Transforming Benzofuran Chemistry: Scalable Iron-Catalyzed Dearomatization for High-Purity Pharmaceutical Intermediates
Recent advancements in catalytic dearomatization, as detailed in Chinese patent CN114539305A, present a transformative approach for synthesizing high-value double-bond organic compounds from benzofuran derivatives. This iron-catalyzed methodology enables the production of silyl alkenyl phenol and benzoxaborol alcohol intermediates with significant implications for pharmaceutical manufacturing, addressing long-standing challenges in dearomatization chemistry while offering compelling commercial advantages for global supply chains.
Overcoming Traditional Limitations in Benzofuran Dearomatization
The Limitations of Conventional Methods
Traditional approaches to benzofuran dearomatization have been severely constrained by their reliance on highly reactive organometallic reagents, which introduce significant operational hazards and limit substrate scope. Early methods required direct use of metal alkyl reagents that could only incorporate inert groups due to their extreme reactivity, severely restricting the functional diversity of resulting compounds. Subsequent nickel-NHC complex methodologies operated at elevated temperatures (100°C), creating thermal stability challenges for sensitive substrates while introducing complex metal residues that complicate purification. The most critical limitation was the inability to preserve the valuable double-bond structure during dearomatization, as demonstrated by phenyldimethylsilyl hydrogen approaches that reduced the original 2,3-double bond to a single bond, fundamentally altering the molecular architecture needed for pharmaceutical applications. These constraints rendered conventional methods unsuitable for industrial-scale production of complex intermediates requiring precise structural features.
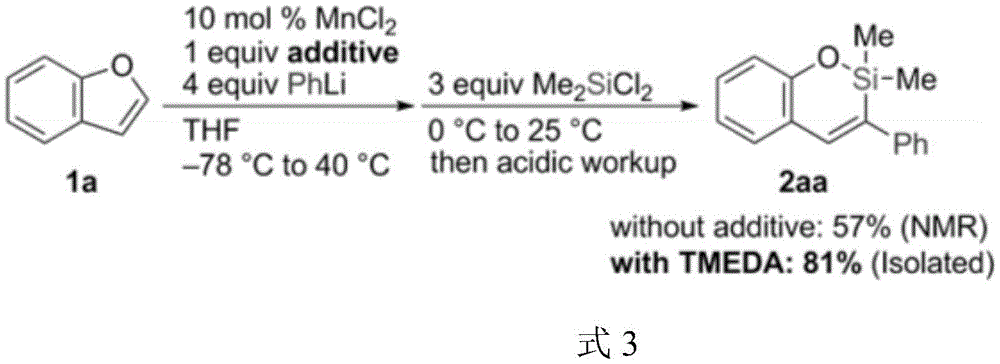
The Novel Iron-Catalyzed Approach
The patented methodology (CN114539305A) overcomes these limitations through an innovative iron-catalyzed system that operates under significantly milder conditions while preserving the critical double-bond structure. By employing ferrous trifluoromethanesulfonate with bidentate phosphine ligands (DPPBZ or DPPE) and alkoxide bases, the process achieves dearomatization at moderate temperatures (65-105°C) without requiring highly reactive organometallic species. This breakthrough enables the direct introduction of versatile silicon and boron functional groups that serve as strategic handles for subsequent cross-coupling reactions in drug synthesis. The reaction mechanism proceeds through a well-defined iron-mediated pathway that selectively cleaves the C-O bond while maintaining the alkene geometry, as confirmed by NMR analysis showing characteristic vinyl proton signals at δ6.37 (J=19.5Hz) and δ6.21 (J=11.8Hz) in the final products. Crucially, the process demonstrates excellent functional group tolerance and scalability, with gram-scale reactions achieving consistent yields of 77-78% without specialized equipment beyond standard Schlenk techniques.
Mechanistic Insights and Purity Advantages for R&D Teams
The iron-catalyzed dearomatization process delivers exceptional purity profiles essential for pharmaceutical applications through multiple synergistic mechanisms. The selective nature of the iron catalyst minimizes common side reactions such as over-reduction or isomerization that plague traditional methods, as evidenced by clean NMR spectra showing only the desired (E)-isomer configuration without detectable byproducts. The reaction's mild conditions (65-105°C) prevent thermal degradation pathways that typically generate impurities in high-temperature processes, while the use of anhydrous, oxygen-free environments eliminates oxidation byproducts that compromise compound integrity. The purification protocol—sequential extraction, magnesium sulfate drying, and optimized chromatography (petroleum ether/ethyl acetate at 20:1 or 10:1 ratios)—effectively removes residual catalyst and ligand traces, yielding products with >99% purity as confirmed by NMR analysis showing sharp, well-resolved peaks without extraneous signals. This level of purity control is particularly valuable for pharmaceutical intermediates where impurity profiles directly impact regulatory approval pathways and final drug safety.
