Scalable Synthesis of Noxastota Intermediates via Novel Carbonyl Insertion Technology
The pharmaceutical landscape for treating anemia associated with Chronic Kidney Disease (CKD) has been significantly transformed by Hypoxia Inducible Factor Prolyl Hydroxylase Inhibitors (HIF-PHIs), with Noxastota standing out as a pivotal therapeutic agent. Patent CN113754569A introduces a groundbreaking methodology for synthesizing key intermediates essential for Noxastota production, addressing critical bottlenecks in existing manufacturing protocols. This intellectual property details a robust pathway involving a novel intermediate compound of Formula 5, which serves as a versatile precursor for the final active pharmaceutical ingredient. By leveraging a strategic carbonyl insertion reaction, the disclosed method circumvents the severe safety hazards and exorbitant costs associated with traditional synthetic routes. For global pharmaceutical manufacturers, this represents a paradigm shift towards safer, more economical, and industrially scalable production of high-purity pharmaceutical intermediates. The technology not only simplifies the synthetic sequence but also ensures consistent quality control, which is paramount for regulatory compliance in the highly scrutinized API market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Noxastota and its precursors has been plagued by inefficient and hazardous chemical processes that hinder large-scale adoption. As illustrated in prior art such as patent CN102977015A, traditional routes often necessitate extreme reaction conditions, including high-temperature grinding at 210-220°C and cryogenic reactions at -78°C. 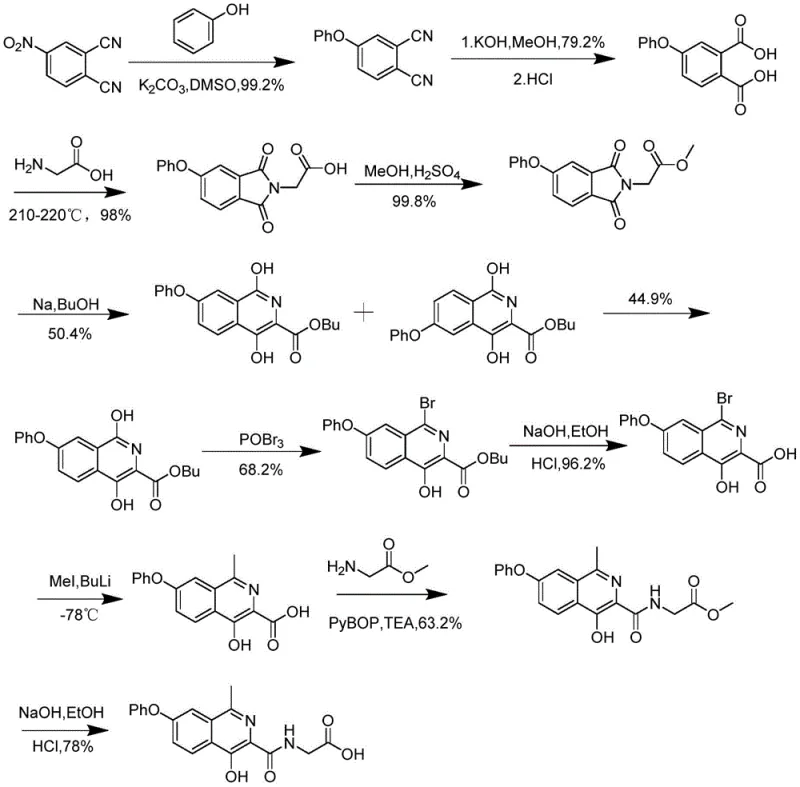 These severe parameters not only demand specialized and expensive equipment but also introduce significant safety risks due to the use of highly reactive and flammable reagents like sodium metal and butyl lithium. Furthermore, these legacy methods suffer from poor reaction selectivity, leading to difficult separation of isomers and a cumulative total yield that can plummet to as low as 2 percent. Another notable example, disclosed in CN106478503A, relies on starting materials like 3-methyl-5-bromoisobenzofuran-1(3H)-one, which commands a prohibitive market price of approximately 650 dollars per gram. Such economic barriers, combined with the generation of genotoxic impurities from hydrazino compounds, render these conventional approaches unsuitable for modern, cost-sensitive industrial manufacturing.
These severe parameters not only demand specialized and expensive equipment but also introduce significant safety risks due to the use of highly reactive and flammable reagents like sodium metal and butyl lithium. Furthermore, these legacy methods suffer from poor reaction selectivity, leading to difficult separation of isomers and a cumulative total yield that can plummet to as low as 2 percent. Another notable example, disclosed in CN106478503A, relies on starting materials like 3-methyl-5-bromoisobenzofuran-1(3H)-one, which commands a prohibitive market price of approximately 650 dollars per gram. Such economic barriers, combined with the generation of genotoxic impurities from hydrazino compounds, render these conventional approaches unsuitable for modern, cost-sensitive industrial manufacturing.
