Scalable Synthesis of 2-(1-Alkyl-1H-pyrazol-4-yl)morpholine Intermediates for Kinase Inhibitors
The pharmaceutical landscape for kinase inhibitors is continuously evolving, with the morpholine-pyrazole scaffold emerging as a critical pharmacophore for targeting Phosphatidylinositol 4-Kinase III beta (PI4KIIIβ). Patent CN111377867B, published on September 6, 2022, introduces a groundbreaking synthetic methodology for producing 2-(1-alkyl-1H-pyrazol-4-yl)morpholine intermediates, specifically addressing the technical voids in current manufacturing capabilities. This innovation provides a robust pathway for generating key building blocks where R represents methyl, ethyl, or isopropyl groups, which are essential for the development of next-generation antiviral and antitumor agents. For R&D directors and procurement strategists, this patent represents a pivotal shift from hazardous, low-yielding legacy processes to a streamlined, industrially viable protocol that enhances both safety and supply chain reliability.
The strategic value of this technology lies in its ability to bypass complex protection-deprotection sequences and hazardous reagents typically associated with morpholine ring construction. By leveraging a Weinreb amide acylation strategy followed by a novel silane-mediated reductive cyclization, the process achieves high purity profiles essential for regulatory compliance. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is crucial for securing a stable supply of high-quality raw materials for clinical and commercial programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in patent WO2008087512, rely on convoluted synthetic routes that pose significant challenges for commercial scale-up. As illustrated in the reaction scheme below, the conventional approach begins with a bromopyridine derivative and requires a multi-step sequence involving epoxide formation and ring opening. A critical bottleneck occurs in the morpholine ring closure step, which utilizes chloroacetyl chloride under harsh conditions, resulting in a dismal yield of only 14% for that specific transformation. Furthermore, the final reduction step necessitates the use of lithium aluminum hydride (LiAlH4), a reagent known for its pyrophoric nature and difficult handling requirements on a large scale.
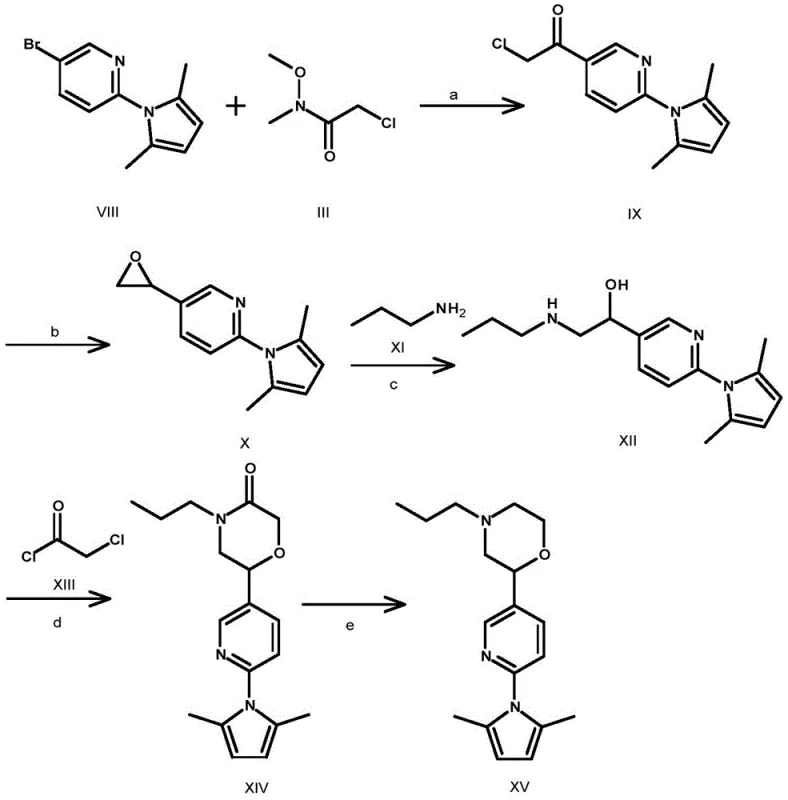
The cumulative effect of these inefficiencies is a process with poor atom economy and substantial waste generation. The requirement for cryogenic conditions (-78°C) in early steps and extended reaction times (up to 4 days for certain transformations) drastically increases energy consumption and operational costs. For supply chain heads, these factors translate into longer lead times and higher vulnerability to production disruptions. The reliance on hazardous reagents like chloroacetyl chloride also imposes strict environmental controls and waste disposal costs, making the conventional route economically unviable for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN111377867B offers a streamlined alternative that directly addresses these pain points. The new route initiates with the metalation of a 4-halo-1-R-1H-pyrazole followed by acylation with 2-chloro-N-methoxy-N-methylacetamide (a Weinreb amide). This strategy ensures precise control over the carbonyl addition, preventing over-alkylation and delivering the ketone intermediate in high purity. Subsequent nucleophilic substitution with N-benzylethanolamine installs the necessary side chain under mild basic conditions, avoiding the extreme temperatures and hazardous reagents of the prior art.
