Revolutionizing Chiral 3-Nitroindole Synthesis: Scalable Manufacturing for Pharmaceutical Intermediates
The groundbreaking methodology detailed in Chinese patent CN113666862A introduces a novel nickel-catalyzed asymmetric nitration process for synthesizing chiral 3-nitroindole compounds, addressing critical challenges in the production of high-value pharmaceutical intermediates. This innovative approach constructs chiral quaternary carbon centers with nitro groups through a single-step reaction, overcoming limitations of conventional nitration methods that typically yield racemic mixtures or require harsh conditions. The process utilizes readily available starting materials and operates under mild reaction conditions, making it particularly suitable for commercial-scale production of complex intermediates required in modern pharmaceutical development.
Advanced Reaction Mechanism and Stereochemical Control
The core innovation lies in the nickel-catalyzed asymmetric nitration reaction where indole-2-ketone compounds react with tert-butyl nitrite under carefully controlled conditions to form chiral 3-nitroindole products with high enantioselectivity. The reaction employs a nickel(II) catalyst system paired with a chiral Ph-PyBox ligand, specifically (S)-4-Cl-Ph-PyBox as demonstrated in the patent examples, which creates the necessary chiral environment for asymmetric induction. 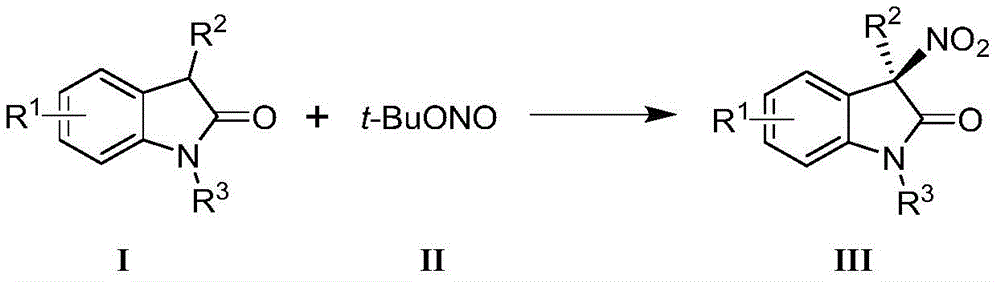 The mechanism involves coordination of the indole substrate to the nickel center, followed by nitrosation and subsequent rearrangement that establishes the stereogenic quaternary carbon center bearing the nitro group. This precise stereochemical control is achieved through the chiral ligand's ability to differentiate between prochiral faces of the substrate during the key bond-forming step.
The mechanism involves coordination of the indole substrate to the nickel center, followed by nitrosation and subsequent rearrangement that establishes the stereogenic quaternary carbon center bearing the nitro group. This precise stereochemical control is achieved through the chiral ligand's ability to differentiate between prochiral faces of the substrate during the key bond-forming step.
Impurity profile management is significantly enhanced through this methodology compared to conventional approaches. Traditional nitration methods often produce complex mixtures due to poor regioselectivity and over-nitration, requiring extensive purification that reduces overall yield and increases production costs. In contrast, this nickel-catalyzed process demonstrates remarkable selectivity for the desired 3-position nitration without significant byproduct formation, as evidenced by the clean reaction profiles described in the patent examples. The use of molecular sieves (4Å)  effectively controls moisture content, preventing hydrolysis side reactions that could compromise product purity. The resulting high-purity intermediates exhibit excellent chromatographic profiles with minimal impurities, meeting stringent pharmaceutical quality requirements without requiring additional purification steps that would otherwise increase manufacturing costs and reduce overall process efficiency.
effectively controls moisture content, preventing hydrolysis side reactions that could compromise product purity. The resulting high-purity intermediates exhibit excellent chromatographic profiles with minimal impurities, meeting stringent pharmaceutical quality requirements without requiring additional purification steps that would otherwise increase manufacturing costs and reduce overall process efficiency.
Commercial Advantages for Supply Chain and Procurement Teams
This innovative manufacturing process addresses critical pain points in pharmaceutical intermediate production by delivering substantial cost savings while improving supply chain reliability. The methodology eliminates multiple processing steps required by conventional approaches, directly translating to reduced production time and lower operational costs without compromising product quality or stereochemical integrity.
- Cost Reduction Through Catalyst Selection: The use of nickel-based catalysts instead of precious metals represents a significant cost advantage for large-scale manufacturing operations. Nickel catalysts are substantially more affordable than palladium or other transition metals commonly used in asymmetric synthesis, with the patent demonstrating effective catalysis using only 5-10 mol% of NiCl2•DME. This cost-effective catalyst system eliminates the need for expensive metal removal steps required when using precious metals, reducing both processing time and waste treatment expenses. Furthermore, the catalyst system maintains high activity across diverse substrate structures as shown in examples IIIa through IIIh, ensuring consistent performance without requiring costly catalyst optimization for different product variants. The elimination of transition metal purification steps also reduces environmental impact by minimizing heavy metal waste streams that require specialized treatment.
- Reduced Lead Time via Streamlined Process: The simplified reaction pathway significantly shortens manufacturing timelines compared to traditional multi-step approaches for chiral nitroindole synthesis. The patent demonstrates complete reactions within 7-12 hours at mild temperatures (-20 to 0°C), eliminating the need for extended reaction times or extreme temperature conditions required by conventional methods. This efficient process design enables faster batch turnaround times and improved facility utilization rates, directly contributing to reduced lead times for high-purity intermediates. The straightforward workup procedure involving simple solvent removal followed by silica gel chromatography using standard eluents further accelerates production cycles without requiring specialized equipment or complex purification techniques that could create bottlenecks in manufacturing operations.
