Advanced Solid Acid Catalysis for High-Purity p-Hydroxyphenylhydantoin Production and Commercial Scale-Up
Advanced Solid Acid Catalysis for High-Purity p-Hydroxyphenylhydantoin Production and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with environmental sustainability, particularly for critical intermediates like p-hydroxyphenylhydantoin. Patent CN110330460B introduces a transformative preparation method that leverages heterogeneous solid acid catalysis to overcome the limitations of traditional liquid-phase synthesis. This technology represents a significant leap forward for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status, as it replaces corrosive liquid acids with recoverable solid catalysts such as acidic ion exchange resins, heteropolyacids, and metal oxides. By utilizing glyoxylic acid, phenol, and urea as feedstocks, this novel approach not only streamlines the reaction workflow but also drastically improves the impurity profile of the final product. The strategic adoption of this patented methodology allows for precise control over reaction kinetics, ensuring consistent quality essential for the synthesis of downstream beta-lactam antibiotics. Furthermore, the elimination of complex neutralization steps inherent in older methods translates directly into operational efficiency and reduced waste treatment burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-hydroxyphenylhydantoin has been plagued by significant technical and environmental hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often rely on homogeneous liquid acid catalysts or toxic raw materials that necessitate rigorous and costly post-reaction processing. These conventional methods typically suffer from low conversion rates, often hovering around suboptimal levels that result in substantial raw material wastage and increased production costs. Moreover, the use of liquid acids generates large volumes of acidic wastewater requiring neutralization, which escalates the environmental footprint and complicates regulatory compliance for modern chemical plants. The difficulty in separating the catalyst from the reaction mixture frequently leads to product contamination, necessitating additional purification steps that erode overall yield and extend lead time for high-purity pharmaceutical intermediates. Consequently, manufacturers have long struggled to find a balance between economic viability and the stringent purity specifications required for antibiotic precursors.
The Novel Approach
In stark contrast, the methodology disclosed in Patent CN110330460B employs a sophisticated solid acid catalytic system that fundamentally redefines the synthesis landscape. By introducing catalysts such as Amberlite-15, phosphotungstic acid, or niobium oxide, the reaction proceeds through a cleaner, more selective pathway that minimizes by-product formation. This innovative strategy facilitates easy catalyst removal via simple hot filtration, thereby eliminating the need for aqueous workups associated with liquid acid neutralization. The process operates effectively under moderate thermal conditions, typically between 60-85°C, which enhances energy efficiency and operational safety compared to harsher traditional protocols. As illustrated in the reaction scheme below, the condensation of glyoxylic acid, phenol, and urea is seamlessly orchestrated by the solid acid surface, promoting high selectivity towards the desired hydantoin ring structure.
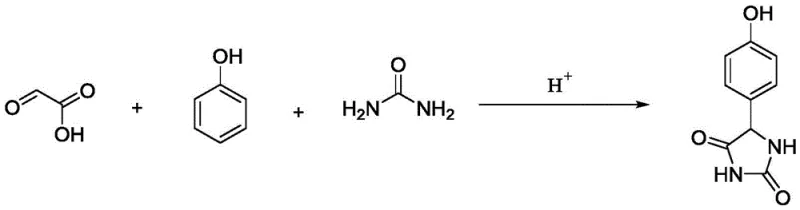
This streamlined approach not only boosts yields to impressive levels, reaching up to 81% in optimized examples, but also ensures product purity exceeds 98%, meeting the rigorous demands of global pharmaceutical supply chains. The versatility of the catalyst selection allows producers to tailor the process to their specific infrastructure, whether utilizing ion exchange resins for continuous flow applications or metal salts for batch processing.
Mechanistic Insights into Solid Acid-Catalyzed Cyclization
The core of this technological advancement lies in the unique mechanistic role played by the solid acid catalyst in facilitating the electrophilic substitution and subsequent cyclization reactions. The surface acid sites of catalysts like sulfonated polystyrene resins or heteropolyacids provide localized proton donors that activate the carbonyl group of glyoxylic acid, making it more susceptible to nucleophilic attack by phenol. This activation lowers the energy barrier for the initial condensation step, driving the reaction forward even at relatively mild temperatures. Unlike homogeneous acids that disperse protons randomly throughout the solution, solid acids offer a structured environment that can enhance regioselectivity, favoring the para-substitution required for the target molecule. The porous structure of certain resins further aids in concentrating reactants within the catalyst matrix, effectively increasing the local concentration and collision frequency. This micro-environment effect is crucial for suppressing side reactions such as polymerization or over-oxidation, which are common pitfalls in non-catalyzed or liquid-acid systems.
Furthermore, the mechanism inherently supports superior impurity control, a critical factor for R&D directors focused on purity and impurity profiles. The solid nature of the catalyst prevents the leaching of metal ions or acidic residues into the product stream, a frequent issue with liquid mineral acids that can complicate downstream crystallization. By maintaining a heterogeneous system, the reaction equilibrium is managed more effectively, allowing for the continuous removal of water or other by-products if the system is designed for azeotropic distillation. The selectivity of the catalyst ensures that the urea component reacts specifically to close the hydantoin ring without degrading the sensitive phenolic moiety. This precision results in a crude product that requires minimal recrystallization to achieve pharmaceutical grade standards. The ability to tune the acidity and pore size of the solid catalyst provides an additional layer of process control, enabling manufacturers to optimize the trade-off between reaction rate and selectivity based on real-time monitoring.
