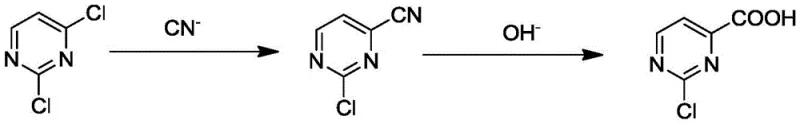
Advanced Nickel-Catalyzed Synthesis of 2-Chloropyrimidine-4-Carboxylic Acid for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic building blocks, and the recent technological advancements detailed in patent CN111393376B represent a significant leap forward for the production of 2-chloropyrimidine-4-carboxylic acid. This specific compound serves as an indispensable key intermediate in the synthesis of benzenesulfonamide pyrazole kinase inhibitors and therapeutic agents for hyperphosphatemia, commanding substantial attention from global R&D teams. The disclosed methodology shifts away from traditional, often problematic oxidation routes, opting instead for a highly selective two-step sequence that begins with 2,4-dichloropyrimidine. By leveraging a sophisticated nickel-catalyzed nucleophilic substitution followed by a controlled hydrolysis, this approach addresses long-standing challenges regarding impurity profiles and overall process efficiency. For procurement specialists and supply chain directors, understanding the nuances of this patent is crucial, as it outlines a pathway that not only enhances chemical purity but also streamlines the manufacturing workflow, thereby offering a more reliable pharmaceutical intermediate supplier option for complex drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloropyrimidine-4-carboxylic acid has relied heavily on methods such as those described in patent CN106083734A, which utilize 2-chloro-4-methylpyrimidine hydrochloride as the primary starting material. While conceptually straightforward, these conventional oxidation strategies suffer from inherent chemical limitations that severely impact commercial viability. The core issue lies in the poor selectivity of the oxidation reaction required to convert the methyl group into a carboxylic acid functionality. This lack of selectivity frequently leads to the formation of numerous difficult-to-separate by-products, which complicates downstream purification processes and drastically reduces the overall yield of the final active ingredient. Furthermore, the harsh conditions often necessary for these oxidations can degrade sensitive heterocyclic rings, leading to batch-to-batch inconsistency that is unacceptable for GMP-compliant manufacturing. These technical bottlenecks result in increased waste generation and higher operational costs, creating significant friction for cost reduction in pharma manufacturing initiatives.
The Novel Approach
In stark contrast to the oxidative degradation seen in older methodologies, the novel approach presented in CN111393376B introduces a constructive synthetic strategy that builds complexity through precise bond formation rather than destructive oxidation. By initiating the synthesis with 2,4-dichloropyrimidine, a readily available and cost-effective commodity chemical, the process bypasses the instability issues associated with methyl oxidation. The first step involves a nickel-catalyzed cyanation, which selectively replaces one chlorine atom with a cyano group under moderate thermal conditions. This is followed by a mild hydrolysis step that converts the nitrile into the desired carboxylic acid. This sequential logic ensures that the pyrimidine ring remains intact throughout the transformation, preserving the structural integrity required for subsequent coupling reactions in drug synthesis. The result is a process that is not only chemically superior but also operationally simpler, offering a clear pathway for commercial scale-up of complex pharmaceutical intermediates without the burden of extensive impurity cleanup.
Mechanistic Insights into Nickel-Catalyzed Cyanation and Hydrolysis
The heart of this innovative synthesis lies in the meticulous design of the catalytic cycle used during the nucleophilic substitution phase. The reaction employs a nickel-based metal catalyst precursor, such as nickel bromide or nickel acetate, which forms an active complex in situ with specialized bidentate phosphine ligands like Xantphos or DPPF. These ligands are critical for stabilizing the nickel center and facilitating the oxidative addition of the aryl chloride bond, a step that is traditionally difficult due to the strength of the C-Cl bond in electron-deficient heterocycles. The presence of the ligand ensures that the cyanating reagent, whether it be cuprous cyanide, zinc cyanide, or the safer acetone cyanohydrin, can effectively displace the chlorine atom at the 4-position with high regioselectivity. This mechanistic precision prevents the formation of bis-cyanated by-products or dehalogenated species, ensuring that the intermediate stream is clean and ready for the subsequent transformation without requiring chromatographic purification.
Following the successful installation of the cyano group, the second stage of the mechanism involves the hydrolysis of the nitrile to the carboxylic acid. This step is notably enhanced by the presence of iodide salts, such as sodium iodide or potassium iodide, alongside a strong base like sodium hydroxide. The iodide ion acts as a potent nucleophilic catalyst, likely facilitating the attack of the hydroxide ion on the nitrile carbon or assisting in the solubilization of intermediates, thereby accelerating the reaction rate at relatively low temperatures of 60-80°C. This mild hydrolysis condition is paramount for maintaining the stability of the 2-chloro substituent, which might otherwise be susceptible to nucleophilic aromatic substitution under more vigorous alkaline conditions. The synergy between the nickel catalyst in the first step and the iodide promoter in the second step creates a robust chemical environment that maximizes yield while minimizing side reactions, ultimately delivering a product with exceptional purity specifications.
