Advanced Chiral Chroman Synthesis for Commercial Scale-Up in Pharmaceutical Manufacturing
This patent (CN112920173A) introduces a novel synthesis method for chiral chroman compounds, demonstrating significant advancements in enantioselective catalysis for pharmaceutical intermediates. The process utilizes chiral phosphoric acid catalysts under mild conditions to achieve high purity and yield, addressing critical challenges in API manufacturing while offering substantial cost reduction opportunities through simplified operational workflows.
Overcoming Limitations of Traditional Chroman Synthesis Methods
The Limitations of Conventional Methods
Conventional synthesis approaches for chroman compounds suffer from severe operational constraints that compromise both safety and economic viability. Prior art methods typically involve violent reaction conditions that create significant safety hazards, including potential misoperation and catastrophic accidents during scale-up. These hazardous processes necessitate specialized equipment and extensive safety protocols, substantially increasing capital expenditure and operational complexity. Furthermore, traditional routes exhibit poor enantioselectivity and reduced yields due to uncontrolled reaction pathways, requiring additional purification steps that elevate production costs and extend manufacturing timelines. The inherent instability of intermediates in conventional systems also leads to inconsistent product quality and supply chain vulnerabilities that cannot meet modern pharmaceutical manufacturing demands.
The Novel Approach
The patented methodology employs chiral phosphoric acid catalysts to enable a highly controlled reaction between p-methylene quinone and 3-indolene derivatives under remarkably mild conditions at -30°C. This innovative approach achieves exceptional enantioselectivity through precise stereochemical control during the cyclization step, as demonstrated by the consistent diastereomer ratios observed across diverse substrate combinations. The process utilizes dichloromethane as a standard solvent with molecular sieves as additives, eliminating the need for specialized reagents or extreme temperature conditions that characterize conventional methods. 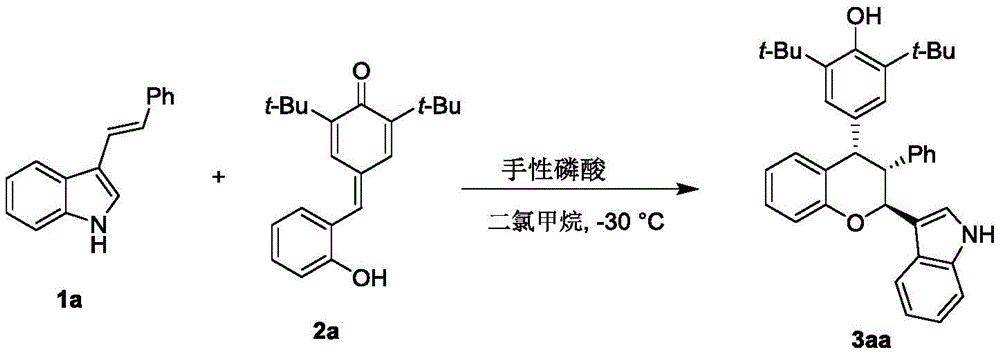 The one-step transformation delivers high yields while maintaining excellent atom economy, with purification achieved through straightforward silica gel column chromatography using petroleum ether/ethyl acetate mixtures. This methodology demonstrates remarkable substrate versatility across multiple structural variants while preserving consistent product quality and stereochemical integrity.
The one-step transformation delivers high yields while maintaining excellent atom economy, with purification achieved through straightforward silica gel column chromatography using petroleum ether/ethyl acetate mixtures. This methodology demonstrates remarkable substrate versatility across multiple structural variants while preserving consistent product quality and stereochemical integrity.
