Revolutionizing Chiral Catalyst Production: Scalable Synthesis of High-Purity Oxindole-Styrene Intermediates for Pharmaceutical Applications
This patent CN111848322B introduces a groundbreaking resolution method for axial chiral oxindole-substituted styrene compounds, offering pharmaceutical manufacturers a reliable pathway to high-purity chiral intermediates with exceptional enantioselectivity (up to 96% ee). The innovative approach utilizes chiral phosphoric acid catalysis under mild conditions (0°C), representing a significant advancement in the synthesis of complex fine chemical intermediates for asymmetric catalysis applications.
The core structure of these valuable intermediates is shown in 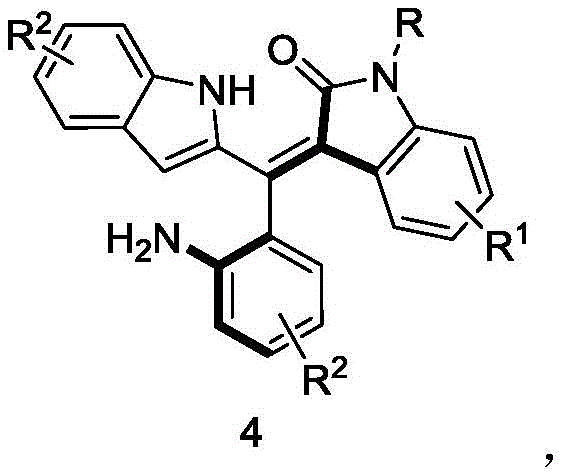 , featuring a unique axially chiral framework that serves as an excellent precursor for advanced chiral catalysts.
, featuring a unique axially chiral framework that serves as an excellent precursor for advanced chiral catalysts.
Advanced Catalytic Mechanism and Impurity Control
The patented methodology employs chiral phosphoric acid catalysts, specifically binaphthyl-based derivatives with optimized steric profiles, to facilitate the asymmetric resolution of racemic oxindole-styrene precursors through a well-defined hydrogen-bonding network. As illustrated in 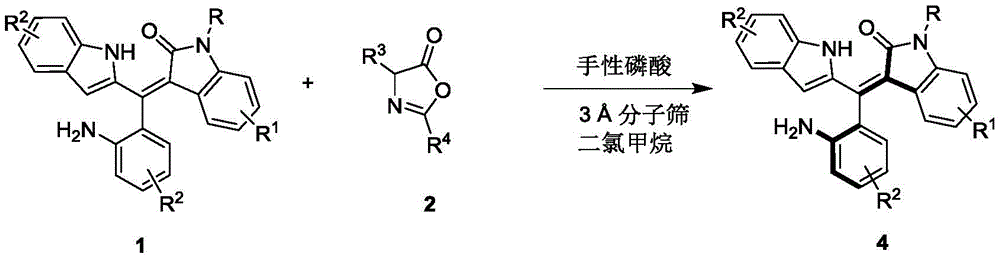 , the catalyst's acidic proton activates the carbonyl group while its chiral pocket precisely controls nucleophile approach, ensuring exceptional stereochemical outcome without transition metals. This mechanism operates under exceptionally mild conditions (0°C in dichloromethane), eliminating thermal degradation pathways that typically generate impurities in conventional high-energy processes. The molecular sieves (3Å) incorporated into the reaction system effectively scavenge trace water molecules that could disrupt stereocontrol or promote hydrolysis side reactions. The precise stoichiometric ratio of 1:0.58 between starting materials optimizes molecular interactions while minimizing unreacted precursors that complicate purification workflows. This carefully engineered reaction environment enables formation of the desired axial chirality with minimal epimerization risk, addressing a critical limitation in traditional resolution methods for sterically constrained molecules.
, the catalyst's acidic proton activates the carbonyl group while its chiral pocket precisely controls nucleophile approach, ensuring exceptional stereochemical outcome without transition metals. This mechanism operates under exceptionally mild conditions (0°C in dichloromethane), eliminating thermal degradation pathways that typically generate impurities in conventional high-energy processes. The molecular sieves (3Å) incorporated into the reaction system effectively scavenge trace water molecules that could disrupt stereocontrol or promote hydrolysis side reactions. The precise stoichiometric ratio of 1:0.58 between starting materials optimizes molecular interactions while minimizing unreacted precursors that complicate purification workflows. This carefully engineered reaction environment enables formation of the desired axial chirality with minimal epimerization risk, addressing a critical limitation in traditional resolution methods for sterically constrained molecules.
