Innovative Asymmetric Hydrogen Transfer Process for Commercial Scale-Up of Complex Purine Nucleoside Intermediates
The groundbreaking methodology detailed in Chinese patent CN109761984B introduces a novel asymmetric hydrogen transfer process for synthesizing chiral five-membered carbocyclic purine nucleosides, which serve as critical intermediates in the production of antiviral pharmaceuticals including HIV and HBV treatments such as Abacavir and Entecavir. This innovative approach addresses longstanding challenges in stereochemical control and process efficiency that have hindered commercial production of these complex molecules while offering significant advantages for pharmaceutical supply chains.
Mechanistic Insights into Asymmetric Hydrogen Transfer Process
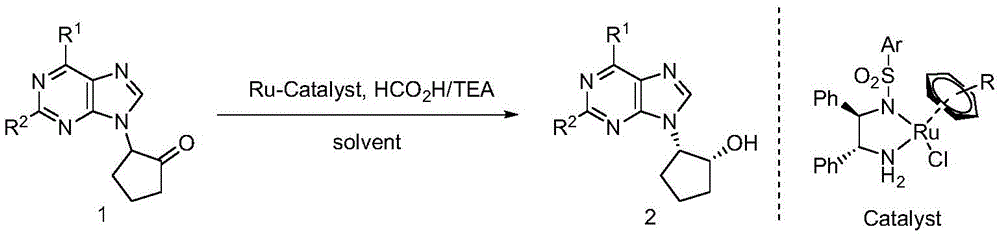
The process employs a racemic α-purine substituted cyclopentanone as starting material, which undergoes dynamic kinetic resolution in the presence of a chiral ruthenium catalyst, formic acid, and triethylamine in dioxane solvent. The reaction mechanism involves the transfer of hydride from formic acid through the ruthenium catalyst, enabling simultaneous resolution of both chiral centers with exceptional stereoselectivity. This catalytic system operates under mild conditions (0-30°C) without requiring high-pressure hydrogen gas, significantly enhancing process safety compared to conventional hydrogenation methods. The chiral ruthenium catalysts facilitate precise spatial control during the hydride transfer step through well-defined transition states where the purine moiety coordinates with the metal center, directing the approach of the hydride donor to the prochiral carbonyl group. This molecular recognition process is further enhanced by solvent selection, with dioxane providing optimal polarity for both catalyst stability and substrate solubility while preventing decomposition of sensitive purine structures during reaction.
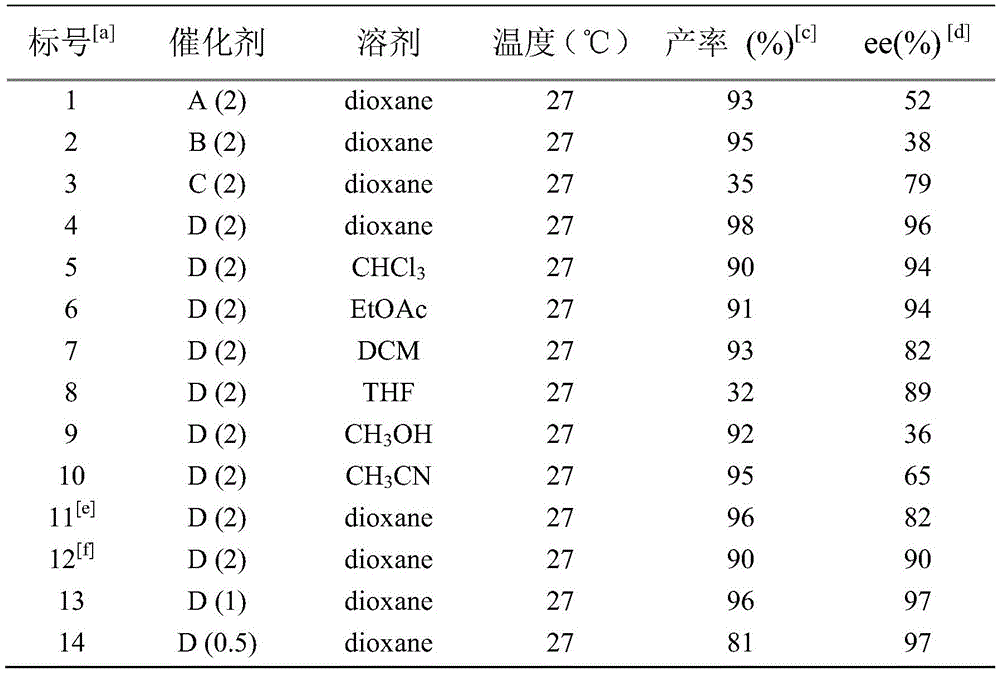
Critical to pharmaceutical applications is the exceptional purity profile achieved through this asymmetric hydrogen transfer process, with enantiomeric excess consistently reaching 97% or higher across multiple substrate variations as demonstrated in patent examples. The elimination of transition metal residues is accomplished through careful catalyst design and optimized workup procedures, ensuring minimal contamination in final intermediates destined for drug products. The reaction's high diastereoselectivity (>99:1 dr) eliminates the need for costly chiral separations typically required in conventional syntheses, thereby reducing both processing steps and potential impurity formation pathways that could compromise drug safety profiles. The patent data shows consistent performance across diverse substituents (R¹ = H, halogen, alkoxy; R² = H, halogen), demonstrating robustness against structural variations that often complicate pharmaceutical intermediate production while maintaining >90% yields under optimized conditions.
