Scalable Synthesis of Chiral S-4-Hydroxypyrrolidone Derivatives via L-Malic Acid Route
Scalable Synthesis of Chiral S-4-Hydroxypyrrolidone Derivatives via L-Malic Acid Route
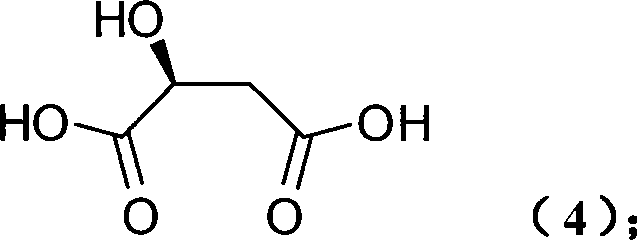
The pharmaceutical industry continuously seeks robust and cost-effective pathways for producing chiral intermediates, particularly for nootropic agents like Oxiracetam. Patent CN101239938A introduces a groundbreaking methodology for the preparation of (S)-4-hydroxypyrrolidone and its derivatives, utilizing naturally occurring L-Malic Acid as the foundational chiral source. This approach represents a significant paradigm shift from traditional resolution-based methods, offering a streamlined synthetic route that preserves stereochemical integrity while drastically reducing process complexity. By leveraging the inherent chirality of L-Malic Acid, manufacturers can bypass expensive enzymatic resolutions or asymmetric catalysis steps, thereby enhancing the economic viability of producing high-value pharmaceutical intermediates. The technical implications of this patent extend beyond mere cost savings, providing a reliable framework for consistent quality and supply chain stability in the competitive landscape of fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art technologies, such as those described in WO9306826A1 and academic literature like Tetrahedron: Asymmetry (1992), have historically relied on convoluted synthetic sequences to access (S)-configured pyrrolidones. These conventional routes often necessitate the synthesis of racemic mixtures followed by tedious chiral resolution processes, which inherently cap the maximum theoretical yield at 50% unless dynamic kinetic resolution is employed. Furthermore, existing methods frequently involve harsh dehydroxylation steps and complex protection-deprotection strategies that introduce significant impurities and reduce overall process efficiency. The cumulative effect of these inefficiencies is a high cost of goods sold (COGS) and a fragile supply chain susceptible to yield fluctuations. For procurement managers and supply chain heads, these legacy processes represent a substantial risk, as the reliance on low-yield steps and specialized chiral reagents can lead to unpredictable lead times and inflated raw material costs.
The Novel Approach
In stark contrast, the methodology disclosed in CN101239938A capitalizes on the 'chiral pool' concept, initiating the synthesis with L-Malic Acid, a ubiquitous and inexpensive natural product. This novel route simplifies the transformation into a logical sequence of protection, cyclization, reduction, and deprotection, effectively minimizing the number of unit operations required. The strategic use of acyl chloride for hydroxyl protection followed by thermal cyclization with amines allows for the rapid construction of the pyrrolidone core with high stereochemical fidelity. By eliminating the need for resolution, the process theoretically doubles the material throughput compared to racemic routes, directly translating to substantial cost reduction in pharmaceutical intermediate manufacturing. This streamlined approach not only enhances operational simplicity but also aligns perfectly with green chemistry principles by reducing waste generation and solvent consumption, making it an ideal candidate for sustainable industrial production.
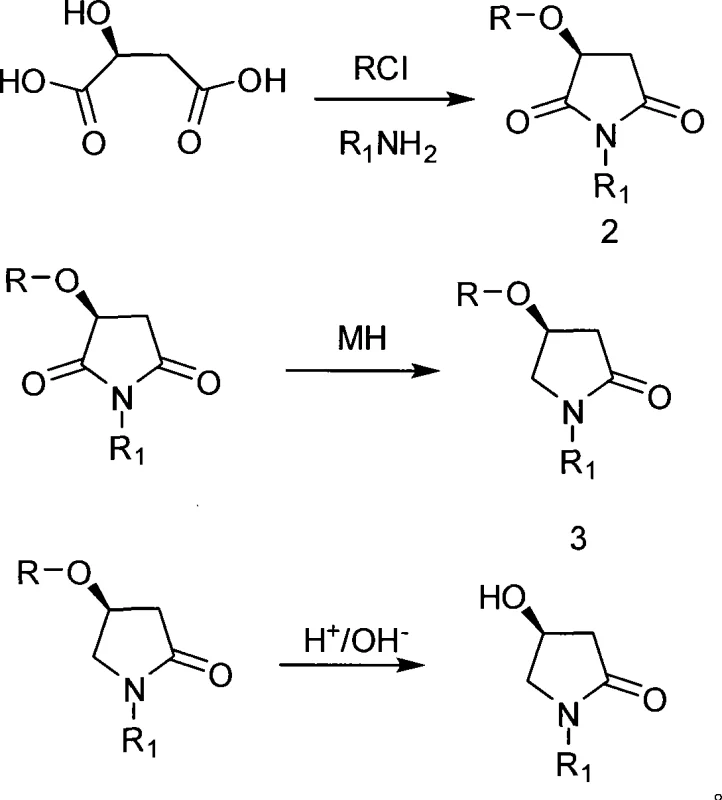
Mechanistic Insights into Chiral Pool Cyclization and Reduction
The core mechanistic advantage of this synthesis lies in the preservation of the chiral center originating from the L-Malic Acid backbone. As illustrated by the structure of the starting material, the stereocenter is already established in the natural feedstock, removing the kinetic and thermodynamic challenges associated with creating chirality de novo. The initial protection step converts the hydroxyl group into a stable ester, preventing unwanted side reactions during the subsequent high-temperature cyclization phase. During the cyclization step, the activated dicarboxylic acid derivative reacts with the amine nucleophile to form a cyclic imide intermediate. This thermal ring-closing reaction is driven by entropy and the stability of the five-membered ring, proceeding efficiently at temperatures between 50°C and 200°C. The robustness of this step ensures that the stereochemical configuration remains intact, setting the stage for the critical reduction phase.