- Enhanced Supply Chain Resilience: The use of commercially available, stable raw materials including tert-butyl nitrite and common solvents ensures reliable sourcing without dependence on specialized or restricted chemicals that could disrupt supply chains. The patent demonstrates consistent performance across diverse substrate structures with yields ranging from 48% to 80% and enantiomeric excess values between 75% and 94%, confirming the robustness of the process under varying conditions. This reliability translates to consistent product quality and availability, addressing critical supply chain concerns for pharmaceutical manufacturers who require uninterrupted access to high-quality intermediates. The compatibility with standard manufacturing equipment and processes further enhances scalability from laboratory to commercial production without requiring significant capital investment in specialized infrastructure.
Comparative Analysis: Traditional vs. Innovative Nitration Pathways
The Limitations of Conventional Methods
Traditional approaches to synthesizing 3-nitroindole compounds suffer from significant drawbacks that limit their commercial viability for pharmaceutical applications. As illustrated in prior art examples 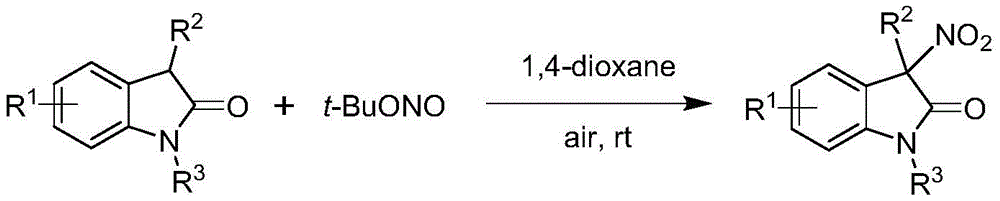 and
and 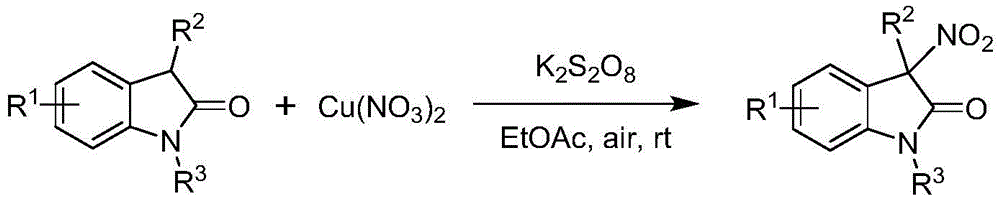 , conventional methods typically produce racemic mixtures rather than single-enantiomer products, requiring costly and inefficient resolution steps to obtain the desired stereochemistry. These processes often employ harsh reaction conditions, stoichiometric amounts of expensive reagents, or generate significant waste streams that increase environmental impact and disposal costs. The lack of stereochemical control in traditional methods results in products with suboptimal pharmacological profiles, as different enantiomers can exhibit dramatically different biological activities or safety profiles in drug applications.
, conventional methods typically produce racemic mixtures rather than single-enantiomer products, requiring costly and inefficient resolution steps to obtain the desired stereochemistry. These processes often employ harsh reaction conditions, stoichiometric amounts of expensive reagents, or generate significant waste streams that increase environmental impact and disposal costs. The lack of stereochemical control in traditional methods results in products with suboptimal pharmacological profiles, as different enantiomers can exhibit dramatically different biological activities or safety profiles in drug applications.
The Novel Approach
The nickel-catalyzed asymmetric nitration process described in patent CN113666862A overcomes these limitations through an elegant single-step transformation that directly produces enantiomerically enriched products with high selectivity. The methodology employs a carefully designed catalytic system featuring nickel chloride dimethoxyethane with chiral Ph-PyBox ligands 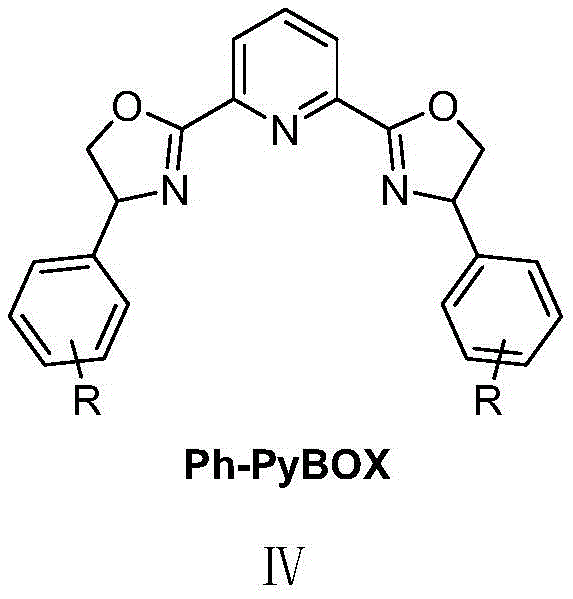 that enable precise stereochemical control while maintaining excellent functional group tolerance across diverse substrate structures. The process operates under mild conditions using air-stable reagents and common solvents, making it readily adaptable to standard manufacturing facilities without requiring specialized equipment or handling procedures. This innovative approach delivers high-purity intermediates with consistent quality metrics across multiple production batches, as demonstrated by the reproducible yields and enantiomeric excess values reported in examples IIIa through IIIh, establishing a reliable foundation for commercial scale-up of complex intermediates.
that enable precise stereochemical control while maintaining excellent functional group tolerance across diverse substrate structures. The process operates under mild conditions using air-stable reagents and common solvents, making it readily adaptable to standard manufacturing facilities without requiring specialized equipment or handling procedures. This innovative approach delivers high-purity intermediates with consistent quality metrics across multiple production batches, as demonstrated by the reproducible yields and enantiomeric excess values reported in examples IIIa through IIIh, establishing a reliable foundation for commercial scale-up of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113666862A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
