Breakthrough 4-Step Synthesis for 3-Azabicyclo[4.1.0]heptane-2-carboxylic Acid Commercialization
The pharmaceutical industry continuously seeks efficient pathways for complex heterocyclic intermediates, and patent CN110092753B represents a pivotal advancement in the synthesis of 3-azabicyclo[4.1.0]heptane-2-carboxylic acid and its hydrochloride salt. This specific chemical entity serves as a crucial homologue of pipecolic acid, finding extensive applications in peptide chemistry and the development of novel therapeutic agents with broad market prospects. Historically, the lack of literature reports on the direct synthesis of this compound created a significant bottleneck for research and development teams aiming to incorporate this unique bicyclic scaffold into their drug candidates. The disclosed invention overcomes these historical barriers by introducing a concise, four-step synthetic route that begins with the commercially viable starting material, tert-butyl 2-oxo-5,6-dihydropyridine-1(2H)-carboxylate. By leveraging a strategic [1+2] cycloaddition followed by reduction, cyanation, and hydrolysis, this methodology not only simplifies the operational complexity but also drastically enhances the overall economic feasibility of producing high-purity pharmaceutical intermediates. For global procurement strategists and R&D directors, this patent signals a shift towards more sustainable and cost-efficient manufacturing paradigms that align with modern green chemistry principles while ensuring a reliable supply chain for critical API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of analogous azabicyclo[4.1.0]heptane structures was plagued by inefficient multi-step sequences that rendered industrial production economically unviable. For instance, earlier methodologies documented in patent literature often required upwards of nine distinct reaction steps to achieve the target molecular architecture, resulting in a cumulative yield that was abysmally low, often hovering around merely 0.69%. Such poor efficiency translates directly into exorbitant production costs and massive waste generation, creating unacceptable burdens for supply chain managers tasked with maintaining environmental compliance and budgetary controls. Furthermore, alternative routes reported in patents such as WO2014141132 relied heavily on hazardous reagents like benzyl bromide, which poses significant safety risks during handling and requires specialized waste treatment protocols to mitigate environmental toxicity. The reliance on difficult-to-obtain starting materials in these legacy processes further exacerbated supply chain vulnerabilities, leading to extended lead times and inconsistent quality batches that could jeopardize downstream drug development timelines. Consequently, the industry urgently required a robust alternative that could bypass these structural inefficiencies and provide a scalable solution for high-purity pharmaceutical intermediates.
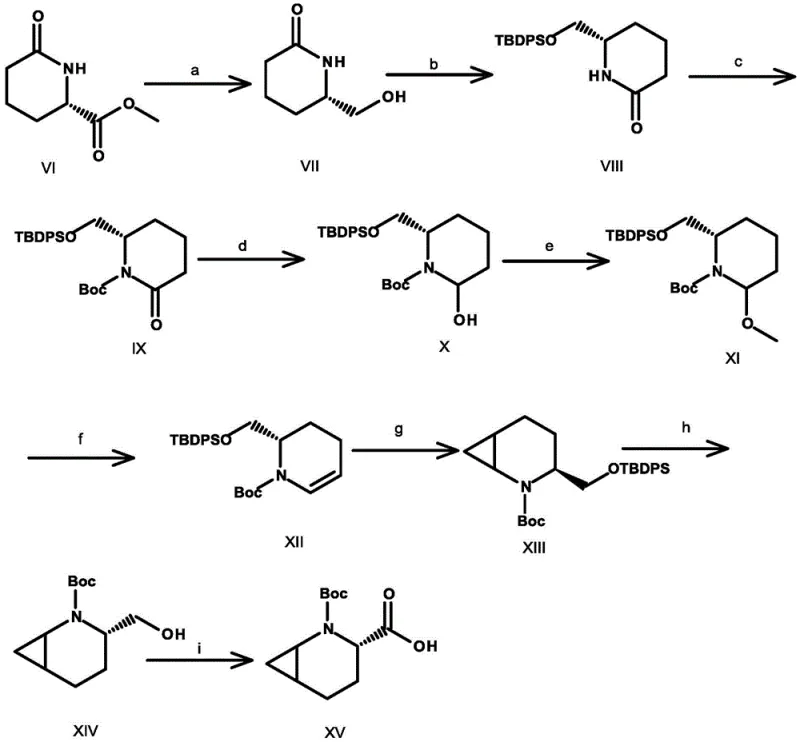
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach detailed in CN110092753B streamlines the entire synthetic sequence into just four highly optimized steps, achieving a remarkable total yield of up to 35.4%. This dramatic improvement in efficiency is achieved through the strategic application of a [1+2] cycloaddition reaction using a sulfur ylide reagent, which rapidly constructs the core bicyclic framework with high precision. The subsequent steps involve a controlled reduction and a Lewis acid-catalyzed cyanation, both of which are performed under mild conditions that minimize side reactions and impurity formation. By eliminating the need for hazardous alkylating agents and reducing the number of purification stages, this new route significantly lowers the operational expenditure and environmental footprint associated with manufacturing. For procurement managers, this translates into a more stable cost structure and reduced risk of supply disruptions, while R&D teams benefit from a cleaner impurity profile that simplifies regulatory filing. The ability to utilize cheap and easily available raw materials further underscores the commercial viability of this method, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
