Advanced Synthesis of Ramelteon Intermediates via Novel Acrylic Ester Cyclization
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing key intermediates, particularly for high-value therapeutic agents like Ramelteon. Patent CN102408337B introduces a groundbreaking methodology for preparing 3-(2,3-dihydrobenzofuran-5-yl)-propionic acid, a critical building block in the synthesis of selective melatonin receptor agonists. This innovation pivots away from traditional, costly aldehyde-based routes, instead leveraging a versatile class of (E)-3-[4-(alkoxy)phenyl]acrylic acid ester compounds. These novel intermediates serve as precursors that enable a more streamlined, high-yielding synthesis through a sequence of alkylation, cyclization, and reduction steps. By redefining the synthetic entry point, this technology addresses long-standing challenges regarding raw material availability and process scalability.
![General chemical structure of the novel (E)-3-[4-(alkoxy)phenyl]acrylic acid ester compounds (I) used as key intermediates](/insights/img/ramelteon-intermediate-synthesis-acrylic-ester-supplier-20260305015022-01.webp)
The structural versatility of these acrylic ester compounds allows for significant flexibility in process design, accommodating various alkyl and aryl groups that can be tuned for optimal solubility and reactivity. This adaptability is crucial for process chemists aiming to optimize crystallization and purification steps during scale-up. Furthermore, the patent details specific embodiments where different ester groups, such as methyl, isopropyl, and benzyl, are successfully employed, demonstrating the broad scope of this chemical platform. The ability to manipulate the R1 and R2 groups provides a strategic advantage in tailoring the physical properties of the intermediate to suit specific manufacturing constraints without compromising the integrity of the final pharmacophore.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-(2,3-dihydrobenzofuran-5-yl)-propionic acid has relied heavily on 2,3-dihydrobenzofuran-5-carbaldehyde as the starting material. As illustrated in the prior art, this approach typically involves a Knoevenagel condensation with malonic acid or a Wittig-Horner reaction with phosphonate esters to extend the carbon chain. These conventional methods suffer from significant drawbacks, primarily stemming from the high cost and limited commercial availability of the starting aldehyde. Additionally, the Wittig-Horner route generates substantial amounts of phosphorus-containing waste, creating severe environmental burdens and complicating waste stream management in large-scale facilities.
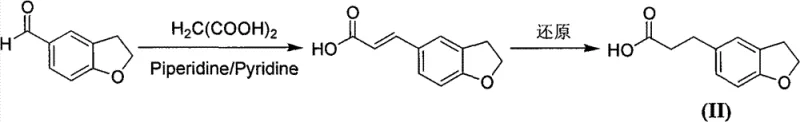
Beyond the economic and environmental penalties, the operational complexity of these traditional routes cannot be overstated. The condensation reactions often require stringent anhydrous conditions and strong bases, which demand specialized equipment and rigorous safety protocols. Subsequent reduction steps using palladium or nickel catalysts must be carefully controlled to avoid over-reduction or side reactions, often resulting in modest overall yields. The cumulative effect of these inefficiencies is a high production cost and a fragile supply chain that is vulnerable to fluctuations in the availability of specialized heterocyclic aldehydes. For procurement managers, this translates to higher input costs and increased risk of supply disruption for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN102408337B initiates the synthesis from readily available (E)-3-(4-hydroxyphenyl)acrylic acid esters. This strategic shift bypasses the need for expensive heterocyclic starting materials entirely. The new route begins with a straightforward alkylation of the phenolic hydroxyl group using 2-haloacetaldehyde acetals, followed by an elegant intramolecular Friedel-Crafts cyclization to construct the dihydrobenzofuran ring. This sequence is depicted clearly in the patent's synthetic scheme, highlighting the logical progression from simple commodity chemicals to the complex target scaffold.
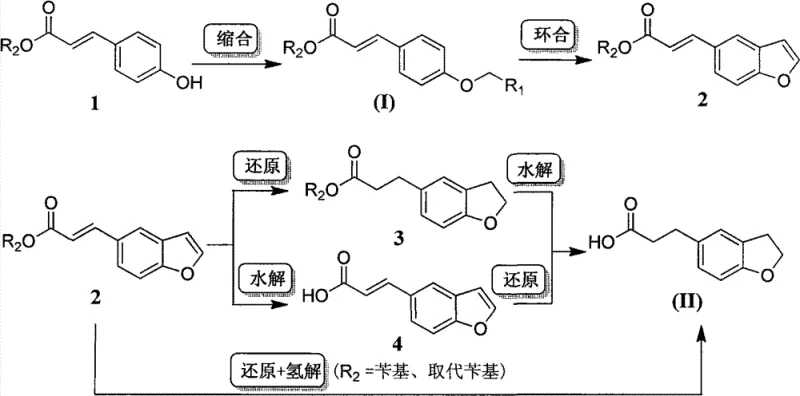
The advantages of this novel approach are manifold, offering a cleaner and more direct path to the target molecule. The cyclization step, catalyzed by common protonic or Lewis acids, proceeds under relatively mild conditions, eliminating the need for cryogenic temperatures or exotic reagents. Following ring closure, the acrylic double bond is easily reduced via catalytic hydrogenation, a unit operation that is well-understood and easily scaled in industrial settings. This route not only improves the overall atom economy but also simplifies the purification workflow, as the intermediates formed are generally more stable and easier to isolate than those in the aldehyde-based pathways. For R&D teams, this represents a significant opportunity to enhance process robustness and reduce the timeline for technology transfer.
