Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Manufacturing
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Manufacturing
The integration of fluorine atoms into heterocyclic scaffolds remains a cornerstone strategy in modern medicinal chemistry, particularly for enhancing metabolic stability and lipophilicity in drug candidates. As detailed in patent CN110467579B, a significant breakthrough has been achieved in the preparation of 5-trifluoromethyl substituted 1,2,4-triazole compounds, a structural motif prevalent in numerous bioactive agents. The 1,2,4-triazole ring system serves as a critical pharmacophore in a wide array of therapeutic classes, ranging from antifungal agents like fluconazole to aromatase inhibitors such as letrozole, as well as iron chelators like deferasirox. 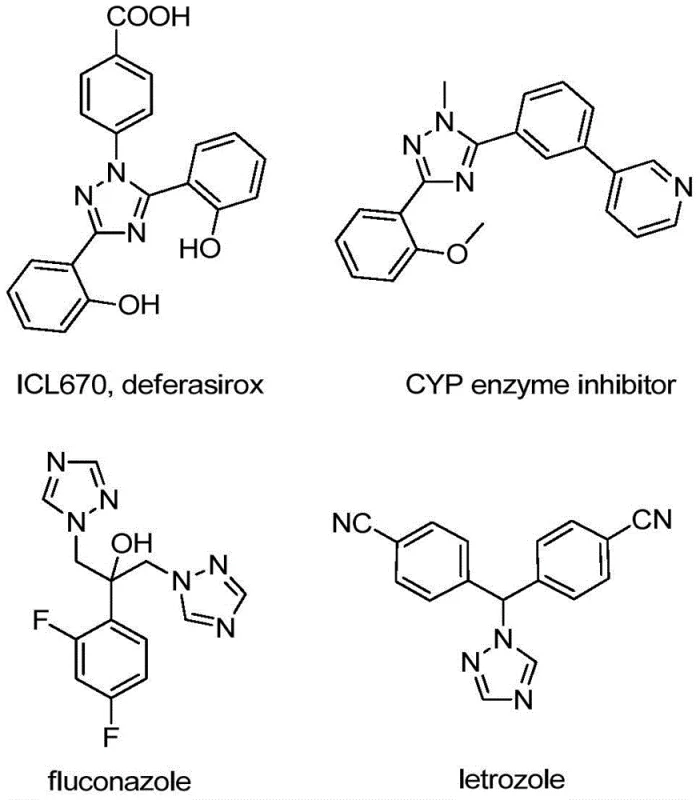 . For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this new synthetic methodology is essential for securing high-purity building blocks that can accelerate drug discovery pipelines while mitigating supply chain risks associated with complex fluorination chemistries.
. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this new synthetic methodology is essential for securing high-purity building blocks that can accelerate drug discovery pipelines while mitigating supply chain risks associated with complex fluorination chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group onto a nitrogen-containing heterocycle has presented substantial synthetic challenges, often relying on methodologies that are ill-suited for large-scale industrial application. Traditional approaches typically involve the direct trifluoromethylation of pre-synthesized triazole rings using specialized reagents such as Togni or Umemoto reagents, which are not only prohibitively expensive but also generate significant amounts of hazardous waste. Alternatively, strategies employing trifluorodiazoethane require stringent safety protocols due to the explosive nature of diazo compounds, posing severe risks for commercial scale-up of complex pharmaceutical intermediates. Furthermore, many existing catalytic systems depend on precious transition metals like palladium or copper, which necessitate rigorous purification steps to remove trace metal residues to meet regulatory standards for active pharmaceutical ingredients (APIs). These legacy methods often suffer from narrow substrate scope, poor functional group tolerance, and the absolute requirement for anhydrous and oxygen-free environments, driving up operational costs and extending lead times for high-purity material delivery.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in patent CN110467579B offers a streamlined, metal-free alternative that leverages inexpensive and commercially available starting materials. This innovative route utilizes trifluoroethylimidoyl chloride and hydrazones as the primary synthons, reacting them in the presence of sodium acetate and elemental iodine to directly construct the trifluoromethylated triazole core. 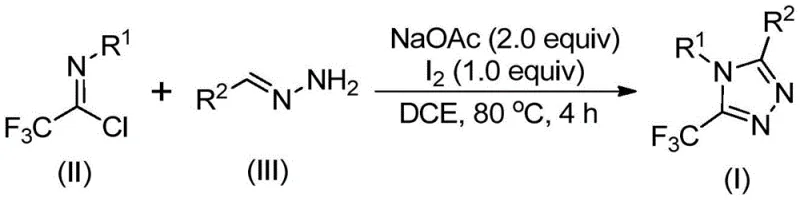 . This approach eliminates the need for exotic fluorinating agents or dangerous diazo precursors, instead relying on a robust oxidative cyclization mechanism that proceeds efficiently in common organic solvents like dichloroethane (DCE). By avoiding the use of heavy metal catalysts, this process inherently simplifies the downstream purification workflow, thereby facilitating cost reduction in API manufacturing and ensuring that the final product meets stringent purity specifications without the need for complex scavenging technologies. The operational simplicity, combined with the ability to tolerate a wide variety of substituents on both the aryl and heteroaryl rings, makes this method highly attractive for the rapid generation of diverse compound libraries.
. This approach eliminates the need for exotic fluorinating agents or dangerous diazo precursors, instead relying on a robust oxidative cyclization mechanism that proceeds efficiently in common organic solvents like dichloroethane (DCE). By avoiding the use of heavy metal catalysts, this process inherently simplifies the downstream purification workflow, thereby facilitating cost reduction in API manufacturing and ensuring that the final product meets stringent purity specifications without the need for complex scavenging technologies. The operational simplicity, combined with the ability to tolerate a wide variety of substituents on both the aryl and heteroaryl rings, makes this method highly attractive for the rapid generation of diverse compound libraries.