The structural evolution of the molecule throughout this process is critical for quality control teams to monitor. The intermediate formed after the first step, 2-chloro-4-cyanopyrimidine, serves as a pivotal checkpoint in the synthesis. Its formation confirms the success of the catalytic cyanation and sets the stage for the final hydrolysis. The preservation of the chlorine atom at the 2-position during both steps is essential, as this handle is often required for further functionalization in the final drug molecule. The final product, 2-chloropyrimidine-4-carboxylic acid, retains this critical functionality while introducing the acidic moiety needed for salt formation or amide coupling. Understanding these structural nuances allows R&D directors to better assess the feasibility of integrating this intermediate into their own proprietary synthesis routes, ensuring that the material supplied meets the rigorous demands of modern medicinal chemistry.
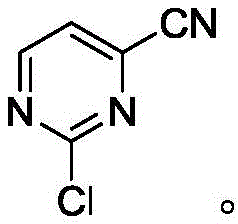
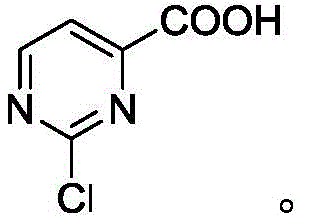
How to Synthesize 2-Chloropyrimidine-4-Carboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to fully realize the benefits outlined in the patent data. The process begins with the preparation of the reaction vessel where 2,4-dichloropyrimidine is combined with the nickel catalyst system in a polar aprotic solvent such as NMP. Precise control of the temperature, maintained between 80-120°C depending on the specific cyanating agent used, is vital to drive the substitution to completion without degrading the catalyst. Once the intermediate is isolated, typically through aqueous workup and organic extraction, it proceeds directly to the hydrolysis stage. Here, the addition of the iodide promoter is the key differentiator that allows the reaction to proceed rapidly at lower temperatures, saving energy and reducing thermal stress on the equipment. Detailed standardized synthesis steps see the guide below.
- Perform nucleophilic substitution on 2,4-dichloropyrimidine using a cyanating reagent and nickel catalyst.
- Isolate the 2-chloro-4-cyanopyrimidine intermediate through extraction and purification.
- Hydrolyze the nitrile group to carboxylic acid using alkaline conditions with iodide promotion.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthetic route offers profound advantages that extend well beyond simple chemical yield. For procurement managers tasked with optimizing budgets, the elimination of harsh oxidation steps translates directly into significant cost savings. Traditional oxidation methods often require expensive oxidants and generate substantial amounts of hazardous waste that incur high disposal fees. By replacing this with a catalytic substitution and mild hydrolysis, the process drastically simplifies the waste stream and reduces the consumption of auxiliary chemicals. Furthermore, the use of 2,4-dichloropyrimidine as a starting material leverages a supply chain that is already well-established and mature, mitigating the risk of raw material shortages that can plague more exotic starting materials. This stability in the upstream supply chain ensures consistent production schedules and reliable delivery timelines for downstream pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high atom economy of the cyanation step and the avoidance of expensive purification protocols. Since the reaction selectivity is inherently high due to the nickel-ligand system, the need for resource-intensive chromatography is eliminated, allowing for simpler crystallization or extraction methods. This reduction in processing complexity lowers the overall cost of goods sold (COGS) and improves the margin profile for the final intermediate. Additionally, the catalytic nature of the nickel system means that only small amounts of metal are required, reducing the burden on heavy metal clearance procedures later in the drug substance manufacturing, which further contributes to substantial cost savings in the overall production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that rely on single-source or unstable reagents. This method utilizes commodity chemicals like zinc cyanide or acetone cyanohydrin and common bases like sodium hydroxide, all of which are available from multiple global suppliers. This diversification of the supply base reduces the risk of disruption and allows for more flexible procurement strategies. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring that production batches remain consistent even when sourcing from different vendors. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates and maintaining just-in-time inventory levels.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability increase, the green chemistry attributes of this synthesis become a major competitive advantage. The process operates at moderate temperatures and avoids the generation of toxic oxidation by-products, aligning with stricter environmental compliance standards. The simplicity of the workup procedure, involving standard liquid-liquid extraction and pH adjustment, is easily adaptable from kilogram to multi-ton scales without requiring specialized high-pressure or high-temperature equipment. This ease of scale-up ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly, allowing manufacturers to respond quickly to market demand fluctuations without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries based on the specific details found within the patent documentation. These questions address the practical concerns of R&D and operations teams who are evaluating this route for potential adoption or outsourcing. Understanding these specifics helps in making informed decisions regarding process validation and vendor qualification. The following insights are derived directly from the experimental data and claims presented in the intellectual property.
Q: What are the advantages of this synthesis over traditional oxidation methods?
A: This method avoids harsh high-temperature oxidation, resulting in fewer by-products, higher purity (up to 99.0%), and significantly improved yield compared to methyl oxidation routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials like 2,4-dichloropyrimidine and operates under moderate temperatures (80-120°C), making it highly scalable and cost-effective for commercial manufacturing.
Q: What catalysts are used in the cyanation step?
A: The reaction employs a nickel-based catalyst system, typically using precursors like nickel bromide or acetate combined with bidentate phosphine ligands such as Xantphos or DPPF to ensure high selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloropyrimidine-4-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN111393376B can be replicated reliably on an industrial scale. Our facilities are equipped with stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. We understand that for a critical intermediate like 2-chloropyrimidine-4-carboxylic acid, consistency is key, and our quality management systems are designed to deliver that consistency batch after batch, supporting your long-term drug development goals.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this nickel-catalyzed route can optimize your specific budget requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique production needs. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior, ensuring that your pipeline moves forward without interruption.
