such as column chromatography or recrystallization from multiple solvent systems significantly lowers the consumption of silica gel and organic solvents, directly impacting the operational expenditure of the manufacturing facility. Furthermore, the use of commercially available starting materials like pentamethylcyclopentadienyl metal dichloride dimers ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors that often have long lead times. The high selectivity of the reaction means that raw material utilization is maximized, with minimal waste generated from side products, aligning with modern green chemistry principles and reducing the burden on waste treatment infrastructure. These factors combined create a compelling economic case for adopting this methodology for the commercial production of high-value organometallic materials.
- Cost Reduction in Manufacturing: The streamlined two-step process eliminates the need for expensive catalysts or harsh reagents, relying instead on simple silver salts and common organic solvents to drive the reaction to completion. By avoiding high-energy inputs such as prolonged heating or cryogenic cooling, the utility costs associated with production are drastically simplified, allowing for more competitive pricing structures in the global market. The high yield reported in the examples indicates that the process is atom-economical, ensuring that the majority of the input mass is converted into saleable product rather than discarded waste. This efficiency translates directly into lower cost of goods sold (COGS), enabling manufacturers to offer high-purity organometallic complexes at price points that were previously unattainable with legacy synthetic methods.
- Enhanced Supply Chain Reliability: The reliance on robust, shelf-stable reagents such as AgOTf and AgPF6 ensures that production schedules are not disrupted by the degradation of sensitive starting materials. The simplicity of the workup procedure, which involves basic filtration and solvent evaporation, reduces the dependency on specialized equipment or highly skilled labor, making it easier to transfer the technology between different manufacturing sites. This operational flexibility enhances supply continuity, as production can be rapidly scaled up or down in response to market demand without the need for extensive retooling or process re-validation. Additionally, the stability of the final macrocyclic products facilitates easier storage and transportation, reducing the risk of spoilage during logistics and ensuring that customers receive materials that meet stringent quality specifications.
- Scalability and Environmental Compliance: The reaction operates effectively at room temperature and atmospheric pressure, removing the safety hazards and engineering challenges associated with high-pressure reactors or extreme thermal conditions. The primary byproduct, silver chloride, is a solid that can be easily filtered and potentially recycled to recover silver, minimizing the environmental footprint of the process and simplifying waste disposal compliance. The use of methanol and dichloromethane, while requiring standard solvent handling protocols, avoids the use of more toxic or persistent organic pollutants, aligning with increasingly strict environmental regulations in the chemical industry. This inherent safety and environmental compatibility makes the process highly attractive for large-scale manufacturing, where regulatory compliance and worker safety are paramount concerns for sustainable operations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these macrocyclic complexes, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating these materials into their own catalytic or material science workflows. The answers provided reflect the specific conditions and outcomes observed in the patented examples, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of the self-assembly method described in CN101693727A?
A: The self-assembly method offers high selectivity and yield under mild conditions (room temperature), eliminating the need for harsh reagents or complex multi-step protection/deprotection sequences typically required in traditional macrocyclization.
Q: Which metals can be utilized in this semi-sandwich macrocyclic structure?
A: The patent specifically discloses the successful formation of stable macrocycles using Iridium (Ir), Rhodium (Rh), and Ruthenium (Ru) centers coordinated with pentamethylcyclopentadienyl (Cp*) ligands.
Q: How does the choice of anion affect the final product properties?
A: The use of non-coordinating anions such as trifluoromethanesulfonate (OTf-) or hexafluorophosphate (PF6-) ensures the stability of the cationic macrocyclic complex and enhances solubility in organic solvents like dichloromethane.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetranuclear Hexanuclear Macrocyclic Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the self-assembly technologies described in CN101693727A for advancing the fields of nanotechnology and homogeneous catalysis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial application is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying the structural integrity and purity of complex organometallic species, guaranteeing that every batch meets your stringent purity specifications. We understand that consistency is key in high-tech applications, and our dedicated process engineering team works tirelessly to optimize every parameter of the synthesis to maximize yield and minimize variability.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with a Customized Cost-Saving Analysis tailored to your volume needs. Whether you require specific COA data for regulatory filings or detailed route feasibility assessments for new derivative structures, our experts are ready to provide the technical depth and commercial agility you need. Partner with us to leverage this cutting-edge chemistry and secure a competitive advantage in the development of next-generation functional materials and catalytic systems.
