Advanced Asymmetric Catalysis for Commercial Scale-Up of Chiral Purine Nucleosides
Patent CN108912123B introduces a groundbreaking asymmetric [3+3] cyclization methodology for synthesizing chiral six-membered carbocyclic purine nucleosides, addressing critical challenges in pharmaceutical intermediate production. This innovative approach eliminates the need for stoichiometric chiral sources and multi-step sequences that characterize conventional synthesis routes, offering a streamlined pathway to high-value nucleoside analogs with exceptional stereoselectivity. The process achieves up to 89% yield with excellent diastereoselectivity (up to 9:1 dr) and enantioselectivity (up to 99% ee), providing significant advantages for pharmaceutical manufacturers seeking reliable API intermediate suppliers with cost reduction in chemical manufacturing capabilities.
Overcoming Traditional Synthesis Limitations in Nucleoside Manufacturing
The Limitations of Conventional Methods
Traditional approaches to constructing chiral six-membered carbocyclic nucleosides require carefully designed stereoconfigured starting materials obtained through multi-step sequences, followed by coupling with purine or pyrimidine bases via nucleophilic substitution, epoxide ring-opening, or Mitsunobu reactions. These conventional methods necessitate stoichiometric chiral sources that are difficult to prepare and costly to acquire, significantly increasing production expenses while introducing multiple purification steps that compromise overall yield. The multi-step nature of these processes creates substantial bottlenecks in supply chain continuity, as each transformation requires separate optimization, validation, and quality control procedures that extend lead times and increase vulnerability to process failures. Furthermore, the reliance on pre-formed chiral building blocks limits structural diversity and complicates the synthesis of novel analogs needed for drug discovery pipelines, creating significant barriers to innovation in nucleoside-based therapeutics development.
The Novel Approach
The patented methodology employs α-purine substituted acetone and β,γ-unsaturated ketoester as readily available achiral starting materials, catalyzed by chiral thiourea compounds under mild conditions without inert gas protection. This direct asymmetric [3+3] cyclization strategy eliminates the need for pre-formed chiral intermediates by creating the stereocenters during the key bond-forming step, dramatically simplifying the synthetic sequence while maintaining exceptional stereocontrol. The reaction proceeds efficiently at room temperature in common solvents like 1,2-dichloroethane, with catalyst loadings as low as 5 mol% achieving meaningful conversion while higher loadings (20 mol%) deliver optimal yields up to 89%. The process demonstrates remarkable substrate scope across diverse R-group variations including halogenated aromatics, heterocycles, and functionalized phenyl groups, enabling production of structurally diverse nucleoside analogs from a single platform technology. This streamlined approach represents a paradigm shift from traditional multi-step syntheses toward a more efficient commercial scale-up of complex intermediates with reduced environmental impact.
![Asymmetric [3+3] cyclization reaction scheme showing synthesis of chiral six-membered carbocyclic purine nucleosides from α-purine substituted acetone and β,γ-unsaturated ketoester](/insights/img/chiral-six-membered-carbocyclic-purine-nucleoside-asymmetric-cyclization-pharmaceutical-cost-reduction-20260228045620-01.png)
Molecular Mechanism and Stereoselectivity Control in Asymmetric Catalysis
The reaction mechanism centers on the dual hydrogen-bonding activation capability of chiral thiourea catalysts, which simultaneously coordinate both the α-purine acetone enol and the β,γ-unsaturated ketoester through a well-defined transition state geometry. This precise molecular recognition creates a chiral environment that directs the stereochemical outcome of the cyclization event, with the catalyst's cyclohexyl backbone and trifluoromethylphenyl groups providing optimal steric differentiation between prochiral faces. The thiourea moiety's hydrogen bonding network activates the electrophile while organizing the nucleophile in a specific orientation that favors formation of the desired stereoisomer. This molecular-level control extends across diverse substrate combinations as demonstrated by consistent high diastereoselectivity (5:1 to 9:1 dr) and enantioselectivity (92-99% ee) across multiple structural variants, indicating the robustness of the catalytic system.
