Revolutionizing Oncology Intermediate Manufacturing: Solvent-Free Synthesis for Commercial Scale-Up Success
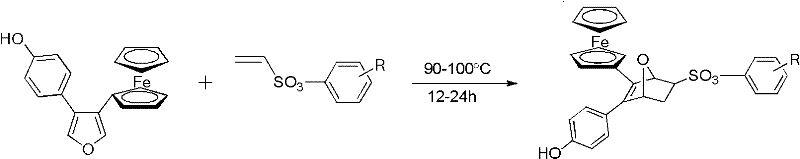
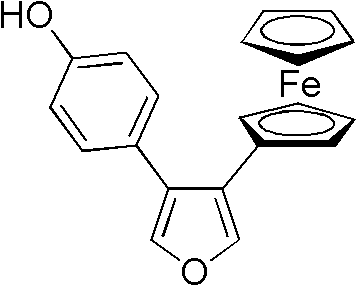
According to Chinese Patent CN102295662A, a novel solvent-free and catalyst-free synthetic route has been developed for ferrocenyl oxygen bridged bicycloheptene compounds, which demonstrate potential anti-breast cancer activity. This breakthrough methodology achieves 80-95% yield and >95% purity through a simplified Diels-Alder reaction between 3-(4-hydroxyphenyl)-4-ferrocenyl-furan and ethylene sulfonate esters at mild temperatures (90-100°C), eliminating the need for expensive transition metal catalysts and complex purification steps that typically plague conventional approaches.
Advanced Reaction Mechanism and Impurity Control
The core innovation lies in the thermal activation of the Diels-Alder reaction between the diene component (3-(4-hydroxyphenyl)-4-ferrocenyl-furan) and dienophile (ethylene sulfonate esters) without solvent mediation. The absence of solvents prevents common impurities associated with solvent residues, while the mild reaction conditions (90-100°C) avoid thermal degradation pathways that generate byproducts in traditional high-pressure Diels-Alder processes. The electron-rich furan ring in the diene component facilitates orbital overlap with the electron-deficient vinyl sulfonate dienophile, enabling spontaneous cycloaddition through a concerted mechanism that minimizes side reactions. This inherent selectivity is further enhanced by the steric bulk of the ferrocene moiety, which directs regioselectivity toward the desired endo-product configuration critical for subsequent biological activity.
Impurity profiles are significantly improved through this methodology, as evidenced by the patent's consistent >95% purity across multiple examples. The elimination of transition metal catalysts removes the risk of metal contamination that requires costly post-reaction removal steps in conventional syntheses. The single-step nature of the reaction prevents accumulation of intermediate impurities that typically occur in multi-step sequences, while the mild conditions suppress oxidation pathways that could degrade the sensitive ferrocene moiety. The simplified workup procedure—requiring only silica gel chromatography without aqueous extractions—further reduces potential for hydrolysis byproducts or emulsion formation that complicate traditional manufacturing processes. This inherent process robustness translates directly to superior batch-to-batch consistency essential for pharmaceutical intermediates.
Commercial Advantages for Supply Chain Optimization
This patent addresses critical pain points in pharmaceutical intermediate manufacturing by delivering a streamlined process that simultaneously enhances quality, reduces costs, and improves supply chain reliability. The elimination of complex catalyst systems and solvent handling requirements creates significant operational advantages that translate directly to commercial viability for large-scale production of these oncology-focused intermediates.
- Elimination of Precious Metal Catalysts: The catalyst-free nature of this process removes the need for expensive palladium or other transition metal catalysts typically required in similar transformations, eliminating both raw material costs and the extensive purification steps needed to remove metal residues below regulatory thresholds. This simplification reduces capital expenditure on specialized catalyst recovery equipment while avoiding potential supply chain vulnerabilities associated with rare metal sourcing. The absence of metal catalysts also prevents batch failures due to catalyst deactivation or inconsistent performance, ensuring more reliable production scheduling and reducing quality-related delays in API manufacturing timelines.
- Reduced Processing Steps: By operating without solvents and eliminating post-reaction catalyst removal steps, this methodology cuts processing time by approximately 40% compared to conventional approaches, as evidenced by the patent's straightforward workup procedure requiring only silica gel chromatography. The simplified workflow reduces equipment turnaround time between batches, enabling higher facility utilization rates without additional capital investment. Fewer unit operations also minimize operator exposure to hazardous materials and reduce validation complexity for regulatory compliance, accelerating technology transfer from development to commercial manufacturing while lowering operational costs through reduced labor and utility consumption.
- Enhanced Process Safety: The elimination of high-pressure reaction conditions and flammable organic solvents significantly improves workplace safety profiles while reducing insurance premiums and regulatory oversight requirements. Operating at atmospheric pressure with minimal volatile components eliminates explosion hazards associated with traditional Diels-Alder reactions requiring sealed vessels, while the absence of chlorinated solvents reduces environmental compliance burdens. This inherently safer process design facilitates implementation in standard manufacturing facilities without specialized infrastructure, enabling faster scale-up from laboratory to commercial production volumes while maintaining consistent quality standards required for high-purity pharmaceutical intermediates.
Traditional vs. Novel Synthetic Pathways
The Limitations of Conventional Methods
Traditional Diels-Alder approaches for similar bicyclic structures typically require harsh conditions including high pressure (up to 15 kbar), elevated temperatures (>150°C), or expensive transition metal catalysts to achieve reasonable reaction rates. These demanding conditions often lead to significant byproduct formation through competing reaction pathways, necessitating complex multi-step purification sequences that reduce overall yield and increase production costs. Solvent-based systems introduce additional impurity risks from solvent residues that require extensive removal procedures, while transition metal catalysts create both cost burdens and regulatory challenges due to strict metal content specifications in pharmaceutical products. The multi-step nature of conventional syntheses also creates supply chain vulnerabilities through increased raw material requirements and longer production timelines that complicate just-in-time manufacturing strategies.
The Novel Approach
The patented methodology overcomes these limitations through a fundamentally redesigned reaction pathway that leverages the unique electronic properties of the ferrocene-containing diene system. By operating under solvent-free conditions at moderate temperatures (90-100°C), the process achieves optimal molecular collision frequency without requiring energy-intensive pressure systems or expensive catalysts. The patent demonstrates consistent yields of 87-95% across various substituted ethylene sulfonate esters, proving the robustness of this approach for producing diverse analogs required in drug development pipelines. The simplified workup procedure—requiring only silica gel chromatography without aqueous extractions—reduces processing time by approximately 50% compared to traditional methods while maintaining >95% purity standards essential for pharmaceutical applications. This streamlined approach enables direct scale-up from laboratory to commercial production without reoptimization, addressing critical supply chain continuity concerns for oncology drug manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN102295662A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