Mechanistic Insights into Iodine-Promoted Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated example of base-promoted heterocycle construction that avoids the pitfalls of radical-based trifluoromethylation. The reaction is initiated by the nucleophilic attack of the hydrazone nitrogen on the electrophilic carbon of the trifluoroethylimidoyl chloride, facilitated by sodium acetate acting as a mild base. This initial intermolecular carbon-nitrogen bond formation yields a trifluoroacetamidine intermediate, which subsequently undergoes isomerization to align the reactive centers for cyclization. The critical step involves the addition of elemental iodine, which acts as a mild oxidant to promote the oxidative iodination of the intermediate species. This generates an iodine-substituted transient species that is primed for intramolecular electrophilic substitution. The final aromatization step releases the iodine species and closes the 1,2,4-triazole ring, resulting in the stable 5-trifluoromethyl substituted product. This mechanism is particularly elegant because it circumvents the need for strong oxidants or harsh acidic conditions that might degrade sensitive functional groups on the substrate, ensuring high fidelity in the synthesis of complex molecular architectures.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed routes. Since the reaction does not involve radical chains that can lead to uncontrolled polymerization or side-reactions with solvent molecules, the impurity profile is generally cleaner and more predictable. The use of sodium acetate ensures that the reaction medium remains buffered, preventing the hydrolysis of the acid-sensitive imidoyl chloride starting material before it can react with the hydrazone. Furthermore, the byproduct of the oxidative step is primarily inorganic iodide salts, which are easily removed during the aqueous workup phase, unlike organometallic byproducts that often co-elute with the product during chromatography. This inherent cleanliness of the reaction profile translates directly to higher isolated yields and reduced solvent consumption during purification, addressing key concerns for high-purity pharmaceutical intermediate production where residual solvent and impurity limits are tightly regulated by global health authorities.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for scalability and ease of operation, making it suitable for both laboratory discovery and pilot plant production. The protocol begins with the simple mixing of stoichiometric amounts of trifluoroethylimidoyl chloride, the corresponding hydrazone derivative, and sodium acetate in a non-protic solvent such as 1,2-dichloroethane. The mixture is heated to a moderate temperature range of 80°C to 100°C, allowing the initial condensation to proceed to completion over a period of 2 to 4 hours. Following this incubation, elemental iodine is introduced to the reaction vessel to drive the oxidative cyclization, with the reaction continuing for an additional 1 to 2 hours. Detailed standardized synthesis steps are provided in the guide below.
- Combine sodium acetate, trifluoroethylimidoyl chloride, and hydrazone substrates in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to approximately 80°C and maintain stirring for 2 to 4 hours to facilitate initial condensation.
- Introduce elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative cyclization and aromatization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical efficiency. The shift away from precious metal catalysis and hazardous reagents fundamentally alters the cost structure and risk profile of producing these valuable heterocyclic intermediates. By leveraging commodity chemicals and simplified processing conditions, manufacturers can achieve significant operational efficiencies that translate into more competitive pricing and reliable delivery schedules for their downstream clients.
- Cost Reduction in Manufacturing: The most immediate economic impact stems from the complete elimination of expensive transition metal catalysts such as palladium or rhodium, which are subject to volatile market pricing and supply constraints. Additionally, the avoidance of specialized trifluoromethylating reagents, which often carry high price tags due to complex synthesis and low atom economy, further drives down the raw material costs. The simplified workup procedure, which does not require specialized metal scavengers or extensive chromatographic purification to remove heavy metals, reduces solvent usage and labor hours, leading to substantial overall cost savings in the production of these intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like hydrazones and trifluoroethylimidoyl chloride ensures a robust supply chain that is less susceptible to disruptions compared to routes dependent on niche reagents. Hydrazones can be easily synthesized from abundant aldehydes and hydrazine, while the imidoyl chlorides are stable and commercially accessible. This accessibility allows for the maintenance of healthy inventory levels and reduces the lead time for high-purity intermediates, ensuring that drug development timelines are not compromised by material shortages. Furthermore, the stability of the reagents allows for long-term storage without significant degradation, adding another layer of security to the supply chain.
- Scalability and Environmental Compliance: The operational simplicity of this method, which does not require strictly anhydrous or anaerobic conditions, makes it highly amenable to scale-up from gram to multi-kilogram quantities without the need for specialized reactor infrastructure. The absence of toxic heavy metals simplifies waste treatment protocols, reducing the environmental footprint and disposal costs associated with the manufacturing process. This alignment with green chemistry principles not only lowers regulatory compliance burdens but also enhances the sustainability profile of the final pharmaceutical product, a factor of increasing importance to global stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-trifluoromethyl-1,2,4-triazoles using this novel methodology. These insights are derived directly from the technical specifications and experimental data provided in the underlying patent literature, offering clarity on the feasibility and advantages of this synthetic route for potential partners and collaborators.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes elemental iodine and sodium acetate, completely avoiding the need for costly palladium or copper catalysts often found in traditional cross-coupling methods.
Q: What is the substrate scope for this trifluoromethylation method?
A: The method demonstrates broad tolerance, successfully accommodating various substituted aryl groups, heteroaryl rings like thiophene, and alkenyl chains on both the imidoyl chloride and hydrazone components.
Q: Are strict anhydrous conditions necessary for this reaction?
A: Unlike many organometallic trifluoromethylations, this protocol operates effectively without rigorous anhydrous or anaerobic conditions, significantly simplifying operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both early-stage clinical trials and full-scale commercial manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in fluorine chemistry allows us to navigate the complexities of trifluoromethylated scaffold synthesis with precision, guaranteeing consistency and reliability for our global partners.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your supply chain objectives.
