Scalable Synthesis of Functionalized Azetidine Esters for Advanced Pharmaceutical Intermediate Production
Scalable Synthesis of Functionalized Azetidine Esters for Advanced Pharmaceutical Intermediate Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A recent technological breakthrough, documented in patent CN113045539B, introduces a novel preparation method for 2-(4,4-dichloro-1-(8-quinolyl)-2-azetidinyl) carboxylic acid ester derivatives. This innovation addresses critical bottlenecks in the synthesis of azetidine scaffolds, which are increasingly recognized as vital structural motifs in modern drug design due to their conformational rigidity and metabolic stability. By leveraging a unique base-mediated rearrangement of beta-lactam precursors, this technology offers a streamlined pathway that bypasses the severe conditions and hazardous reagents associated with legacy methods. For R&D directors and procurement strategists alike, understanding the implications of this patent is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of azetidine rings has been a formidable challenge in organic synthesis, often necessitating multi-step sequences that suffer from poor atom economy and low overall yields. Traditional approaches frequently rely on direct nucleophilic substitution to close the ring or the reduction of beta-lactams using powerful, moisture-sensitive hydride sources such as lithium aluminum hydride (LiAlH4). These conventional methodologies present significant drawbacks for industrial application; LiAlH4, for instance, poses severe safety risks due to its pyrophoric nature and requires rigorous anhydrous conditions, driving up infrastructure and handling costs. Furthermore, alternative routes utilizing Lewis acids like aluminum trichloride often generate substantial amounts of corrosive waste and require complex purification protocols to remove residual metal contaminants, which is a critical failure point for pharmaceutical grade intermediates. The reliance on chiral starting materials like 1,3-butanediol in some prior art further restricts the economic viability and substrate scope, limiting the ability to rapidly generate diverse analog libraries needed for structure-activity relationship (SAR) studies.
The Novel Approach
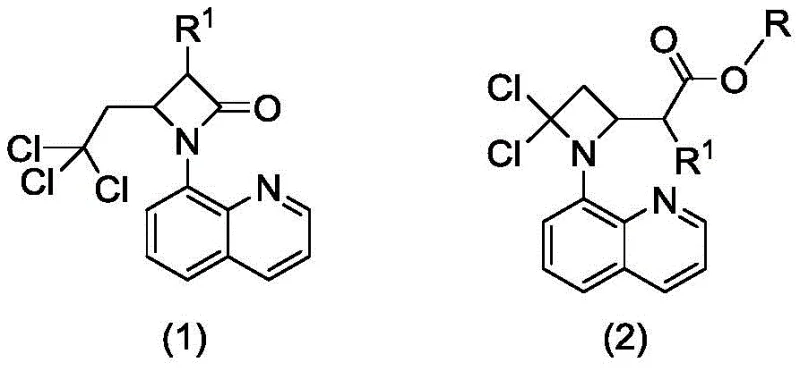
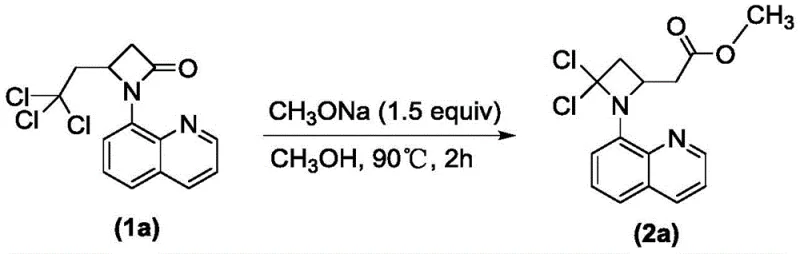
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN113045539B utilizes a remarkably simple yet effective intramolecular nucleophilic substitution strategy. The core of this innovation lies in the transformation of readily available substituted 4-(2,2,2-trichloroethyl)-1-(8-quinolyl)-beta-lactam derivatives into the target azetidine esters using sodium alkoxide in a monohydric alcohol solvent. This single-pot transformation proceeds under mild thermal conditions, typically between 80°C and 110°C, eliminating the need for cryogenic temperatures or ultra-dry environments. The reaction effectively repurposes the trichloroethyl side chain and the lactam carbonyl functionality to construct the dichloro-azetidine core with an appended ester group, achieving a level of molecular complexity in a single step that previously required multiple operations. This paradigm shift not only drastically reduces the number of unit operations but also utilizes commodity chemicals like sodium methoxide and methanol, making the process inherently more sustainable and economically attractive for large-scale production.
