Advanced Asymmetric Hydrogenation for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
Introduction to Breakthrough Catalytic Technology in Fine Chemical Synthesis
The landscape of fine chemical manufacturing, particularly for chiral pharmaceutical intermediates, is constantly evolving to meet stricter purity standards and sustainability goals. Patent CN1753857A introduces a transformative method for preparing optically active carboxylic acids with high optical purity, addressing one of the most persistent challenges in homogeneous catalysis: the separation and recovery of expensive transition metal catalysts. This technology leverages a specialized sulfonated BINAP-Ru complex that functions effectively in an aqueous or biphasic solvent system, fundamentally altering the economic and operational dynamics of asymmetric hydrogenation. By enabling the catalyst to reside in a distinct aqueous phase while the product partitions into an organic phase, this innovation eliminates the need for energy-intensive distillation or complex recrystallization steps traditionally required to remove residual metals. For R&D directors and process chemists, this represents a significant leap forward in designing greener, more efficient synthetic routes for high-value chiral building blocks used in drugs and liquid crystal materials.
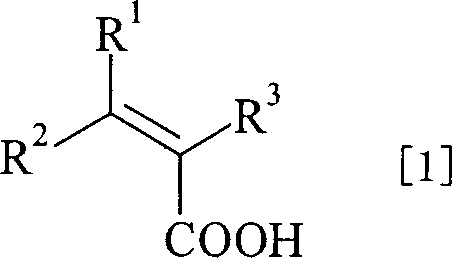
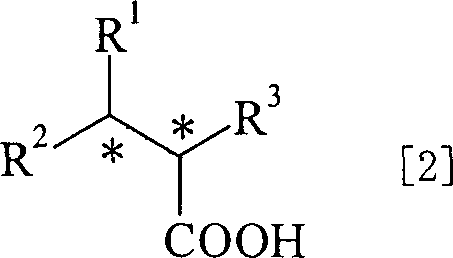
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In traditional homogeneous catalytic processes for synthesizing chiral carboxylic acids, the catalyst and the product typically share the same organic phase, creating a formidable downstream processing bottleneck. Most drug intermediates produced via these methods are solids that are notoriously difficult to separate from the catalyst solely through distillation, often necessitating multiple purification cycles such as heavy metal scavenging, extensive washing, or repeated recrystallization. These additional unit operations not only increase the overall production cost but also lead to significant yield losses and generate substantial chemical waste, complicating environmental compliance. Furthermore, the inability to easily recover the precious metal catalyst means that each batch requires a fresh charge of expensive ligands and metals, driving up the raw material costs significantly. The accumulation of metal impurities in the final product is also a critical quality concern for pharmaceutical applications, where strict limits on residual heavy metals must be met, often requiring dedicated and costly purification technologies.
The Novel Approach
The novel approach detailed in the patent data utilizes a water-soluble sulfonated BINAP-Ru complex to create a biphasic reaction system that inherently solves the separation problem. By conducting the asymmetric hydrogenation in a mixture of water and a water-insoluble organic solvent, the reaction exploits the differential solubility of the components: the hydrophobic product dissolves in the organic layer, while the hydrophilic catalyst remains dissolved in the aqueous layer. This physical separation allows for the catalyst to be recovered simply by decanting or separating the aqueous phase after the reaction is complete, bypassing the need for complex chemical treatments. Moreover, this aqueous catalyst solution can be directly recycled and reused for subsequent batches without extensive reprocessing, maintaining high catalytic activity and enantioselectivity over multiple cycles. This methodology not only streamlines the workflow but also drastically reduces the consumption of precious metals, aligning perfectly with the principles of green chemistry and sustainable manufacturing practices.
Mechanistic Insights into Sulfonated BINAP-Ru Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the unique structure of the sulfonated BINAP-Ru complex, specifically represented by the formula [RuX(aromatic){(SO3M)2-BINAP}]X, where the introduction of sulfonate groups (-SO3M) onto the BINAP ligand backbone imparts crucial water solubility. Unlike standard BINAP ligands which are strictly lipophilic, the sulfonated derivative allows the ruthenium center to operate effectively at the interface or within the aqueous phase of a biphasic system. The mechanism involves the coordination of the alpha,beta-unsaturated carboxylic acid substrate to the ruthenium center, followed by the stereoselective addition of hydrogen across the double bond. The steric and electronic environment created by the bulky binaphthyl backbone and the diphenylphosphine groups ensures high enantiofacial discrimination, leading to the formation of the desired optical isomer with high excess. The presence of the sulfonate groups does not merely aid solubility; it stabilizes the catalyst in the aqueous medium, preventing leaching into the organic product phase and ensuring that the metal stays where it is needed for reuse.
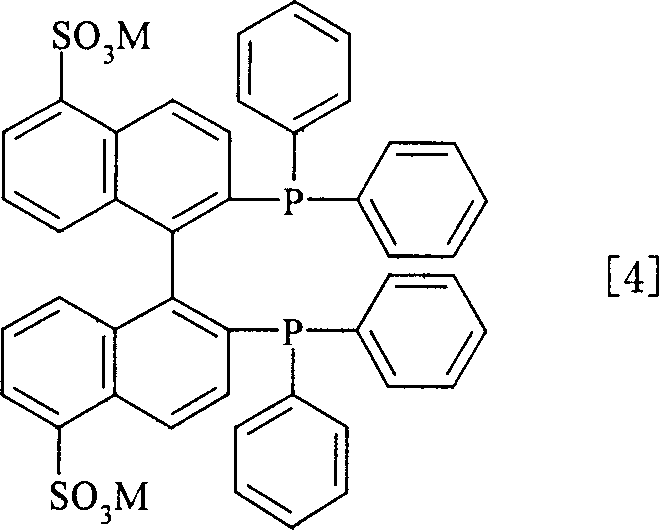
Controlling impurities in this system is inherently managed by the phase separation mechanism, which acts as a continuous purification step during the reaction workup. Since the catalyst is confined to the aqueous phase, the risk of metal contamination in the final organic product is minimized compared to single-phase systems where equilibrium distribution can lead to trace metal retention. The patent data indicates that high optical purity, often exceeding 90% ee and reaching up to 94.8% ee in specific examples like (2R)-methylbutanoic acid synthesis, is consistently achieved. This high level of stereocontrol is maintained even when the catalyst solution is recycled, suggesting that the active catalytic species remains stable and does not degrade into non-selective forms under the reaction conditions. For quality control teams, this implies a robust process with a predictable impurity profile, reducing the analytical burden and ensuring batch-to-batch consistency essential for regulatory filings.