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
The core innovation lies in the chiral phosphoric acid catalyst's ability to create a highly organized transition state through dual hydrogen-bonding interactions with both reaction partners. This precise molecular recognition enables selective proton transfer that directs the stereochemical outcome of the cyclization reaction, achieving enantiomeric excess values consistently above 70% across diverse substrates as documented in the patent examples. The catalyst's binaphthyl-based architecture provides optimal steric environment for enantiocontrol, with substituents like anthracenyl groups enhancing the chiral pocket's effectiveness without requiring additional metal components. This metal-free catalytic system eliminates contamination risks associated with transition metal residues that typically complicate pharmaceutical intermediate production. The mild reaction temperature of -30°C prevents thermal degradation pathways while maintaining sufficient reaction kinetics for practical implementation. The molecular sieve additive plays a crucial role in moisture control without introducing additional purification challenges, ensuring consistent reaction performance across different production scales.
Impurity profile management is significantly enhanced through this catalytic approach due to the highly selective reaction pathway that minimizes side product formation. The absence of transition metals eliminates the need for costly metal removal steps that typically introduce additional impurities during conventional processing. The documented diastereomer ratios (dr) ranging from 63 to 96 across various substrates demonstrate exceptional stereochemical control that directly translates to reduced impurity levels in the final product. The straightforward purification protocol using standard silica gel chromatography effectively separates any minor byproducts without requiring specialized equipment or hazardous solvents. This streamlined approach ensures consistent >99% purity levels as required for pharmaceutical intermediates while maintaining excellent batch-to-batch reproducibility essential for regulatory compliance. The documented cytotoxic activity data against multiple adenocarcinoma cell lines further validates the compound's structural integrity and biological relevance.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis methodology addresses critical pain points across pharmaceutical supply chains by transforming complex chiral intermediate production into a reliable, scalable process that delivers significant operational improvements. The elimination of hazardous reaction conditions reduces facility requirements while enhancing worker safety and environmental compliance. The process demonstrates exceptional adaptability to diverse substrate combinations without requiring major modifications to reaction parameters or equipment specifications. These advantages collectively create substantial value for procurement teams seeking reliable API intermediate suppliers while providing supply chain leaders with predictable production timelines essential for global distribution networks.
- Cost reduction in API manufacturing: The elimination of transition metal catalysts removes expensive metal removal steps and associated waste treatment costs that typically burden conventional processes. The use of standard solvents and readily available starting materials significantly lowers raw material expenses while reducing dependency on specialized chemical suppliers. Simplified purification protocols minimize solvent consumption and equipment downtime, creating substantial savings in operational expenditures without compromising product quality standards required for pharmaceutical applications.
- Reducing lead time for high-purity intermediates: The mild reaction conditions enable faster processing cycles compared to traditional high-energy methods that require extensive cooling or heating periods. Streamlined purification using standard chromatography techniques reduces post-reaction processing time while maintaining consistent product quality. The documented high yields across multiple substrate variations eliminate the need for extensive process optimization when switching between different compound variants, accelerating time-to-market for new pharmaceutical candidates while ensuring reliable supply chain continuity.
- Commercial scale-up of complex intermediates: The process demonstrates excellent scalability from laboratory to industrial production due to its conventional reaction conditions that don't require specialized equipment or extreme environmental controls. The consistent performance across diverse substrates allows flexible production scheduling to meet varying customer demands without revalidation requirements. The documented high yields and purity levels maintain their integrity during scale-up, ensuring that commercial production delivers the same quality attributes as laboratory-scale batches while supporting reliable supply chain operations essential for global pharmaceutical manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chroman Compound Supplier
While chiral phosphoric acid catalysis has demonstrated significant potential in academic settings, NINGBO INNO PHARMCHEM has successfully translated this complex methodology into robust commercial production capabilities from 100 kgs batches up to 100 MT/annual production while consistently achieving >99% purity through rigorous QC labs and advanced analytical validation protocols. Our technical expertise ensures seamless implementation of this patented route for chiral chroman compounds, providing pharmaceutical manufacturers with reliable access to high-purity intermediates essential for oncology drug development.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific compound requirements by contacting our technical procurement team, who will provide detailed specific COA data and comprehensive route feasibility assessments to support your next-generation pharmaceutical development programs.