![Novel 4-step synthesis route for 3-azabicyclo[4.1.0]heptane-2-carboxylic acid](/insights/img/3-azabicyclo-heptane-acid-synthesis-supplier-20260304171330-03.webp)
Mechanistic Insights into Sulfur Ylide Cycloaddition and Cyanation
The cornerstone of this synthetic breakthrough lies in the initial [1+2] cycloaddition reaction between the starting enone and the sulfur ylide reagent, which effectively installs the cyclopropane ring essential for the bicyclic structure. This transformation proceeds through a concerted mechanism where the nucleophilic carbon of the ylide attacks the electron-deficient double bond of the pyridinone derivative, followed by ring closure to form the strained three-membered ring system. The choice of base, such as potassium tert-butoxide or sodium hydride, is critical in generating the active ylide species in situ, ensuring high conversion rates while minimizing decomposition of the sensitive intermediates. Following this, the reduction of the ketone moiety using triethylborohydride at low temperatures ranging from -78°C to -30°C ensures stereochemical control and prevents over-reduction or ring-opening side reactions. This precise temperature control is vital for maintaining the integrity of the bicyclic scaffold, which is prone to strain-induced degradation under harsher conditions. Finally, the introduction of the nitrile group via reaction with trimethylsilyl cyanide in the presence of a Lewis acid like tin tetrachloride activates the hydroxyl group for nucleophilic substitution, setting the stage for the final hydrolysis to the carboxylic acid.
Impurity control is inherently built into this mechanistic design, as the stepwise nature of the synthesis allows for intermediate purification that removes byproducts before they can propagate through the sequence. The use of specific Lewis acids and controlled stoichiometry during the cyanation step prevents the formation of oligomeric side products that are common in less optimized nitrile syntheses. Moreover, the final hydrolysis step, whether conducted under acidic or basic conditions, is designed to simultaneously remove the protecting group and convert the nitrile to the carboxylic acid, thereby reducing the total number of unit operations required. This convergence of deprotection and functional group transformation not only saves time but also reduces the solvent usage and waste generation per kilogram of product. For quality assurance teams, this means a more consistent impurity spectrum that is easier to characterize and control, ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications. The robustness of these reaction conditions suggests that the process is highly transferable to large-scale reactors without significant loss of yield or selectivity.
How to Synthesize 3-Azabicyclo[4.1.0]heptane-2-carboxylic Acid Efficiently
To implement this synthesis effectively, operators must adhere to strict temperature controls and reagent addition rates, particularly during the exothermic cycloaddition and the low-temperature reduction phases. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and workup procedures necessary to achieve the reported yields. It is essential to maintain an inert atmosphere during the ylide formation and cyanation steps to prevent moisture interference, which could deactivate the reagents and lower the overall efficiency. Proper quenching protocols are also critical, especially when handling reactive borohydrides and Lewis acids, to ensure operator safety and product stability. By following these optimized parameters, manufacturing teams can reliably reproduce the high-quality results demonstrated in the patent examples.