Mechanistic Insights into Base-Mediated Beta-Lactam Rearrangement
The chemical elegance of this process stems from a sophisticated base-mediated rearrangement mechanism that leverages the electron-withdrawing nature of the trichloromethyl group. Upon exposure to the alkoxide base, the beta-lactam ring undergoes a specific activation where the nucleophile likely attacks the carbonyl carbon or facilitates an elimination-addition sequence involving the trichloroethyl moiety. The presence of the 8-quinolyl group on the nitrogen atom plays a pivotal role, potentially stabilizing transition states through electronic effects or acting as a temporary directing group that favors the formation of the four-membered azetidine ring over other potential oligomerization pathways. The loss of a chloride ion from the trichloroethyl group, coupled with the incorporation of the alkoxy group from the solvent, results in the formation of the ester functionality at the C2 position while simultaneously establishing the gem-dichloro motif at the C4 position. This intricate dance of bond breaking and forming occurs with high regioselectivity, ensuring that the desired 2-substituted azetidine is formed predominantly, thereby minimizing the formation of difficult-to-separate regioisomers that often plague azetidine synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over reduction-based routes. Since the reaction does not involve over-reduction or the use of heavy metal catalysts, the impurity profile is significantly cleaner, primarily consisting of unreacted starting material or simple hydrolysis byproducts that are easily removed. The absence of metal residues is particularly beneficial for downstream processing, as it removes the necessity for expensive scavenging resins or complex aqueous workups designed to meet strict ICH Q3D guidelines for elemental impurities. Furthermore, the robustness of the reaction conditions allows for a wide tolerance of functional groups on the R1 substituent, whether it be alkyl, benzyl, or substituted benzyl moieties, without compromising the integrity of the sensitive azetidine ring. This mechanistic resilience ensures consistent product quality across different batches, a critical factor for maintaining supply chain reliability in the production of high-value pharmaceutical intermediates.
How to Synthesize 2-(4,4-dichloro-1-(8-quinolyl)-2-azetidinyl) Carboxylate Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward, requiring standard glassware and heating equipment without the need for specialized high-pressure reactors. The process begins by dissolving the beta-lactam precursor and the sodium alkoxide reagent in the chosen alcohol solvent, creating a homogeneous reaction mixture that is then heated to the optimal temperature range. Monitoring the reaction progress is easily achieved via thin-layer chromatography (TLC), allowing operators to quench the reaction precisely upon completion to maximize yield and minimize degradation. The workup procedure is equally benign, typically involving concentration of the reaction mixture followed by purification via silica gel column chromatography using a standard petroleum ether and ethyl acetate gradient. This simplicity makes the technology highly transferable from gram-scale discovery to kilogram-scale production, facilitating rapid access to these valuable building blocks for drug development programs.
- Dissolve the substituted 4-(2,2,2-trichloroethyl)-1-(8-quinolyl)-beta-lactam derivative and sodium alkoxide (e.g., sodium methoxide) in a monohydric alcohol solvent such as methanol.
- Heat the reaction mixture to a temperature between 80°C and 110°C, preferably 90°C, and maintain stirring for 1 to 10 hours until TLC indicates completion.