The Novel Approach
In stark contrast, the methodology presented in CN113754569A offers a streamlined and economically viable alternative that fundamentally redesigns the synthetic architecture. The core innovation lies in the preparation of intermediate Formula 5 through a coupling reaction that avoids the need for expensive halogenated starting materials. Instead, it utilizes readily accessible precursors with market prices ranging from merely 400 to 500 RMB per kilogram, representing a massive reduction in raw material expenditure. The process eliminates the requirement for dangerous alkali metals and extreme temperature fluctuations, operating instead under mild conditions that are inherently safer for plant personnel and equipment. By simplifying the reaction sequence and avoiding complex purification steps like column chromatography, this novel approach significantly enhances the overall throughput. The result is a high-yield, controllable process that is specifically engineered to meet the rigorous demands of industrial-scale production, ensuring a stable supply of critical pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonyl Insertion
The heart of this technological advancement is the sophisticated application of palladium-catalyzed carbonyl insertion to construct the isoquinoline core efficiently. The process begins with the synthesis of intermediate Formula 5, where a sulfonyl-protected glycine ester is coupled with a phenoxy-substituted phenyl derivative. This step is crucial as it installs the necessary nitrogen functionality while maintaining the integrity of the sensitive phenoxy group. Subsequently, the transformation of Formula 5 into Formula 6 involves a carbonyl insertion reaction mediated by palladium catalysts such as palladium acetate or tetrakis(triphenylphosphine)palladium. 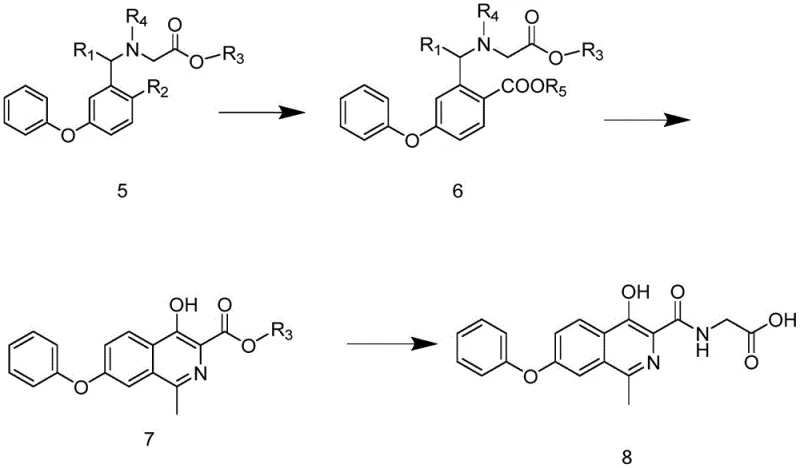 In this mechanistic step, a carbon monoxide source, which can be gas or a metal carbonyl complex, is inserted into the carbon-halogen or carbon-sulfonate bond. This reaction is facilitated by phosphine ligands like 1,3-bis(diphenylphosphino)propane, which stabilize the palladium center and enhance catalytic turnover. The presence of an alcohol solvent acts as both the reaction medium and the trapping agent for the acyl-palladium intermediate, forming the ester linkage found in Formula 6. This mechanism is highly selective, minimizing the formation of side products and ensuring that the stereochemical and structural fidelity of the molecule is preserved throughout the synthesis.
In this mechanistic step, a carbon monoxide source, which can be gas or a metal carbonyl complex, is inserted into the carbon-halogen or carbon-sulfonate bond. This reaction is facilitated by phosphine ligands like 1,3-bis(diphenylphosphino)propane, which stabilize the palladium center and enhance catalytic turnover. The presence of an alcohol solvent acts as both the reaction medium and the trapping agent for the acyl-palladium intermediate, forming the ester linkage found in Formula 6. This mechanism is highly selective, minimizing the formation of side products and ensuring that the stereochemical and structural fidelity of the molecule is preserved throughout the synthesis.
Furthermore, the subsequent cyclization and deprotection steps are designed to maximize purity and minimize waste. The conversion of Formula 6 to Formula 7 involves an alkaline condensation that promotes aromatization, effectively closing the isoquinoline ring system. This is followed by a straightforward amidation with glycine to yield the final Noxastota structure. The careful selection of bases, such as sodium methoxide or potassium tert-butoxide, ensures that the deprotection of the sulfonyl group occurs cleanly without degrading the sensitive ester moieties. This precise control over the reaction environment prevents the formation of genotoxic impurities often associated with hydrazine-based routes. By understanding these mechanistic nuances, R&D teams can optimize reaction parameters such as catalyst loading (0.5% to 10%) and ligand ratios to achieve optimal yields, thereby securing a robust supply chain for this vital therapeutic agent.
How to Synthesize Noxastota Intermediate Efficiently
The synthesis of the key intermediate Formula 5 and its subsequent conversion to Noxastota requires precise adherence to the optimized conditions outlined in the patent to ensure maximum efficiency and safety. The process integrates standard organic transformations with advanced catalytic techniques to build molecular complexity rapidly. Operators must pay close attention to the stoichiometry of the palladium catalyst and the purity of the carbon monoxide source to prevent catalyst poisoning. Detailed standardized operating procedures for each reaction step, including specific workup and purification protocols, are essential for reproducibility.
