Revolutionizing Herbicide Intermediate Production via Direct C-H Functionalization Technology
Revolutionizing Herbicide Intermediate Production via Direct C-H Functionalization Technology
The global agrochemical industry is constantly seeking more sustainable and cost-effective pathways for producing high-volume herbicides, and the recent disclosure in patent CN110872283A presents a transformative approach to synthesizing 2-aryloxynicotinamide compounds. This intellectual property introduces a novel methodology that bypasses traditional reliance on halogenated pyridine derivatives, instead utilizing direct carbon-hydrogen bond functionalization to construct the critical ether linkage found in molecules like diflufenican. By shifting the synthetic paradigm from nucleophilic aromatic substitution to copper-catalyzed C-H activation, this technology addresses long-standing pain points regarding waste generation and raw material costs that have plagued the sector for decades. The structural versatility of the resulting compounds, as defined by the general formula (I), opens new avenues not only for established herbicides but also for the discovery of next-generation crop protection agents with improved safety profiles. For R&D teams and procurement strategists alike, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-purity agrochemical intermediates.
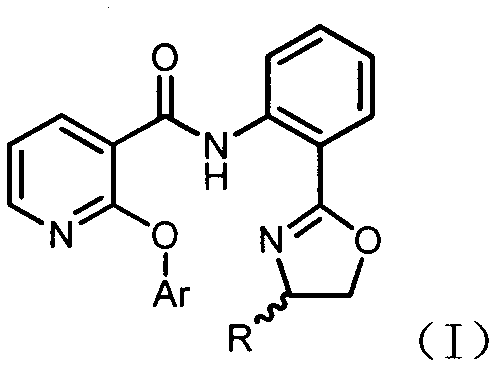
Furthermore, the strategic importance of this invention lies in its ability to utilize nicotinic acid, a ubiquitous and inexpensive commodity chemical, as the foundational building block. This stands in stark contrast to legacy processes that depend on specialized, often imported, chlorinated heterocycles which are subject to volatile pricing and supply chain disruptions. The patent outlines a robust sequence involving condensation followed by a key etherification step, demonstrating high efficiency and selectivity that is critical for industrial adoption. As we delve deeper into the technical specifics, it becomes evident that this methodology represents a significant leap forward in green chemistry principles applied to large-scale agrochemical manufacturing. Companies capable of mastering this route will be well-positioned to offer reliable agrochemical intermediate supplier services with superior margin structures and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aryloxynicotinamide scaffolds, which are pivotal for herbicides like diflufenican, has relied heavily on the use of 2-chloronicotinic acid or similar halogenated precursors. These traditional routes typically necessitate harsh reaction conditions, such as high-temperature Ullmann coupling or strong base-mediated nucleophilic substitutions, to displace the chlorine atom with the desired phenolic moiety. Such processes are inherently inefficient, often suffering from moderate yields and the generation of substantial quantities of salt waste and chlorine-containing byproducts that require complex and costly disposal protocols. Moreover, the starting materials themselves are derived from energy-intensive chlorination processes, making the overall carbon footprint of the synthesis unnecessarily high and susceptible to regulatory scrutiny regarding halogenated waste. From a supply chain perspective, reliance on these specific chlorinated intermediates creates a bottleneck, as their production is concentrated among a limited number of manufacturers, leading to potential shortages and price volatility that can disrupt downstream herbicide production schedules.
The Novel Approach
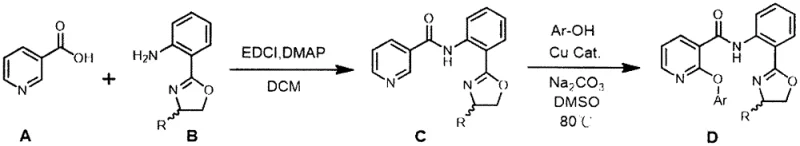
In a groundbreaking departure from these legacy methods, the technology described in CN110872283A leverages a copper-catalyzed direct C-H etherification strategy that fundamentally alters the economic and environmental landscape of this synthesis. By employing nicotinic acid and a removable oxazoline-based directing group, the process achieves regioselective functionalization at the C2 position of the pyridine ring without the need for pre-installed leaving groups. This innovative route, illustrated comprehensively in the reaction scheme below, allows for the use of mild conditions and readily available phenols, drastically simplifying the operational complexity and reducing the hazard profile of the manufacturing process. The ability to recycle the directing group after the synthesis further enhances the atom economy, making this a truly sustainable solution for cost reduction in herbicide manufacturing. This approach not only mitigates the risks associated with halogenated waste but also diversifies the supply base by enabling the use of bulk commodity chemicals, thereby ensuring greater supply chain resilience for global agrochemical producers.
Mechanistic Insights into Copper-Catalyzed C-H Etherification
The core of this technological advancement lies in the sophisticated mechanism of the copper-catalyzed C-H activation step, which transforms the nicotinamide intermediate into the desired 2-aryloxy derivative. The reaction proceeds through a coordination-assisted pathway where the oxazoline nitrogen acts as a directing group, chelating the copper catalyst to bring it into close proximity with the target C-H bond at the ortho position of the pyridine ring. This pre-organization lowers the activation energy required for C-H bond cleavage, allowing the reaction to proceed under relatively mild thermal conditions compared to traditional cross-coupling reactions. The subsequent oxidative addition of the phenol and reductive elimination steps are carefully balanced by the choice of ligand and base, ensuring high turnover numbers and minimizing the formation of homocoupling byproducts that often plague copper chemistry. Understanding this mechanistic nuance is vital for R&D directors aiming to optimize the process for commercial scale-up of complex agrochemical intermediates, as slight variations in catalyst loading or base selection can significantly impact the impurity profile and overall yield.