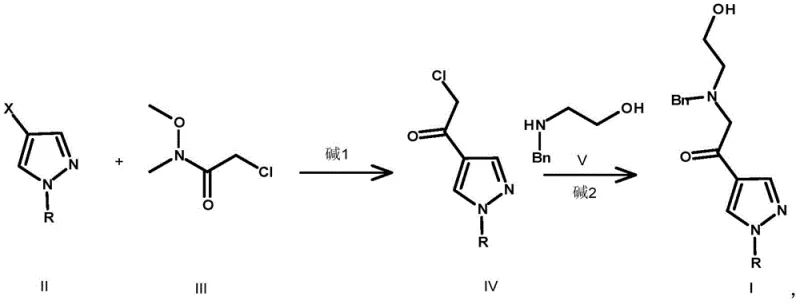
The true innovation lies in the one-pot reductive cyclization step, where trifluoroacetic acid and triethylsilane facilitate the simultaneous reduction of the ketone and cyclization to form the morpholine ring. This telescoped operation eliminates the need for isolating unstable intermediates and removes the requirement for LiAlH4. Finally, the benzyl protecting group is cleanly removed via catalytic hydrogenation or chloroethyl chloroformate treatment. This approach not only improves the overall yield significantly but also simplifies the purification workflow, offering a clear path for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Silane-Mediated Reductive Cyclization
The core chemical transformation in this novel pathway is the conversion of the amino-alcohol ketone intermediate (Compound I) into the protected morpholine (Compound VI). This process relies on the generation of an iminium ion species under acidic conditions (TFA), which is subsequently reduced by triethylsilane. The mechanism proceeds through the protonation of the carbonyl oxygen, followed by nucleophilic attack from the pendant hydroxyl group to form a hemiketal-like intermediate. Dehydration leads to an oxocarbenium or iminium species, which is rapidly reduced by the hydride source (Et3SiH) to lock in the morpholine ring structure. This chemoselective reduction is highly advantageous as it tolerates the pyrazole nitrogen and other sensitive functional groups without requiring orthogonal protection strategies.
Impurity control is inherently built into this mechanism. The use of the Weinreb amide in the initial acylation step prevents the formation of tertiary alcohol byproducts that often plague Grignard reactions with esters or acid chlorides. Furthermore, the mild conditions of the cyclization step (0-100°C) minimize thermal degradation and polymerization side reactions common in high-temperature closures. The final deprotection step using Pd/C and hydrogen ensures that any remaining olefinic impurities are also reduced, resulting in a final API intermediate with a superior impurity profile suitable for stringent regulatory filings.
How to Synthesize 2-(1-Alkyl-1H-pyrazol-4-yl)morpholine Efficiently
The synthesis protocol outlined in the patent provides a detailed roadmap for laboratory and pilot-scale production. The process begins with the preparation of the ketone intermediate using either n-butyllithium at cryogenic temperatures or isopropylmagnesium chloride at ambient temperatures, offering flexibility based on available infrastructure. The subsequent alkylation and cyclization steps are robust and have been demonstrated with various alkyl substituents (methyl, ethyl, isopropyl), confirming the method's versatility. For detailed operational parameters and specific workup procedures, please refer to the standardized guide below.
- React 4-halo-1-R-1H-pyrazole with 2-chloro-N-methoxy-N-methylacetamide using n-BuLi or Grignard reagents to form the ketone intermediate.
- Perform nucleophilic substitution with N-benzylethanolamine using carbonate bases to install the ethanolamine side chain.
- Execute a one-pot reductive cyclization using trifluoroacetic acid and triethylsilane to close the morpholine ring.
- Remove the benzyl protecting group via catalytic hydrogenation or chloroethyl chloroformate treatment to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers transformative benefits for procurement managers and supply chain directors. The elimination of lithium aluminum hydride and chloroacetyl chloride removes two of the most significant safety and environmental liabilities from the manufacturing process. This shift not only reduces the cost of specialized safety equipment and waste disposal but also broadens the pool of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis, thereby enhancing supply chain resilience.
- Cost Reduction in Manufacturing: The new process achieves substantial cost savings by replacing expensive and hazardous reagents with commodity chemicals like triethylsilane and carbonate bases. The telescoping of the reduction and cyclization into a single pot significantly reduces solvent usage, reactor occupancy time, and labor costs associated with intermediate isolation. By avoiding the low-yielding steps of the prior art, the overall material throughput is improved, leading to a lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials, including 4-bromo-1-alkylpyrazoles and N-benzylethanolamine, are commercially available from multiple global suppliers, reducing the risk of raw material shortages. The robustness of the reaction conditions, particularly the ability to use Grignard reagents at higher temperatures compared to organolithiums, allows for more flexible scheduling and reduces the dependency on specialized cryogenic equipment, ensuring consistent delivery timelines.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, minimizing the generation of heavy metal waste (aluminum salts) and chlorinated organic byproducts. The simplified workup procedures, often involving straightforward extractions and crystallizations, facilitate easier scale-up from kilogram to ton quantities. This environmental compatibility aligns with increasingly strict global regulations on pharmaceutical manufacturing emissions and waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN111377867B, providing a factual basis for decision-making.
Q: What are the critical advantages of this new synthesis route over prior art?
A: The novel route eliminates the use of hazardous chloroacetyl chloride and expensive lithium aluminum hydride (LiAlH4), replacing them with milder silane-mediated cyclization and safer carbonate bases, significantly improving process safety and environmental compliance.
Q: Can this process be scaled for commercial production of kinase inhibitor intermediates?
A: Yes, the process utilizes robust reagents like isopropylmagnesium chloride and potassium carbonate, and features a convergent one-pot cyclization step that reduces unit operations, making it highly suitable for multi-kilogram to ton-scale manufacturing.
Q: What specific alkyl groups are compatible with this synthetic methodology?
A: The patent explicitly demonstrates successful synthesis for R groups including methyl, ethyl, and isopropyl, covering the most common substituents required for PI4KIIIβ inhibitor drug candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(1-Alkyl-1H-pyrazol-4-yl)morpholine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of kinase inhibitor development programs. Our technical team has thoroughly analyzed the pathways described in CN111377867B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 2-(1-alkyl-1H-pyrazol-4-yl)morpholine meets the exacting standards required for clinical trials and commercial launch.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your project timelines while reducing overall expenditure.