- Perform [1+2] cycloaddition on compound II using sulfur ylide reagent to form the bicyclic ketone compound III.
- Reduce compound III using triethylborohydride at low temperature (-78 to -30°C) to obtain the hydroxy intermediate compound IV.
- React compound IV with trimethylsilyl cyanide and a Lewis acid catalyst to introduce the cyano group, yielding compound V.
- Hydrolyze the cyano group and remove the protecting group under acidic or basic conditions to finalize 3-azabicyclo[4.1.0]heptane-2-carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from a nine-step legacy process to this streamlined four-step methodology offers profound commercial advantages that extend far beyond simple yield improvements. For procurement managers, the reduction in step count directly correlates to a significant decrease in raw material consumption, solvent usage, and labor hours, all of which contribute to substantial cost savings in API manufacturing. The elimination of hazardous reagents like benzyl bromide removes the need for expensive safety containment systems and specialized waste disposal services, further driving down the operational overhead. Additionally, the use of readily available starting materials mitigates the risk of supply chain bottlenecks, ensuring a continuous flow of production inputs even during periods of market volatility. This reliability is crucial for maintaining production schedules and meeting the demanding delivery timelines expected by downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The drastic simplification of the synthetic route eliminates multiple isolation and purification stages, which are typically the most cost-intensive parts of chemical manufacturing. By reducing the number of unit operations, the process minimizes solvent recovery costs and energy consumption associated with heating and cooling cycles across multiple reactors. Furthermore, the higher overall yield means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold. This economic efficiency allows for more competitive pricing strategies without compromising on margin, providing a distinct advantage in the global marketplace for specialty chemicals.
- Enhanced Supply Chain Reliability: Sourcing raw materials for complex multi-step syntheses often involves managing a fragmented supplier base, which increases the risk of delays and quality inconsistencies. This new method relies on commodity chemicals that are widely produced and stocked by multiple vendors, thereby diversifying the supply base and enhancing resilience against disruptions. The simplified logistics of managing fewer reagents also reduce the administrative burden on procurement teams, allowing them to focus on strategic sourcing rather than fire-fighting supply shortages. Consequently, this leads to more predictable lead times and a more stable inventory profile, which is essential for just-in-time manufacturing models.
- Scalability and Environmental Compliance: The operational simplicity of this route makes it highly amenable to scale-up, as there are fewer critical control points that could fail during technology transfer from lab to plant. The avoidance of toxic reagents and the reduction in waste volume align perfectly with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for the manufacturer. This green chemistry profile not only facilitates smoother regulatory approvals but also enhances the corporate sustainability image, which is becoming a key differentiator for suppliers in the eyes of major pharmaceutical partners. The ability to scale from kilograms to tons with consistent quality ensures that the supply chain can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, raw material sourcing, and environmental impact. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for their own production lines or supply chains.
Q: How does the new synthesis route compare to conventional methods in terms of yield?
A: The novel 4-step route described in patent CN110092753B achieves a total yield of up to 35.4%, which is a substantial improvement over the 0.69% yield reported in previous 9-step conventional methods.
Q: Are the raw materials for this process readily available for large-scale production?
A: Yes, the process utilizes tert-butyl 2-oxo-5,6-dihydropyridine-1(2H)-carboxylate as a starting material, which is inexpensive and easily accessible, facilitating cost-effective commercial scale-up.
Q: Does this method involve hazardous reagents that complicate environmental compliance?
A: Unlike prior art routes that require hazardous reagents like benzyl bromide, this method employs safer reagents and simpler operational conditions, significantly reducing environmental burden and safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Azabicyclo[4.1.0]heptane-2-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in delivering high-value pharmaceutical intermediates to the global market. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-azabicyclo[4.1.0]heptane-2-carboxylic acid meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this process further, ensuring cost-effectiveness and supply continuity for our partners.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and growth in your supply chain.