How to Synthesize p-Hydroxyphenylhydantoin Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits of the solid acid catalyst. The process begins with the precise weighing and mixing of glyoxylic acid, phenol, and urea, typically in a molar ratio optimized to drive the equilibrium towards product formation while minimizing excess reagent costs. The choice of solvent, ranging from water to organic mediums like toluene or butanone, depends on the solubility profile of the specific catalyst chosen and the desired isolation method. Once the reactants are homogenized, the solid acid catalyst is introduced, and the mixture is heated to the target temperature range, where it is maintained for a duration sufficient to achieve high conversion. Detailed standard operating procedures regarding catalyst loading, filtration temperatures, and crystallization cooling rates are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the guide below.
- Mix glyoxylic acid, phenol, and urea in a reactor with a molar ratio ranging from 3-6: 2-4:5-7, optionally adding a solvent like water or toluene.
- Add a solid acid catalyst such as Amberlite-15, phosphotungstic acid, or metal oxides (e.g., Nb2O5) at a loading of 0.01-10 molar ratio relative to glyoxylic acid.
- Heat the mixture to 60-85°C and react for 4-24 hours, then filter hot to remove the catalyst and cool the filtrate to crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solid acid-catalyzed process offers profound strategic advantages that extend beyond mere chemical yield. The elimination of liquid acid neutralization steps drastically simplifies the downstream processing workflow, reducing the consumption of bases and the generation of saline waste streams. This simplification directly correlates to cost reduction in pharmaceutical intermediate manufacturing by lowering utility costs associated with wastewater treatment and hazardous waste disposal. Additionally, the robustness of solid catalysts like ion exchange resins allows for potential regeneration and reuse, further diminishing the recurring cost of consumables over long production campaigns. The improved safety profile, characterized by moderate operating temperatures and the absence of corrosive liquid acids, reduces the risk of workplace incidents and lowers insurance and compliance overheads. These factors collectively contribute to a more resilient and cost-effective supply chain capable of withstanding market fluctuations.
- Cost Reduction in Manufacturing: The shift to heterogeneous catalysis removes the expensive and time-consuming neutralization and extraction steps typically required with liquid acids. By simplifying the workup to a mere filtration and crystallization, manufacturers can significantly reduce labor hours and solvent consumption. The high selectivity of the catalyst minimizes the formation of hard-to-remove impurities, thereby increasing the overall mass balance and reducing the loss of valuable starting materials. This efficiency gain translates into substantial cost savings per kilogram of finished product, enhancing competitiveness in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available and stable solid acid catalysts mitigates the risk of supply disruptions often associated with specialized or hazardous liquid reagents. The process tolerance for various solvent systems, including water, provides flexibility in sourcing raw materials based on regional availability and price volatility. Furthermore, the simplified process flow reduces the likelihood of batch failures due to operational errors, ensuring a consistent and reliable output of critical intermediates. This stability is paramount for maintaining uninterrupted production schedules for downstream antibiotic manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the solid acid method make it ideally suited for commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production. The reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing facility against tighter emission standards. The ability to operate in aqueous or green solvent systems further enhances the sustainability profile of the production process, appealing to eco-conscious partners and stakeholders. This alignment with green chemistry principles not only reduces regulatory friction but also enhances the brand reputation of the manufacturer as a responsible corporate citizen.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the process feasibility. Understanding these nuances is crucial for making informed decisions about technology adoption and process integration. The answers reflect the practical realities of scaling this chemistry in an industrial setting.
Q: What are the primary advantages of using solid acid catalysts over traditional liquid acids for p-hydroxyphenylhydantoin synthesis?
A: Solid acid catalysts eliminate the need for neutralization steps, significantly reducing aqueous waste generation and simplifying product isolation through simple filtration, thereby lowering environmental compliance costs.
Q: What is the typical yield and purity achievable with this patented method?
A: According to Patent CN110330460B, the method achieves yields between 70% and 81% with product purity consistently exceeding 98%, making it highly suitable for downstream antibiotic synthesis.
Q: Can this process be scaled for industrial production of beta-lactam intermediates?
A: Yes, the process utilizes robust catalysts like ion exchange resins and operates at moderate temperatures (60-85°C), ensuring excellent thermal safety and scalability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Hydroxyphenylhydantoin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain, particularly for life-saving beta-lactam antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. By leveraging technologies such as the solid acid catalysis method described in Patent CN110330460B, we can offer our partners a superior balance of cost, quality, and sustainability. Our infrastructure is designed to handle complex chemistries with the utmost safety and precision, guaranteeing supply continuity even in volatile market conditions.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your long-term strategic partner in the fine chemical sector.