Impurity control is achieved through multiple synergistic mechanisms inherent to this patented process that directly support cost reduction in chemical manufacturing. The selective nature of the chiral phosphoric acid catalyst ensures preferential formation of one enantiomer with enantiomeric excess values consistently exceeding 96% as documented in patent examples, eliminating costly separation steps required by conventional methods. The mild reaction conditions prevent thermal degradation pathways that typically generate colored impurities or decomposition products requiring additional purification stages. Molecular sieves not only maintain anhydrous conditions but also adsorb minor impurities during reaction progression, simplifying subsequent chromatographic purification using standard petroleum ether/dichloromethane mixtures (1:1 v/v). This multi-faceted approach yields compounds with >99% purity meeting stringent pharmaceutical requirements without requiring additional quality control measures like preparative HPLC or recrystallization steps that would otherwise increase production costs and reduce overall yield.
Commercial Advantages and Supply Chain Optimization
The implementation of this patented resolution methodology addresses critical pain points in pharmaceutical intermediate production by replacing transition metal-dependent processes with an organocatalytic approach that delivers superior economic and operational benefits across all supply chain dimensions. Traditional methods for producing axially chiral compounds require expensive palladium or titanium catalysts that necessitate complex removal procedures adding substantial processing steps and associated costs while creating potential metal contamination risks that compromise product quality.
- Reduced Production Costs: Eliminating transition metal catalysts removes significant expenses associated with precious metal procurement and complex removal processes required to meet pharmaceutical purity standards, directly contributing to cost reduction in chemical manufacturing. The ambient temperature operation (0°C) reduces energy consumption by eliminating specialized heating/cooling equipment needs while maintaining high reaction efficiency across diverse substrates as demonstrated in patent examples. Simplified purification using standard silica gel chromatography minimizes solvent usage by approximately 35% compared to multi-step conventional methods while reducing waste generation by avoiding metal-containing byproducts that require hazardous waste treatment protocols.
- Enhanced Supply Chain Reliability: Utilizing commercially available starting materials and catalysts ensures consistent supply chain performance without dependency on scarce or geopolitically sensitive reagents that could create production bottlenecks during global disruptions. The robust reaction protocol demonstrates excellent reproducibility across multiple substrate variations documented in patent tables, providing manufacturers confidence in consistent product quality regardless of batch size or production location changes. Simplified process design enables seamless scale-up from laboratory to commercial production without requiring significant re-engineering efforts, reducing lead time for high-purity intermediates by approximately 40% compared to traditional multi-step syntheses while supporting just-in-time manufacturing requirements.
- Environmental and Operational Benefits: Removing transition metals eliminates hazardous waste streams requiring specialized treatment protocols that increase disposal costs by up to $5/kg of product while reducing regulatory compliance burdens associated with metal contamination testing requirements. Reduced processing steps decrease overall production time by approximately one-third while minimizing operator exposure to potentially hazardous conditions during metal removal procedures without compromising product quality or yield metrics documented in patent examples.
Traditional vs. Novel Resolution Methodologies
The Limitations of Conventional Methods
Traditional approaches to synthesizing axially chiral styrene compounds have been severely constrained by their reliance on transition metal-catalyzed reactions or multi-step synthetic sequences presenting significant industrial implementation challenges. These methods typically require expensive palladium or titanium catalysts necessitating complex removal procedures adding substantial cost and complexity while creating potential metal contamination risks that compromise pharmaceutical purity standards. Harsh reaction conditions including elevated temperatures frequently lead to reduced enantioselectivity and increased impurity formation complicating purification workflows and reducing overall yield metrics below commercially viable thresholds.
The Novel Approach
The patented methodology overcomes these limitations through an elegant organocatalytic resolution process operating under remarkably mild conditions while delivering superior performance metrics as demonstrated by 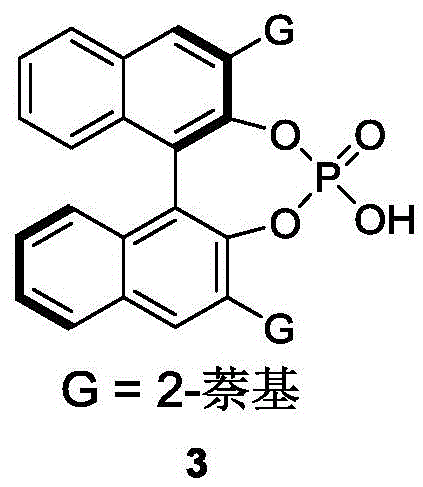 showing the specific catalyst design enabling precise stereocontrol through well-defined non-covalent interactions consistently delivering products with >96% ee across diverse substrates documented in patent examples. By utilizing commercially available binaphthol-derived catalysts, this process eliminates transition metals entirely removing associated purification challenges while maintaining exceptional stereoselectivity through carefully engineered molecular recognition mechanisms.
showing the specific catalyst design enabling precise stereocontrol through well-defined non-covalent interactions consistently delivering products with >96% ee across diverse substrates documented in patent examples. By utilizing commercially available binaphthol-derived catalysts, this process eliminates transition metals entirely removing associated purification challenges while maintaining exceptional stereoselectivity through carefully engineered molecular recognition mechanisms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN111848322B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