Impurity control is another critical aspect where this mechanism offers distinct advantages over conventional nucleophilic substitution. In traditional S_NAr reactions, there is always a risk of over-reaction or attack at other electrophilic sites, leading to difficult-to-remove impurities that compromise the purity specifications required for regulatory approval. In contrast, the directing group strategy employed here ensures exquisite regioselectivity, effectively shielding other positions on the pyridine ring from functionalization. Furthermore, the hydrolysis step that follows the etherification allows for the cleavage of the amide bond, which serves a dual purpose: it releases the final 2-aryloxynicotinic acid product and simultaneously regenerates the oxazoline-aniline auxiliary. This regeneration capability means that the auxiliary does not become a waste stream but rather a reusable asset, significantly reducing the material cost per kilogram of the final active ingredient and simplifying the downstream purification workflow.
How to Synthesize 2-Aryloxynicotinamide Efficiently
The practical implementation of this synthesis involves a streamlined sequence that begins with the condensation of nicotinic acid and the oxazoline-aniline derivative using standard peptide coupling reagents like EDCI and DMAP. Following the isolation of the nicotinamide intermediate, the key copper-catalyzed etherification is performed in a polar aprotic solvent such as DMSO, utilizing a carbonate base and a copper salt at temperatures around 80°C. The detailed standardized synthesis steps see the guide below for precise stoichiometric ratios and workup procedures that ensure maximum recovery of both the product and the recyclable auxiliary. This protocol has been validated to produce high-purity intermediates suitable for subsequent conversion into final herbicidal active ingredients, demonstrating the robustness of the method across different aryl substituents.
- Condense nicotinic acid with 2-(2-oxazolinyl)aniline using EDCI and DMAP in dichloromethane to form the nicotinamide intermediate.
- Perform copper-catalyzed direct C-H etherification with aryl phenols in DMSO at 80°C to install the aryloxy group.
- Hydrolyze the amide bond under alkaline conditions to recover the directing group and obtain the 2-aryloxynicotinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this C-H activation technology translates into tangible strategic benefits that go beyond mere technical novelty. The shift away from chlorinated starting materials eliminates the need for handling hazardous halogenated waste streams, which significantly reduces the environmental compliance burden and associated disposal costs that can erode profit margins in chemical manufacturing. Additionally, the reliance on nicotinic acid, a globally produced vitamin precursor, ensures a stable and diversified supply base that is less prone to the geopolitical and logistical disruptions often seen with specialized fine chemical intermediates. This stability is crucial for maintaining continuous production schedules for high-volume herbicides, preventing costly downtime and ensuring that customer commitments are met without delay. The overall simplification of the synthetic route also implies a reduction in unit operations, which directly correlates to lower capital expenditure requirements for new production lines and reduced operational overheads.
- Cost Reduction in Manufacturing: The elimination of expensive halogenated precursors and the ability to recycle the directing group create a compelling economic case for adopting this new route. By removing the cost of the chlorine leaving group and the waste treatment associated with it, the variable cost of goods sold is significantly optimized, allowing for more competitive pricing in the global agrochemical market. Furthermore, the milder reaction conditions reduce energy consumption, contributing to lower utility costs and a smaller carbon footprint, which is increasingly valued by end-customers and regulators alike.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like nicotinic acid and simple phenols decouples the production of these critical intermediates from the bottlenecks of specialized halogenation facilities. This diversification of raw material sources enhances supply chain resilience, ensuring that production can continue even if specific niche suppliers face disruptions. The robustness of the chemistry also means that technology transfer to multiple manufacturing sites is feasible, further de-risking the supply chain against regional instabilities or force majeure events.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents and catalysts that are easily sourced in bulk quantities for commercial production. The reduction in chlorine waste aligns perfectly with tightening global environmental regulations, future-proofing the manufacturing process against stricter discharge limits. This proactive approach to green chemistry not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner in the agrochemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway for 2-aryloxynicotinamides. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What is the primary advantage of this C-H activation method over traditional Ullmann coupling?
A: The primary advantage is the avoidance of expensive and hazardous halogenated starting materials like 2-chloronicotinic acid. This method uses cheap nicotinic acid and generates significantly less chlorine-containing waste, improving the environmental profile and reducing raw material costs.
Q: Can the 2-(2-oxazolinyl)aniline auxiliary be recovered?
A: Yes, the patent explicitly details a hydrolysis step where the amide bond is cleaved under alkaline conditions. This allows for the recovery and recycling of the 2-(2-oxazolinyl)aniline directing group, which further enhances the economic viability of the process.
Q: Is this process suitable for large-scale manufacturing of diflufenican?
A: Absolutely. The reaction conditions are relatively mild (80°C) compared to traditional high-temperature Ullmann reactions, and the starting materials are commodity chemicals. The simplified purification and reduced waste load make it highly scalable for commercial agrochemical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryloxynicotinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the C-H activation technologies described in CN110872283A and have invested heavily in mastering these advanced synthetic methodologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-aryloxynicotinamide intermediate meets the exacting standards required for agrochemical registration and formulation. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking to secure their supply chains for critical herbicide intermediates.
We invite you to engage with our technical procurement team to discuss how this novel route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your operation, backed by our real-world manufacturing data. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and profitability in your agrochemical production networks.