Mechanistic Insights into Friedel-Crafts Cyclization and Reduction
The core of this innovative synthesis lies in the acid-catalyzed intramolecular Friedel-Crafts alkylation, which effectively closes the furan ring onto the aromatic backbone. In this mechanism, the acetal moiety introduced in the first step serves as a masked aldehyde equivalent. Under the influence of catalysts such as polyphosphoric acid, boron trifluoride etherate, or aluminum chloride, the acetal is activated to form an electrophilic species. This electrophile is then attacked by the electron-rich aromatic ring at the ortho position relative to the acrylic ester side chain. The regioselectivity of this attack is driven by the electronic directing effects of the substituents, ensuring that the five-membered oxygen-containing ring forms exclusively at the desired position.
Following the successful construction of the coumaran (dihydrobenzofuran) skeleton, the process requires the saturation of the exocyclic double bond. The patent describes multiple reduction strategies, including catalytic hydrogenation using palladium on carbon (Pd/C), palladium hydroxide, or Raney nickel. The choice of catalyst and solvent system—ranging from alcohols to aromatic hydrocarbons—allows for fine-tuning the reaction kinetics to prevent unwanted side reactions, such as the hydrogenolysis of benzyl ester protecting groups if they are present. In cases where benzyl esters are used, a single hydrogenation step can simultaneously reduce the alkene and cleave the benzyl group, streamlining the synthesis further. This mechanistic flexibility ensures that the process can be adapted to various substrate derivatives without losing efficiency.
How to Synthesize 3-(2,3-Dihydrobenzofuran-5-yl)-propionic Acid Efficiently
The synthesis of this high-value pharmaceutical intermediate is achieved through a logical sequence of organic transformations that prioritize yield and purity. The process begins with the alkylation of a commercially available hydroxy-cinnamate ester, followed by a cyclization step that builds the core heterocyclic structure. Detailed operational parameters, including specific solvent choices like acetone or acetonitrile for alkylation and dichloroethane or nitromethane for cyclization, are critical for success. The subsequent reduction and hydrolysis steps are designed to be telescoped where possible, minimizing isolation losses. For a comprehensive understanding of the specific molar ratios, temperature profiles, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Alkylation of (E)-3-(4-hydroxyphenyl)acrylic ester with 2-haloacetaldehyde acetals under basic conditions to form the alkoxy intermediate.
- Intramolecular Friedel-Crafts cyclization using protonic or Lewis acids to construct the dihydrobenzofuran ring system.
- Catalytic hydrogenation to reduce the acrylic double bond and subsequent hydrolysis to yield the final propionic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for supply chain stability and cost management. The primary driver of value is the substitution of expensive, specialty heterocyclic aldehydes with commodity phenolic acrylic esters. These starting materials are produced on a massive scale for other industries, ensuring a consistent and reliable supply base that is less susceptible to the bottlenecks often associated with custom fine chemical synthesis. This shift fundamentally alters the cost structure of the intermediate, moving it from a low-volume, high-margin category to a more commoditized tier, which translates directly into lower procurement costs for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of phosphorus-based reagents, such as those used in Wittig-Horner reactions, removes a significant cost center associated with both reagent purchase and hazardous waste disposal. Furthermore, the use of robust, heterogeneous catalysts like Pd/C allows for catalyst recovery and reuse, further driving down operating expenses. The simplified purification requirements, resulting from fewer side products and cleaner reaction profiles, reduce the consumption of solvents and chromatography media, leading to substantial savings in utility and material costs throughout the production lifecycle.
- Enhanced Supply Chain Reliability: By relying on raw materials that are widely available from multiple global suppliers, the risk of single-source dependency is drastically mitigated. The synthetic route avoids the use of unstable or highly regulated precursors, simplifying logistics and storage requirements. This resilience is critical for maintaining continuous production schedules, especially in the face of global supply chain disruptions. The ability to source key inputs from the bulk chemical market rather than the fine chemical niche ensures that lead times remain short and predictable, facilitating better inventory planning and Just-In-Time manufacturing strategies.
- Scalability and Environmental Compliance: The process conditions are inherently safer and more environmentally friendly, operating at moderate temperatures and pressures without the need for cryogenic cooling or pyrophoric reagents. This makes the technology highly amenable to scale-up from pilot plant to multi-ton commercial production without requiring extensive re-engineering of existing infrastructure. Additionally, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for manufacturing sites. This green chemistry profile enhances the corporate sustainability metrics of any organization adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and process descriptions found within the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary cost drivers eliminated by this new synthetic route?
A: The novel route eliminates the need for expensive 2,3-dihydrobenzofuran-5-carbaldehyde and avoids phosphorus-heavy Wittig reagents, significantly reducing raw material and waste treatment costs.
Q: How does the Friedel-Crafts cyclization step impact impurity profiles?
A: The acid-catalyzed intramolecular cyclization is highly regioselective, minimizing isomeric byproducts often seen in intermolecular coupling reactions, thus simplifying downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes robust catalysts like Pd/C or Raney-Ni and operates under mild pressure and temperature conditions, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2,3-Dihydrobenzofuran-5-yl)-propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of drug development and commercialization. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the advanced cyclization and reduction techniques described in CN102408337B positions us as a strategic partner capable of meeting the demanding quality standards of the global pharmaceutical market.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal benchmarks. Let us collaborate to drive efficiency and reliability in your supply chain for Ramelteon and related therapeutic programs.