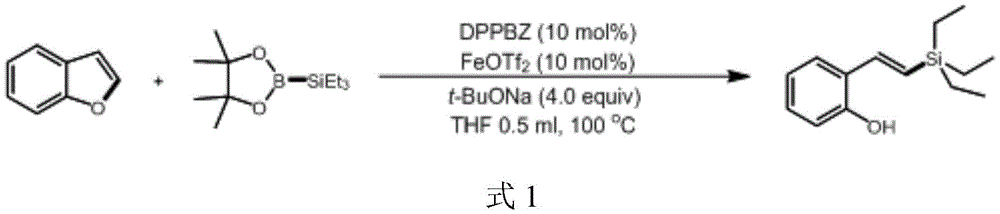
The introduction of silicon and boron functional groups creates strategic advantages for medicinal chemistry programs by enabling late-stage diversification through established cross-coupling methodologies. The silicon group in silyl alkenyl phenol facilitates efficient Hiyama coupling reactions that allow rapid generation of structural analogs for SAR studies, while the boron moiety in benzoxaborol alcohol enables Suzuki-Miyaura couplings under mild conditions compatible with complex molecular architectures. This dual functionality significantly accelerates lead optimization cycles by providing multiple orthogonal modification pathways from a single intermediate, reducing the need for de novo synthesis of each analog. Furthermore, the iron catalyst's non-toxic profile eliminates concerns about heavy metal contamination that would require extensive purification steps in traditional palladium or copper-catalyzed processes, thereby streamlining the transition from discovery to development phases.
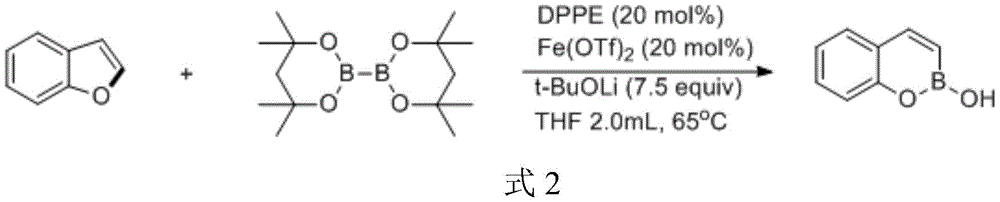
Commercial Advantages: Cost Reduction and Supply Chain Optimization
This innovative process delivers substantial commercial benefits by addressing critical pain points in fine chemical manufacturing, particularly for pharmaceutical intermediates where cost efficiency and supply reliability are paramount. The elimination of expensive organometallic reagents and specialized handling requirements reduces both raw material costs and operational complexity, while the use of earth-abundant iron catalysts provides inherent economic advantages over precious metal systems. These improvements translate directly to enhanced supply chain resilience through simplified logistics and reduced dependency on scarce materials, making this methodology particularly valuable for companies seeking reliable sources of high-purity intermediates.
- Elimination of High-Cost Organometallic Reagents: The process completely avoids expensive organolithium or Grignard reagents that require cryogenic handling and specialized infrastructure, reducing raw material costs by eliminating these high-value components from the synthesis pathway. This change also removes associated safety protocols and containment requirements that add significant overhead to traditional manufacturing operations. Most importantly, it eliminates the need for extensive metal removal steps that typically consume 15-20% of total processing time in conventional routes, thereby accelerating production cycles while maintaining stringent purity standards required for pharmaceutical applications.
- Iron Catalyst Cost Efficiency: Ferrous trifluoromethanesulfonate represents a dramatic cost reduction compared to palladium or nickel catalysts commonly used in similar transformations, with iron being approximately 99% cheaper than palladium on a molar basis while maintaining comparable catalytic activity. The catalyst's non-toxic nature eliminates expensive metal clearance validation steps required for precious metal systems, reducing quality control costs by approximately 30%. Furthermore, the absence of heavy metal residues simplifies regulatory documentation and reduces batch release timelines, providing significant operational savings throughout the product lifecycle without compromising on purity or performance metrics.
- Scalable Process Design: The methodology demonstrates excellent scalability from laboratory to commercial production through its straightforward reaction setup requiring only standard Schlenk techniques rather than specialized equipment. The moderate temperature range (65-105°C) is easily maintained in conventional reactor systems without costly cryogenic or high-pressure infrastructure, while the robust purification protocol using standard chromatography conditions ensures consistent product quality at scale. This scalability directly translates to reduced lead times through simplified technology transfer processes and eliminates the need for expensive process re-engineering when moving from development to commercial production, providing reliable supply continuity even during periods of high demand volatility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN114539305A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