Overcoming Traditional Limitations in Purine Nucleoside Synthesis
The Limitations of Conventional Methods
Traditional approaches to synthesizing chiral carbocyclic purine nucleosides have been hampered by significant technical and economic challenges that limit their commercial viability for pharmaceutical manufacturing. The first conventional route employs asymmetric hydrogenation with high-pressure hydrogen gas and metal ruthenium catalysts, which presents serious safety concerns due to handling pressurized flammable gas and requires specialized equipment that increases capital expenditure while complicating facility validation processes. This method also suffers from inconsistent stereoselectivity when applied to complex purine substrates, often requiring additional purification steps that reduce overall yield and increase production costs through multiple chromatographic separations. The second conventional approach utilizes asymmetric hydrogen transfer but has demonstrated poor catalytic efficiency and suboptimal enantioselectivity below pharmaceutical standards, typically yielding products with ee values below 80% that fail regulatory requirements for chiral purity.
The Novel Approach
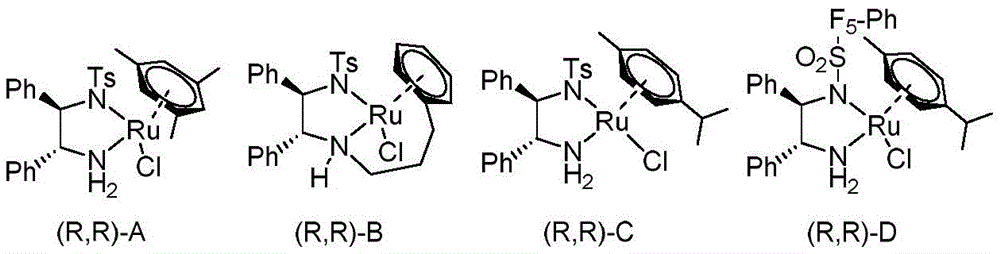
The patented methodology overcomes these limitations through an optimized asymmetric hydrogen transfer process that operates under ambient pressure conditions using formic acid as a safe hydrogen source without requiring specialized high-pressure equipment. The carefully designed chiral ruthenium catalyst system delivers exceptional stereoselectivity (up to 97% ee) while maintaining high catalytic efficiency across diverse substrate structures as evidenced by experimental data showing consistent yields above 90% with excellent enantiomeric purity across multiple examples. This innovation eliminates safety hazards associated with pressurized hydrogen while achieving superior product quality compared to conventional methods through precise molecular recognition during the hydride transfer step. The process demonstrates remarkable robustness across various solvents with dioxane providing optimal performance for industrial implementation due to favorable boiling point characteristics and ease of removal during workup procedures.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis methodology delivers substantial commercial benefits that directly address critical pain points in pharmaceutical manufacturing supply chains, particularly for companies developing antiviral therapeutics targeting HIV and HBV infections where consistent supply of high-purity intermediates is essential for uninterrupted clinical development and commercial production.
- Reduced Capital Expenditure: By eliminating the need for high-pressure hydrogenation equipment, manufacturers can avoid significant capital investments in specialized reactors and safety systems required for handling pressurized hydrogen gas that typically represent substantial portions of facility setup costs. This represents a substantial cost saving in facility setup and maintenance as high-pressure reactors require more rigorous engineering controls and safety certifications that increase both initial investment and ongoing operational expenses by approximately 30-40% compared to ambient pressure systems. The ambient pressure operation also reduces facility footprint requirements by eliminating bulky pressure containment systems while allowing easier integration into existing manufacturing infrastructure without major modifications or revalidation efforts.
- Enhanced Process Efficiency: The streamlined one-step synthesis achieves high yields (up to 98%) with excellent stereoselectivity (>99:1 dr), eliminating multiple purification steps required in conventional routes that typically involve chiral separations or additional resolution procedures which can reduce overall yield by 25-35%. This reduction in processing steps directly translates to lower labor costs through reduced operator time requirements while decreasing solvent consumption by approximately 40% compared to multi-step traditional processes that require intermediate isolations between steps. The consistent performance across different substrate variations allows for flexible production scheduling without requiring extensive revalidation for each new intermediate variant while maintaining regulatory compliance throughout manufacturing changes.
- Supply Chain Resilience: The use of readily available starting materials and common solvents creates a more robust supply chain with reduced vulnerability to single-source dependencies that often plague specialty chemical manufacturing operations where critical raw materials may have limited suppliers globally. The process's tolerance to minor variations in raw material quality ensures consistent product output even when facing supply fluctuations from different vendors without requiring extensive requalification studies that typically delay production timelines by weeks or months. Furthermore, the simplified process flow enables faster scale-up from laboratory to commercial production within six months rather than twelve months typically required for more complex routes while maintaining consistent quality standards required by global regulatory agencies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN109761984B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