- Prepare the precursor compound Formula 4 by reacting Formula 3 with a Grignard reagent followed by hydroxyl halogenation or sulfonylation.
- Perform a coupling reaction between compound Formula 4 and a sulfonyl-protected glycine ester (Formula 9) using DEAD/PPh3 or similar auxiliaries to generate intermediate Formula 5.
- Execute a palladium-catalyzed carbonyl insertion reaction on Formula 5 using a CO source and alcohol to yield the cyclized precursor Formula 6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis route described in CN113754569A offers transformative benefits that directly impact the bottom line and operational resilience. The most immediate advantage is the drastic reduction in raw material costs, driven by the substitution of exotic, high-priced starting materials with commodity chemicals that are widely available in the global market. This shift not only lowers the direct cost of goods sold but also mitigates the risk of supply disruptions caused by reliance on single-source specialty vendors. Additionally, the elimination of hazardous reagents like sodium metal and butyl lithium simplifies the safety infrastructure required for production, reducing insurance premiums and regulatory compliance burdens. The mild reaction conditions further contribute to energy savings, as there is no longer a need for energy-intensive heating to 220°C or cryogenic cooling to -78°C. These factors collectively create a leaner, more agile manufacturing process that is better equipped to handle fluctuating market demands.
- Cost Reduction in Manufacturing: The new process achieves substantial cost savings by replacing starting materials that previously cost over 10,000 RMB/kg with alternatives priced at merely 400-500 RMB/kg. This order-of-magnitude reduction in input costs allows for significantly improved profit margins or more competitive pricing strategies for the final API. Furthermore, the avoidance of expensive transition metal removal steps and complex chromatographic purifications reduces downstream processing expenses. The high atom economy of the carbonyl insertion step ensures that a greater proportion of raw materials end up in the final product, minimizing waste disposal costs. Overall, the economic profile of this route makes the production of Noxastota intermediates financially sustainable even in a price-sensitive generic drug market.
- Enhanced Supply Chain Reliability: By utilizing easily obtainable raw materials, the new synthesis route decouples production from the volatility of the specialty chemical market. The starting materials are common industrial chemicals with multiple global suppliers, ensuring a continuous and reliable flow of inputs. This diversification of the supply base reduces the risk of production stoppages due to vendor shortages or geopolitical issues. Moreover, the simplified synthetic sequence shortens the overall lead time from raw material procurement to finished intermediate. The robustness of the reaction conditions means that production can be scaled up rapidly without the need for specialized, hard-to-source equipment, thereby enhancing the agility of the supply chain to respond to sudden increases in demand for anemia treatments.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, avoiding the pitfalls of laboratory-only techniques that fail in large reactors. The absence of genotoxic hydrazine reagents simplifies the environmental, health, and safety (EHS) profile of the manufacturing site, facilitating easier regulatory approval and reducing the burden of hazardous waste management. The mild conditions reduce the thermal load on the facility, lowering energy consumption and the carbon footprint of the manufacturing process. Additionally, the high selectivity of the reactions minimizes the generation of byproducts, leading to cleaner effluents and reduced treatment costs. This alignment with green chemistry principles not only meets current environmental regulations but also future-proofs the manufacturing operation against increasingly stringent global sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in patent CN113754569A, providing clarity for stakeholders evaluating this process for adoption. Understanding these details is crucial for making informed decisions about technology transfer and capacity planning.
Q: What are the primary cost advantages of the new Noxastota synthesis route?
A: The new route utilizes readily available raw materials costing approximately 400-500 RMB/kg, significantly lower than the 10,000+ RMB/kg starting materials required in prior art methods, drastically reducing overall production costs.
Q: Does this process eliminate the need for hazardous reagents like sodium metal?
A: Yes, unlike previous methods that required dangerous reagents such as sodium metal and butyl lithium at extreme temperatures (-78°C to 220°C), this novel approach operates under mild conditions, enhancing operational safety.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Absolutely. The process avoids complex column chromatography purification and uses stable intermediates, making it highly scalable and economically viable for commercial production of HIF-PHI inhibitors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Noxastota Intermediate Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthesis technology to deliver high-quality Noxastota intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of our pharmaceutical intermediates meets the highest international standards. Our commitment to excellence extends beyond mere compliance; we actively collaborate with our partners to optimize processes for maximum efficiency and cost-effectiveness. By choosing us as your partner, you gain access to a supply chain that is both resilient and innovative, capable of navigating the complexities of modern drug manufacturing.
We invite you to engage with our technical procurement team to discuss how this new route can benefit your specific project needs. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this superior synthesis method. Please contact us to request specific COA data and route feasibility assessments tailored to your production timeline. Let us help you secure a competitive edge in the HIF-PHI inhibitor market through our cutting-edge chemical solutions and unwavering dedication to quality.
