Advanced Synthesis of 3D Polyimidazolium Salt Cage Compounds for High-Performance Materials
Advanced Synthesis of 3D Polyimidazolium Salt Cage Compounds for High-Performance Materials
The development of sophisticated three-dimensional molecular architectures represents a frontier in modern synthetic chemistry, particularly for applications requiring precise host-guest interactions or specific electronic properties. Patent CN109232586B introduces a groundbreaking methodology for the efficient synthesis of novel three-dimensional polyimidazolium salt cage-shaped compounds. This technology leverages a metal carbene template reaction strategy to construct complex 3D polyimidazolium salt molecules of varying sizes, overcoming significant limitations associated with traditional synthetic routes. By utilizing imidazolium salt compounds as starting materials and employing a photochemical post-modification of an N-heterocyclic carbene organic metal framework, this process achieves remarkable control over molecular topology. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent outlines a robust pathway to access high-value cage compounds that were previously difficult to manufacture at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyimidazolyl cyclophane compounds has relied on stepwise nitrogen alkylation reactions. This conventional approach typically involves reacting dihalogenated alkanes or arenes with imidazole derivatives under basic conditions to form alkane-bridged bisimidazole compounds, followed by further quaternization to close the ring. However, as the number of imidazole groups increases, the complexity of the reaction mixture grows exponentially. Traditional methods are plagued by the formation of numerous complex by-products, particularly when attempting to create three-dimensional structures. The statistical nature of these cyclization reactions often leads to linear oligomers or ill-defined polymers rather than the desired discrete cage structures. Furthermore, yields decrease significantly with increased structural complexity, and the separation of the target molecule from the crude reaction mixture becomes an arduous and costly task, often requiring extensive chromatographic purification that is not feasible for commercial scale-up.
The Novel Approach
In stark contrast, the method disclosed in CN109232586B utilizes a metal carbene template strategy that fundamentally alters the reaction landscape. By first assembling a tridentate imidazole salt ligand and then coordinating it with a metal oxide (specifically silver oxide in the preferred embodiments), the system pre-organizes the reactive olefin groups in close proximity. This templating effect drastically reduces the entropic penalty associated with cyclization. The subsequent [2+2] photochemical reaction locks these pre-organized groups into a cyclobutane ring, effectively stitching the 3D cage together with high precision. This approach not only simplifies the operational procedure but also ensures that the reaction proceeds with high selectivity towards the desired cage structure, minimizing the formation of unwanted oligomeric by-products and enabling the synthesis of size-controllable 3D imidazolium salts that are inaccessible via standard alkylation techniques.
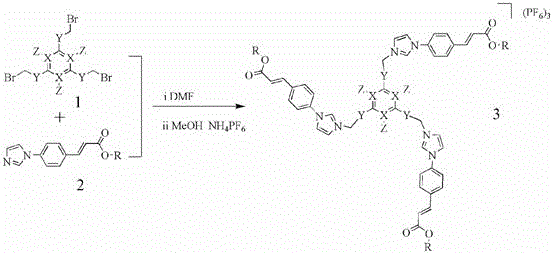
Mechanistic Insights into Metal Carbene Template Photochemical Cyclization
The core of this innovation lies in the formation and utilization of the silver carbene complex. In the first stage, the tridentate imidazole salt ligand reacts with silver oxide (Ag2O) to form a stable silver carbene complex. In this intermediate, the silver ions coordinate with the carbene carbons generated from the imidazolium salts, effectively acting as a structural scaffold. This coordination holds the pendant styryl groups (derived from the 4-imidazole-phenylacrylate moieties) in a specific spatial arrangement. The proximity and orientation of the double bonds are critical for the subsequent photochemical step. When irradiated with UV light (typically at 365 nm), the pre-aligned olefins undergo a [2+2] cycloaddition reaction. This photochemical process is highly efficient because the metal template has already done the heavy lifting of overcoming the entropic barrier, bringing the reactive centers within bonding distance. The result is the formation of a cyclobutane ring that covalently links the arms of the ligand, creating the rigid 3D cage architecture while the silver ions remain coordinated.
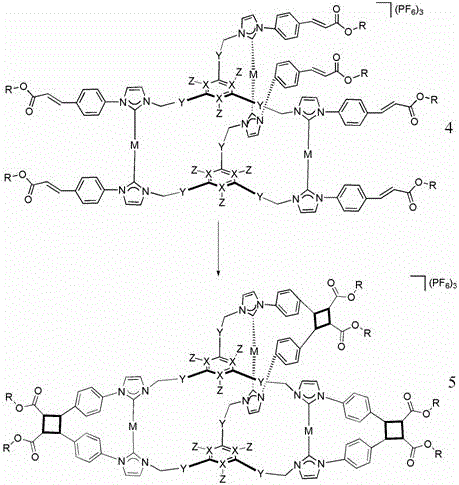
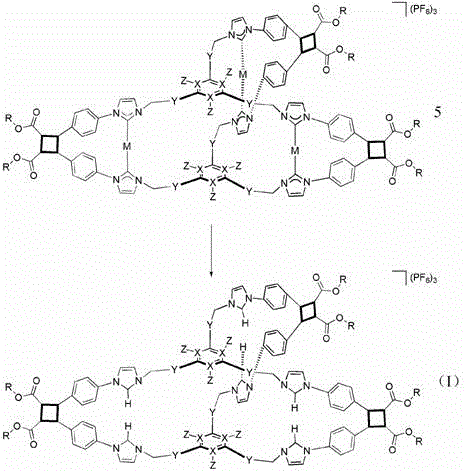
Impurity control in this process is inherently superior due to the template-directed nature of the reaction. In traditional statistical cyclizations, any two reactive ends can meet, leading to a distribution of ring sizes and linear chains. Here, the stoichiometry and geometry of the silver carbene complex dictate that only specific intramolecular reactions can occur. The metal center enforces a specific geometry that favors the formation of the desired cyclobutane linkage between the intended pairs of olefins. Following the photocyclization, the silver template is removed via treatment with ammonium chloride, which precipitates silver chloride and releases the free polyimidazolium salt cage. This demetallation step is clean and quantitative, ensuring that the final product is free from heavy metal contaminants, a critical requirement for pharmaceutical and electronic applications. The entire sequence demonstrates how supramolecular principles can be harnessed to drive covalent bond formation with high fidelity.
