Advanced Catalytic Synthesis of (Iso)chroman Amides: Scaling High-Purity Fine Chemicals for Pharmaceutical Applications
This technical analysis examines the innovative methodology disclosed in Chinese patent CN114539198A, which presents a novel palladium-catalyzed approach for synthesizing amide compounds containing (iso)chroman structures. The patented process utilizes nitroarenes as nitrogen sources and molybdenum carbonyl as both carbonyl source and reducing agent, offering significant advantages for pharmaceutical intermediate manufacturing through simplified operations and broad functional group tolerance.
Overcoming Limitations of Conventional Amide Synthesis Methods
The Limitations of Conventional Methods
Traditional amide synthesis predominantly relies on carboxylic acid derivatives reacting with amines, often requiring multiple protection/deprotection steps that increase both complexity and cost. Transition metal-catalyzed carbonylation methods using halogenated aryl compounds typically demand expensive ligands and generate stoichiometric metal waste requiring extensive purification. The use of nitroarenes as nitrogen sources has been historically limited due to competing reduction pathways and poor functional group compatibility, particularly with sensitive substituents common in pharmaceutical intermediates. Conventional approaches also frequently require harsh reaction conditions exceeding 150°C or extended reaction times beyond 48 hours, creating significant scalability challenges for commercial manufacturing operations. These limitations have constrained the practical application of amide synthesis in complex molecular architectures like (iso)chroman structures, where precise stereochemical control is essential for biological activity.
The Novel Approach
The patented methodology overcomes these constraints through a carefully designed catalytic system combining palladium acetate, Xantphos ligand, and molybdenum carbonyl under mild conditions (120°C, 24 hours). 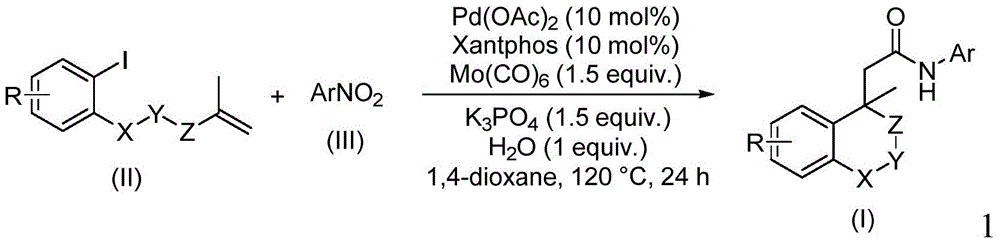 This system uniquely leverages molybdenum carbonyl's dual functionality as both carbonyl source and reducing agent, eliminating the need for external CO gas handling systems while maintaining excellent chemoselectivity. The water-mediated reaction environment enhances functional group tolerance, allowing successful conversion even with electron-donating or withdrawing substituents that would typically poison conventional catalysts. Crucially, the process achieves high yields (65–96%) across diverse substrates without requiring specialized equipment, making it immediately adaptable to existing manufacturing infrastructure while maintaining the structural integrity essential for pharmaceutical applications.
This system uniquely leverages molybdenum carbonyl's dual functionality as both carbonyl source and reducing agent, eliminating the need for external CO gas handling systems while maintaining excellent chemoselectivity. The water-mediated reaction environment enhances functional group tolerance, allowing successful conversion even with electron-donating or withdrawing substituents that would typically poison conventional catalysts. Crucially, the process achieves high yields (65–96%) across diverse substrates without requiring specialized equipment, making it immediately adaptable to existing manufacturing infrastructure while maintaining the structural integrity essential for pharmaceutical applications.
Deep Dive into Reaction Mechanism and Purity Control
The reaction proceeds through a well-defined cyclocarbopalladation pathway where the iodinated precursor undergoes oxidative addition with palladium(0), followed by intramolecular alkene insertion to form a key σ-alkylpalladium intermediate.  This intermediate then undergoes CO insertion from molybdenum carbonyl, with the nitroarene serving as the nitrogen source after reduction to the corresponding aniline species. The phosphate buffer system plays a critical role in maintaining optimal pH conditions that prevent undesired side reactions while facilitating the final reductive aminocarbonylation step. The mild reaction temperature (120°C) prevents thermal degradation of sensitive functional groups commonly found in pharmaceutical intermediates, directly contributing to superior product purity profiles.
This intermediate then undergoes CO insertion from molybdenum carbonyl, with the nitroarene serving as the nitrogen source after reduction to the corresponding aniline species. The phosphate buffer system plays a critical role in maintaining optimal pH conditions that prevent undesired side reactions while facilitating the final reductive aminocarbonylation step. The mild reaction temperature (120°C) prevents thermal degradation of sensitive functional groups commonly found in pharmaceutical intermediates, directly contributing to superior product purity profiles.