Impurity profile management is inherently addressed through the reaction's high stereoselectivity and clean conversion characteristics. The absence of transition metals eliminates concerns about heavy metal contamination that would require extensive purification steps in traditional catalytic systems. The mild reaction conditions (room temperature, no inert atmosphere) prevent thermal degradation pathways that commonly generate impurities in conventional syntheses requiring elevated temperatures or harsh reagents. The well-defined reaction pathway minimizes side products through precise substrate orientation within the catalyst's chiral pocket, resulting in cleaner crude reaction mixtures that simplify downstream processing. This inherent selectivity translates directly to higher purity intermediates with reduced need for costly purification steps, addressing critical quality requirements for pharmaceutical applications while supporting cost reduction in chemical manufacturing through fewer unit operations.
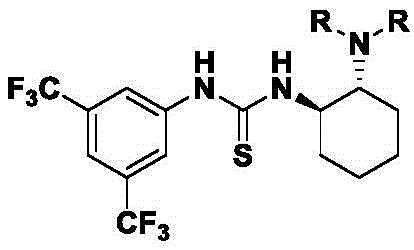
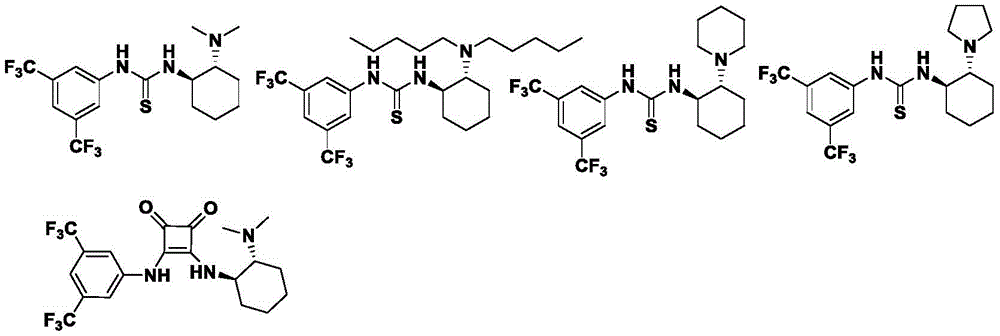
Commercial Advantages for Pharmaceutical Supply Chain Optimization
This innovative catalytic process directly addresses three critical pain points in pharmaceutical manufacturing supply chains: excessive production costs from multi-step syntheses, extended lead times from complex purification requirements, and supply continuity risks associated with specialized chiral building blocks. By converting readily available achiral starting materials into high-value chiral intermediates through a single catalytic transformation, the methodology creates significant opportunities for cost reduction in chemical manufacturing while enhancing supply chain resilience. The elimination of multiple synthetic steps reduces both capital equipment requirements and operational complexity, creating a more robust manufacturing platform that can better withstand market fluctuations and raw material shortages.
- Reduced equipment investment and operational costs: The simplified single-step process eliminates the need for specialized equipment required in traditional multi-step syntheses, including cryogenic reactors for low-temperature transformations and dedicated metal removal systems. This capital expenditure reduction translates directly to lower production costs per kilogram while improving facility utilization rates through faster campaign turnover. The room temperature operation avoids energy-intensive heating/cooling cycles that represent significant operational expenses in conventional processes, while the absence of inert atmosphere requirements simplifies reactor design and maintenance protocols. These combined factors create substantial cost reduction in chemical manufacturing without compromising product quality or yield consistency.
- Accelerated time-to-market for new therapeutics: The streamlined synthetic route reduces manufacturing lead time by eliminating multiple intermediate isolation and purification steps that typically add weeks to production schedules. This acceleration is particularly valuable during clinical development phases where rapid access to high-purity intermediates can significantly shorten drug development timelines. The robustness of the catalytic system across diverse substrate combinations enables faster route scouting and optimization for new analogs, allowing pharmaceutical companies to respond more rapidly to evolving clinical data or competitive pressures. The simplified process also reduces validation complexity for regulatory submissions, further compressing development timelines while maintaining regulatory compliance.
- Enhanced supply chain continuity and sustainability: By utilizing commodity chemicals as starting materials instead of specialized chiral building blocks with limited suppliers, this methodology creates a more resilient supply chain less vulnerable to single-source dependencies. The elimination of transition metals removes concerns about catalyst supply constraints and associated quality control challenges related to metal residues in final products. The reduced number of synthetic steps decreases solvent consumption and waste generation per kilogram of product, lowering environmental impact while reducing disposal costs associated with hazardous waste streams. This sustainable manufacturing approach aligns with growing regulatory emphasis on green chemistry principles while supporting corporate ESG initiatives through reduced carbon footprint per production batch.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN108912123B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