How to Synthesize Optically Active Carboxylic Acids Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting, emphasizing the simplicity of the biphasic operation. The process begins with charging a reactor with the alpha,beta-unsaturated carboxylic acid substrate and the sulfonated BINAP-Ru catalyst, followed by the addition of a degassed mixture of water and a water-insoluble organic solvent such as dichloromethane or diisopropyl ether. The detailed standardized synthesis steps see the guide below for specific molar ratios and safety precautions regarding hydrogen handling.
- Prepare the reaction mixture by combining the alpha,beta-unsaturated carboxylic acid substrate with the sulfonated BINAP-Ru complex catalyst in a biphasic solvent system consisting of water and a water-insoluble organic solvent.
- Conduct asymmetric hydrogenation under controlled hydrogen pressure (0.5-10 MPa) and temperature (30-100°C) to ensure high conversion and enantioselectivity.
- Upon completion, separate the aqueous phase containing the catalyst from the organic phase containing the product, allowing for direct recycling of the catalyst solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous biphasic hydrogenation technology offers compelling economic and logistical advantages that extend beyond mere technical feasibility. The primary value driver is the drastic simplification of the downstream processing workflow, which translates directly into reduced operational expenditures and shorter manufacturing cycle times. By eliminating the need for complex catalyst removal steps like column chromatography or specialized metal scavenging resins, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. The ability to recycle the catalyst solution multiple times means that the effective cost per kilogram of the precious metal catalyst is amortized over many batches, leading to substantial raw material savings. Furthermore, the use of water as a primary solvent component reduces the reliance on large volumes of volatile organic compounds, lowering solvent procurement costs and waste disposal fees associated with hazardous organic waste streams.
- Cost Reduction in Manufacturing: The economic impact of this process is profound because it removes several expensive unit operations from the production line. Traditional methods often require stoichiometric amounts of additives or expensive purification media to meet metal specifications, whereas this method relies on physical phase separation which is virtually cost-free in terms of consumables. The recycling capability of the catalyst means that the initial investment in the sophisticated ruthenium complex is spread across a much larger output volume, effectively lowering the variable cost of goods sold. Additionally, the high conversion rates and selectivity reported in the patent examples minimize the formation of by-products, thereby maximizing the yield of the desired API intermediate and reducing the cost associated with waste treatment and lost material.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this catalytic system enhances reliability by reducing dependency on single-use reagents that might face availability fluctuations. The catalyst precursors and ligands, while specialized, are used in such small quantities and with such high efficiency due to recycling that supply risks are mitigated. The process operates under moderate temperatures and pressures that are standard in most multipurpose chemical plants, meaning there is no need for exotic equipment that could introduce bottlenecks or long lead times for installation. This compatibility with existing infrastructure allows for faster technology transfer and quicker ramp-up to commercial production, ensuring a steady supply of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward because the biphasic mixing and separation principles remain consistent regardless of vessel size. The reduced generation of hazardous waste, particularly heavy metal-contaminated organic waste, simplifies environmental compliance and permits management. Using water as a major solvent component aligns with increasingly stringent global environmental regulations regarding VOC emissions and solvent usage. This 'green' credential not only future-proofs the manufacturing site against regulatory changes but also appeals to end-clients who are prioritizing sustainable supply chains, potentially opening up new market opportunities for eco-friendly pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology, derived directly from the experimental data and claims within the patent documentation. These answers are designed to provide clarity on the operational parameters and the tangible benefits observed during the development of this process.
Q: How does the sulfonated BINAP-Ru catalyst improve separation efficiency compared to traditional homogeneous catalysts?
A: Traditional homogeneous catalysts often require complex distillation or recrystallization steps for separation because they dissolve in the same organic phase as the product. The sulfonated BINAP-Ru complex described in patent CN1753857A is water-soluble due to its sulfonate groups, enabling a biphasic system where the catalyst remains in the aqueous layer while the product extracts into the organic layer, allowing for simple phase separation.
Q: Can the catalyst solution be reused without loss of optical purity?
A: Yes, the patent data demonstrates that the aqueous catalyst solution can be recycled multiple times. Experimental results show that even after multiple cycles, the optical purity (ee) remains stable, typically maintaining levels above 92% ee, which ensures consistent quality for pharmaceutical intermediates.
Q: What are the typical reaction conditions for this asymmetric hydrogenation process?
A: The process operates effectively under moderate conditions, with reaction temperatures generally ranging from 30°C to 100°C and hydrogen pressures between 0.5 MPa and 10 MPa. These conditions are compatible with standard industrial hydrogenation equipment, facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the sulfonated BINAP-Ru hydrogenation process are successfully translated into robust industrial operations. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to verify optical purity and residual metal levels for every batch. Our capability to handle complex chiral syntheses allows us to offer customized solutions that optimize both cost and quality for our international partners.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise in asymmetric hydrogenation can enhance your supply chain efficiency and product quality.