How to Synthesize 3D Polyimidazolium Salt Cage Compounds Efficiently
The synthesis protocol described in the patent provides a clear, step-by-step guide for producing these complex architectures. The process begins with the preparation of the tridentate ligand in DMF, followed by metalation with Ag2O in acetonitrile. The key transformation occurs under UV irradiation, where the conversion to the cyclobutane-containing complex is monitored to reach completion. Finally, the silver is stripped to yield the target cage compound. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and reaction times optimized for maximum yield and purity, are outlined in the guide below.
- Prepare tridentate imidazole salt ligands by reacting tribenzylbromoarene with 4-imidazole-phenylacrylate in DMF at 110°C.
- Form the silver carbene complex by reacting the ligand with Ag2O in acetonitrile at 55°C under dark conditions.
- Perform [2+2] photochemical template reaction using 365 nm UV light to close the cyclobutane ring.
- Remove the silver template using ammonium chloride to obtain the final 3D polyimidazolium salt cage compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional alkylation methods to this metal carbene template technology offers substantial strategic benefits. The primary advantage lies in the drastic simplification of the purification process. Because the template reaction minimizes the formation of complex by-product mixtures, the need for resource-intensive chromatographic separations is significantly reduced or eliminated. This directly translates to lower manufacturing costs and reduced solvent consumption, aligning with green chemistry principles. Furthermore, the high yields reported in the patent examples demonstrate a robust process that maximizes raw material utilization, ensuring that expensive precursors are converted efficiently into the final high-value product.
- Cost Reduction in Manufacturing: The elimination of difficult separation steps and the high efficiency of the photochemical cyclization lead to significant cost savings. By avoiding the generation of vast amounts of oligomeric waste typical of statistical cyclizations, the process reduces waste disposal costs and improves overall atom economy. The use of readily available starting materials like tribenzylbromoarenes and imidazole derivatives further supports a cost-effective supply chain, making the commercial production of these specialized cage compounds economically viable for the first time.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The reactions are performed under relatively mild temperatures (e.g., 55°C for metalation) and standard atmospheric pressure, reducing the need for specialized high-pressure equipment that can be prone to maintenance issues. The scalability of the photochemical step, combined with the high conversion rates observed, ensures consistent batch-to-batch quality. This reliability is crucial for downstream customers who require a steady supply of high-purity intermediates for their own manufacturing processes without the risk of production delays caused by complex synthesis failures.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operational simplicity. The use of common solvents like DMF, methanol, and acetonitrile facilitates easy integration into existing manufacturing infrastructure. Moreover, the high selectivity of the reaction reduces the environmental footprint by minimizing chemical waste. The final demetallation step produces silver chloride, which can be easily recovered and recycled, adding a layer of sustainability to the process. This alignment with environmental compliance standards makes the technology attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes of complex specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these 3D polyimidazolium salt cage compounds. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and technical stakeholders.
Q: What are the advantages of the metal carbene template method over traditional alkylation?
A: The metal carbene template method offers mild reaction conditions, shorter reaction times, and significantly higher yields compared to traditional methods which often suffer from complex by-products and low yields due to statistical cyclization issues.
Q: Can the size of the 3D imidazolium cage be controlled?
A: Yes, by selecting different tribenzylbromoarene starting materials (varying X, Y, Z groups), the method allows for the synthesis of three-dimensional imidazolium salt compounds with controllable sizes and cavity dimensions.
Q: What is the typical yield for the photochemical cyclization step?
A: The patent data indicates that the photochemical conversion rate for the cyclization step can reach 100%, demonstrating high efficiency in forming the cyclobutane-containing complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyimidazolium Salt Cage Compound Supplier
The synthesis method detailed in CN109232586B represents a significant leap forward in the construction of complex 3D molecular cages, offering a pathway to materials with unique properties for pharmaceutical and electronic applications. NINGBO INNO PHARMCHEM stands ready to leverage this advanced technology to support your development goals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for high-performance intermediates. We understand the critical nature of supply continuity and are committed to delivering consistent quality.
We invite you to discuss how our capabilities can accelerate your project timelines. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target molecules. Let us collaborate to bring these innovative 3D cage compounds from the laboratory to the global market efficiently and sustainably.