- Purify the crude reaction product using silica gel column chromatography with a petroleum ether and ethyl acetate eluent system to isolate the high-purity azetidine ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond the laboratory bench. The shift from hazardous, expensive reagents to commodity chemicals fundamentally alters the cost structure of producing these complex heterocycles. By eliminating the dependency on lithium aluminum hydride and exotic chiral auxiliaries, manufacturers can secure raw materials from a broader, more competitive supplier base, thereby mitigating supply risk and stabilizing input costs. The simplified operational profile also means that production can be scheduled with greater flexibility, as the reaction does not require the extensive safety protocols and dedicated equipment lines associated with pyrophoric reagents. This operational agility allows for faster turnaround times on custom synthesis orders, enabling pharmaceutical partners to accelerate their own development timelines and bring life-saving therapies to market more rapidly.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the replacement of high-cost, high-hazard reagents with inexpensive, bulk-available sodium alkoxides and alcohols. The elimination of expensive metal catalysts and the associated downstream purification steps—such as activated carbon treatment or specialized resin columns for metal scavenging—results in a drastic reduction in both material and processing costs. Furthermore, the high isolated yields reported in the patent examples, consistently hovering around 70% to 72%, ensure that raw material utilization is maximized, minimizing waste disposal fees and improving the overall mass balance of the manufacturing campaign. This efficiency gain directly contributes to a lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of stable, non-hazardous reagents that are not subject to the same stringent transportation and storage regulations as pyrophoric hydrides. Sodium methoxide and methanol are widely produced commodities with robust global supply networks, ensuring that production schedules are not disrupted by raw material shortages or logistical bottlenecks. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and ancillary equipment, leading to lower maintenance costs and higher asset availability. This reliability is crucial for long-term supply agreements, giving pharmaceutical clients the confidence that their critical intermediate needs will be met consistently without unexpected delays or quality deviations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant step forward in green chemistry. The avoidance of heavy metals and the use of alcohol solvents, which are easier to recover and recycle compared to chlorinated solvents often used in older methods, align with increasingly strict global environmental regulations. The simplified waste stream, devoid of toxic metal sludge, reduces the burden on wastewater treatment facilities and lowers the cost of hazardous waste disposal. This environmental compatibility not only future-proofs the manufacturing process against tightening regulatory frameworks but also enhances the corporate sustainability profile of the supply chain, a key metric for many multinational pharmaceutical corporations today.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel azetidine synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a transparent view of the process capabilities. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios or sourcing strategies.
Q: What are the primary advantages of this new azetidine synthesis method over traditional reduction techniques?
A: Unlike traditional methods requiring hazardous reducing agents like LiAlH4 or harsh Lewis acids, this patented process utilizes mild sodium alkoxide bases in alcohol solvents. This eliminates the need for expensive metal removal steps, significantly improves operational safety, and simplifies the post-reaction workup, leading to higher overall process efficiency.
Q: Can this synthesis route accommodate diverse substituent groups on the azetidine ring?
A: Yes, the method demonstrates excellent substrate scope. The patent data confirms successful synthesis with various R1 groups including hydrogen, methyl, ethyl, benzyl, and substituted benzyl moieties. This versatility allows for the generation of a wide library of intermediates suitable for different drug discovery programs without altering the core reaction conditions.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Absolutely. The reaction operates at moderate temperatures (80-110°C) using common, inexpensive solvents like methanol and reagents like sodium methoxide. The isolation involves standard silica gel chromatography, which is easily scalable. The reported yields of approximately 70-72% combined with the simplicity of the one-pot transformation make it highly viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4,4-dichloro-1-(8-quinolyl)-2-azetidinyl) Carboxylic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the next generation of pharmaceutical intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to seamlessly translate this laboratory-scale innovation into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is smooth and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of 2-(4,4-dichloro-1-(8-quinolyl)-2-azetidinyl) carboxylic acid ester derivative adheres to the highest industry standards for identity, potency, and impurity profiles.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development needs. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this efficient synthesis platform can drive value and accelerate your path to clinical success.
