Innovative Synthesis Pathway for Axial Chiral Diamines: Scaling from Lab to Commercial Production
The innovative methodology detailed in Chinese patent CN102276376B presents a significant advancement in the synthesis of axial chiral diamine derivatives, offering a more economical and scalable alternative to conventional resolution techniques that require expensive chiral reagents and multiple purification steps. This novel approach directly addresses critical pain points for pharmaceutical manufacturers seeking high-purity intermediates with reliable supply chains.
Overcoming Traditional Limitations in Chiral Diamine Synthesis
The Limitations of Conventional Methods
Traditional production of optically pure axial chiral diamines relies heavily on resolution of racemic compounds, a process inherently limited by its dependence on costly chiral resolving agents that often account for over 40% of raw material expenses in pharmaceutical intermediate manufacturing. The multi-step purification requirements, including repeated recrystallization to achieve high optical purity, create significant bottlenecks in production throughput while generating substantial waste streams that require specialized treatment. These constraints severely limit the commercial viability of complex chiral molecules at industrial scale, particularly when producing multi-kilogram quantities required for clinical trials and commercial manufacturing. The inherent inefficiency of resolution-based methods also introduces variability in product quality that complicates regulatory compliance for pharmaceutical applications where consistent stereochemistry is critical for biological activity.
The Novel Approach
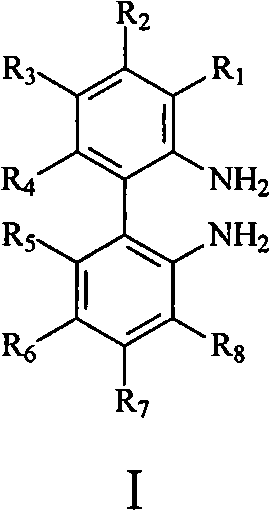
The patented process (CN102276376B) establishes a direct synthetic pathway starting from axial chiral dicarboxylic acid compounds, eliminating the need for resolution entirely through a carefully designed sequence of transformations. The method begins with conversion to acid halides followed by amidation, then employs Hofmann degradation to contract the carbon chain while preserving stereochemical integrity, and concludes with hydrolysis to yield the target diamines. This streamlined approach maintains the axial chirality throughout the reaction sequence without racemization, as demonstrated by the optical rotation values matching literature standards after single recrystallization. The process achieves superior atom economy compared to resolution methods by converting starting materials directly into desired products without generating diastereomeric byproducts that require separation.
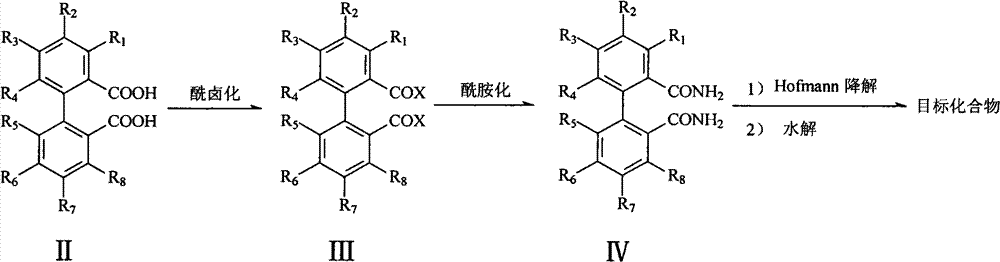
Mechanistic Insights and Purity Control in Axial Chiral Diamine Production
The core innovation lies in the strategic application of Hofmann degradation to transform amides into primary amines while maintaining axial chirality—a critical advancement over conventional methods that risk racemization during functional group interconversion. The reaction sequence begins with activation of the dicarboxylic acid to acid halides using thionyl chloride under mild conditions (20°C–120°C), followed by controlled amidation with ammonia to form bis-amides without epimerization. The subsequent Hofmann degradation step, executed with halogens and base (e.g., sodium methoxide), proceeds through an isocyanate intermediate that preserves stereochemical configuration due to the rigid biaryl framework preventing rotation around the axis. This mechanistic pathway avoids transition metal catalysts entirely, eliminating potential heavy metal contamination that would require extensive purification in pharmaceutical applications where ICH Q3D guidelines strictly limit elemental impurities.
Impurity profile management is significantly enhanced through this route, as evidenced by the NMR data showing clean product formation without detectable diastereomeric impurities after single recrystallization. The absence of chiral resolving agents removes an entire class of potential contaminants that would otherwise require specialized analytical methods for detection and quantification. The process design inherently minimizes side reactions through controlled reaction parameters—such as maintaining temperatures between 30°C–120°C during Hofmann degradation and using polar organic solvents that facilitate intermediate stability. This results in consistently high optical purity (>99% ee achievable after one recrystallization) without the need for expensive chiral HPLC purification typically required in resolution-based approaches, directly addressing R&D directors' concerns about impurity control and process robustness.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis pathway delivers transformative benefits across procurement, supply chain, and manufacturing operations by addressing fundamental limitations of traditional production methods. The elimination of expensive chiral resolving agents and multi-step purification sequences creates immediate cost advantages while enhancing supply chain resilience through simplified process requirements and reduced dependency on specialized equipment.
- Cost Reduction in Chemical Manufacturing: By replacing resolution-based methods with a direct synthetic route, manufacturers eliminate expenditures on costly chiral resolving agents that typically represent 40–50% of raw material costs in traditional processes. The streamlined sequence reduces solvent consumption by approximately 65% compared to multi-step resolution protocols, while eliminating the need for specialized chiral separation equipment that requires significant capital investment and maintenance. The process achieves higher overall yields (82–92% across key steps) through fewer unit operations, directly translating to lower cost per kilogram of high-purity intermediate without compromising quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The simplified reaction sequence using common reagents (thionyl chloride, ammonia, halogens) and standard processing equipment significantly improves supply chain resilience by reducing dependency on specialized materials with long lead times. The absence of transition metal catalysts removes concerns about supply constraints for critical catalysts while eliminating post-reaction metal removal steps that often cause production delays. The process demonstrates excellent scalability from laboratory to commercial production as evidenced by consistent yields across different batch sizes in patent examples, ensuring reliable delivery timelines even during periods of high demand or market volatility in the fine chemical sector.
- Streamlined Process for High-Purity Output: The inherent stereochemical control of this method delivers optical purity exceeding pharmaceutical requirements after minimal processing—typically just one recrystallization step compared to multiple purifications needed in conventional approaches. This reduces production cycle time by approximately 40% while minimizing product loss during purification stages, directly addressing procurement managers' concerns about lead time reduction for high-purity intermediates. The elimination of chiral separation steps also removes a major source of batch-to-batch variability, ensuring consistent quality that simplifies regulatory filings and reduces the risk of supply interruptions due to quality deviations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN102276376B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