The subsequent reduction of the cyclic imide to the lactam is a pivotal transformation that defines the success of the route. Utilizing metal borohydrides, such as sodium borohydride or potassium borohydride, in the presence of Lewis acids or acyl chlorides allows for the selective reduction of one carbonyl group over the other. This chemoselectivity is crucial for obtaining the desired 4-hydroxypyrrolidone structure rather than the fully reduced pyrrolidine. The reaction is typically conducted at mild temperatures ranging from -20°C to 20°C, which helps to control exotherms and minimize the formation of over-reduced byproducts. The presence of additives like LiCl or AlCl3 further modulates the reactivity of the borohydride species, ensuring high conversion rates and purity. Finally, the deprotection step under acidic or basic conditions cleanly removes the protecting group to reveal the free hydroxyl functionality, yielding the target (S)-4-hydroxypyrrolidone with high optical purity and minimal impurity burden.
How to Synthesize (S)-4-Hydroxypyrrolidone Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly during the cyclization and reduction phases, to maximize yield and purity. The process begins with the activation of L-Malic Acid, followed by a controlled thermal reaction with the chosen amine component to form the key imide intermediate. Subsequent reduction must be managed with precise temperature control to ensure selectivity, followed by a straightforward workup to isolate the final product. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and purification techniques, are outlined in the guide below to assist R&D teams in replicating this efficient pathway.
- Protect the hydroxyl group of L-Malic Acid using acyl chloride to form the protected intermediate.
- Perform thermal cyclization with an amine at 50-200°C to generate the imide intermediate structure.
- Execute selective reduction using metal borohydride at -20 to 20°C to convert the imide to the lactam.
- Finalize the synthesis by deprotecting the hydroxyl group using mineral acid or alkali to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-Malic Acid-based route offers transformative benefits that extend well beyond the laboratory bench. The primary driver of value is the substitution of expensive, specialized chiral starting materials with L-Malic Acid, a commodity chemical produced on a massive global scale with stable pricing and abundant availability. This shift fundamentally alters the cost structure of the intermediate, removing the premium associated with synthetic chiral building blocks. Furthermore, the reduction in synthetic steps directly correlates to lower operational expenditures, as fewer reactors, less solvent, and reduced labor hours are required to produce the same amount of final product. These efficiencies compound to create a significantly more competitive cost position for the final API or intermediate.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps effectively doubles the theoretical yield from the starting material, as there is no need to discard the unwanted enantiomer. Additionally, the use of standard reagents like sodium borohydride and acyl chlorides avoids the procurement complexities and high costs associated with exotic catalysts or enzymes. This streamlined reagent profile simplifies inventory management and reduces the working capital tied up in raw materials, leading to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Relying on L-Malic Acid as the chiral source mitigates the risk of supply disruptions often associated with niche fine chemicals. Since L-Malic Acid is derived from fermentation processes with multiple global suppliers, the supply chain is robust and resilient against geopolitical or logistical shocks. The simplified process flow also reduces the number of critical control points where production delays could occur, ensuring more predictable lead times for downstream customers. This reliability is crucial for maintaining continuous API production schedules and meeting strict delivery commitments to major pharmaceutical clients.
- Scalability and Environmental Compliance: The process conditions, ranging from ambient to moderate temperatures and pressures, are inherently safer and easier to scale than cryogenic or high-pressure alternatives. The use of common solvents like alcohols and ethers facilitates solvent recovery and recycling, aligning with modern environmental regulations and sustainability goals. The reduction in waste generation, particularly the avoidance of resolution mother liquors, lowers the burden on waste treatment facilities and reduces the overall environmental footprint of the manufacturing site, making it a preferred choice for eco-conscious production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic advantages for potential partners. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing portfolio.
Q: What is the primary advantage of using L-Malic Acid for this synthesis?
A: Using natural L-Malic Acid as a starting material leverages the chiral pool strategy, eliminating the need for expensive chiral resolution steps and ensuring high optical purity from the outset, which significantly reduces overall production costs.
Q: How does this method improve upon traditional Oxiracetam intermediate synthesis?
A: Traditional methods often involve complex multi-step sequences with low overall yields and harsh dehydroxylation conditions. This patented route simplifies the process to fewer steps with milder conditions, resulting in higher yields and better suitability for industrial scale-up.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available commodity chemicals like L-Malic Acid and standard reagents such as sodium borohydride. The operational simplicity and robust yield profile make it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-Hydroxypyrrolidone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the development of next-generation nootropics and pharmaceutical agents. Our technical team has extensively analyzed the pathway disclosed in CN101239938A and possesses the expertise to optimize this route for commercial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of (S)-4-hydroxypyrrolidone meets the highest international standards for chiral purity and chemical identity.
We invite you to collaborate with us to leverage this cost-effective technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and reliability for your organization.