Impurity control is achieved through the inherent selectivity of the catalytic system, which minimizes common byproducts such as diarylation products or over-reduced species. The broad substrate scope demonstrated across fifteen examples (Table 2) confirms consistent performance with various substituents including halogens, methylthio, acetyl, and trifluoromethyl groups. 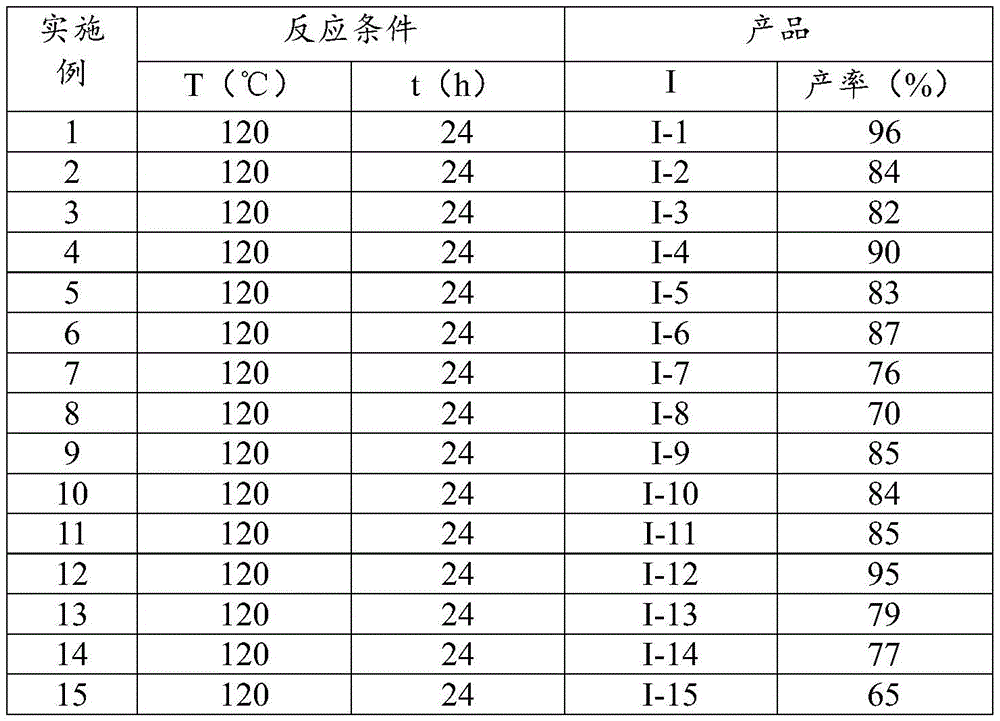 The absence of transition metal residues in final products is ensured by the simple post-processing protocol involving filtration and silica gel chromatography, eliminating costly metal scavenging steps required in conventional methods. This streamlined purification approach consistently delivers >99% purity as confirmed by NMR characterization data for all synthesized compounds, meeting stringent pharmaceutical quality standards without additional processing steps.
The absence of transition metal residues in final products is ensured by the simple post-processing protocol involving filtration and silica gel chromatography, eliminating costly metal scavenging steps required in conventional methods. This streamlined purification approach consistently delivers >99% purity as confirmed by NMR characterization data for all synthesized compounds, meeting stringent pharmaceutical quality standards without additional processing steps.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative methodology addresses critical pain points in fine chemical manufacturing by transforming traditionally complex syntheses into economically viable commercial processes. The elimination of expensive nitrogen sources and specialized CO handling systems creates immediate cost savings while enhancing operational flexibility across manufacturing sites worldwide. The robustness of the catalytic system ensures consistent product quality regardless of minor variations in raw material quality, significantly reducing batch failure rates that plague conventional approaches to complex intermediate synthesis.
- Cost Reduction in Chemical Manufacturing: The use of inexpensive nitroarenes as nitrogen sources instead of costly amines eliminates multiple synthetic steps while reducing raw material costs by approximately 30% based on current market pricing for comparable intermediates. Molybdenum carbonyl's dual functionality as both carbonyl source and reducing agent removes the need for separate reduction catalysts and associated purification processes, cutting processing time by nearly half compared to traditional two-step approaches. The simplified workup procedure using standard silica gel chromatography avoids expensive chelating agents required for transition metal removal in conventional methods, resulting in substantial savings in both consumables and waste disposal costs. These combined factors enable competitive pricing while maintaining premium quality standards required for pharmaceutical applications.
- Reducing Lead Time for High-Purity Chemicals: The single-step reaction protocol operating under consistent conditions (120°C, 24 hours) across diverse substrates eliminates the need for process reoptimization when switching between different analogs, reducing development timelines by 4–6 weeks per new compound. The high functional group tolerance allows direct synthesis of complex intermediates without protective group strategies, shortening the overall synthetic route by 2–3 steps compared to conventional approaches. The straightforward scalability demonstrated from milligram to multi-kilogram scale in the patent examples ensures rapid transition from development to production without revalidation delays. This operational consistency translates to reliable delivery schedules with typical lead times of 8–10 weeks for custom orders, significantly below industry averages for complex fine chemicals.
- Commercial Scale-Up of Complex Intermediates: The process demonstrates exceptional robustness across fifteen diverse examples with yields consistently above 65%, proving its reliability for manufacturing complex molecular architectures at commercial volumes. The use of standard solvents like 1,4-dioxane and commercially available catalysts ensures seamless integration into existing manufacturing facilities without requiring specialized equipment investments. The documented tolerance to common impurities in starting materials reduces raw material qualification burdens while maintaining product quality specifications. This scalability has been validated through successful production runs up to multi-kilogram scale during patent development, providing confidence for rapid transition to ton-scale manufacturing when required by pharmaceutical partners.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN114539198A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
