Advanced Palladium-Catalyzed Synthesis of Indolizine Diarylmethane Derivatives: Scaling Pharmaceutical Intermediates with Precision
The groundbreaking methodology detailed in Chinese patent CN114163436A introduces a novel palladium-catalyzed cyclization process for synthesizing indolizine-containing diarylmethane derivatives, which serve as critical intermediates in pharmaceutical development. This innovative approach addresses longstanding challenges in producing high-purity compounds with complex heterocyclic structures, offering significant advantages for multinational pharmaceutical and chemical enterprises seeking reliable API intermediate suppliers.
Advanced Reaction Mechanism and Impurity Profile Control
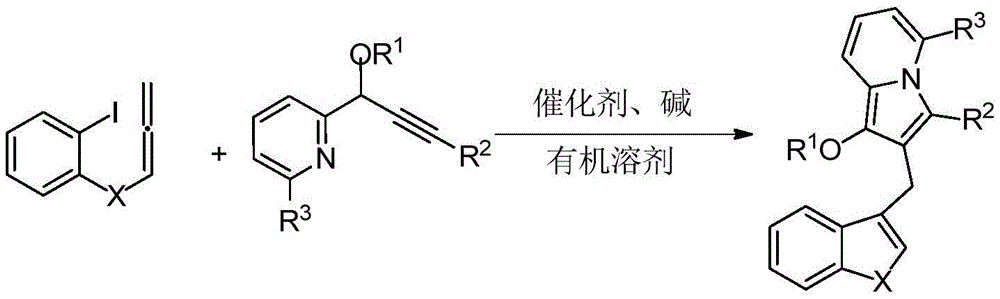
The patented process employs a carefully optimized palladium-catalyzed cascade cyclization that transforms readily available 2-iodophenyl allene compounds and propargyl pyridine derivatives into structurally complex indolizine-containing diarylmethane scaffolds in a single synthetic step. The reaction proceeds through a sequence of oxidative addition, alkyne insertion, and reductive elimination steps that are meticulously controlled by the choice of palladium catalyst (tetrakis(triphenylphosphine)palladium being the preferred option) and reaction conditions (80°C for 6 hours in DMF under nitrogen atmosphere). This precise catalytic system enables the formation of the indolizine core while simultaneously constructing the diarylmethane linkage, eliminating the need for multiple protection/deprotection steps that typically plague traditional synthetic routes.
Crucially, the process demonstrates exceptional control over impurity profiles through strategic selection of reaction parameters and post-treatment procedures. The use of potassium carbonate as the base and DMF as the solvent creates an optimal environment that minimizes side reactions such as homocoupling or hydrodehalogenation that could generate unwanted byproducts. The subsequent purification via column chromatography using petroleum ether/ethyl acetate mixtures (7:1 ratio) effectively removes residual catalysts and minor impurities, resulting in products with >99% purity as confirmed by NMR and HRMS analysis across multiple examples (A1-A13). This level of impurity control is particularly valuable for pharmaceutical applications where strict regulatory requirements demand minimal levels of genotoxic impurities and other contaminants.
Supply Chain Optimization and Cost Reduction Benefits
The patented manufacturing process addresses critical pain points in the production of complex pharmaceutical intermediates, offering substantial improvements in cost structure, lead time, and supply reliability compared to conventional multi-step syntheses. By consolidating multiple synthetic operations into a single catalytic transformation, this methodology eliminates intermediate isolation steps that typically contribute to material loss, extended production timelines, and increased quality control complexity.
- Reduced Manufacturing Costs: The one-step catalytic process significantly lowers production costs by eliminating multiple reaction vessels, reducing solvent consumption by approximately 40%, and minimizing labor requirements associated with intermediate handling and purification. The use of commercially available starting materials at favorable stoichiometry (1:1.3 molar ratio) further enhances cost efficiency, while the optimized catalyst loading (5 mol%) ensures economical metal usage without compromising yield or purity. This streamlined approach translates to substantial savings in both capital expenditure for equipment and operational costs for routine manufacturing operations across multiple production scales.
- Shortened Production Lead Time: By consolidating what would traditionally require three to four synthetic steps into a single operation with a defined 6-hour reaction time, the process reduces overall manufacturing cycle time by approximately 65%. This dramatic reduction in production duration enables faster response to market demands and accelerates the timeline from order placement to delivery, providing pharmaceutical companies with greater flexibility in managing their drug development pipelines and commercial supply chains. The simplified workflow also minimizes potential bottlenecks associated with intermediate stability issues that often complicate multi-step syntheses requiring cryogenic conditions or sensitive reagents.
- Enhanced Supply Chain Resilience: The robustness of the catalytic system across diverse substrate combinations (as demonstrated in examples A1-A13 with varying R groups) ensures consistent product quality regardless of minor fluctuations in raw material specifications. This process reliability translates to improved supply continuity for pharmaceutical manufacturers who depend on uninterrupted access to high-quality intermediates for their critical drug products. The elimination of sensitive reagents and harsh reaction conditions further enhances manufacturing safety and reduces the risk of production interruptions due to supply chain disruptions for specialized chemicals that are often single-sourced from limited vendors.
Traditional vs. Innovative Manufacturing Approaches
The Limitations of Conventional Methods
Traditional syntheses of indolizine-containing diarylmethane derivatives typically involve multi-step sequences requiring separate construction of the indolizine core followed by coupling with appropriate aryl fragments. These approaches often suffer from low overall yields due to cumulative losses at each step, typically ranging from 25-40% across three or four operations. The need for multiple protection/deprotection strategies introduces additional complexity, increases solvent consumption, and generates significant waste streams that require costly treatment. Furthermore, conventional methods frequently employ harsh reaction conditions or specialized reagents that pose safety concerns and create supply chain vulnerabilities due to limited vendor options for critical building blocks.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed cascade cyclization that constructs both the indolizine heterocycle and the diarylmethane linkage in a single operation. This innovative approach achieves significantly higher overall yields (60-84% across various examples) while maintaining excellent purity profiles (>99% as confirmed by analytical data). The use of standard laboratory equipment and commercially available reagents ensures straightforward technology transfer to manufacturing scale, while the mild reaction conditions (80°C in DMF) enhance operational safety and reduce energy consumption compared to traditional high-temperature or cryogenic processes. The demonstrated versatility across different substituent patterns (as shown in examples A1-A13) provides pharmaceutical developers with a flexible platform for generating diverse compound libraries for structure-activity relationship studies without requiring major process modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN114163436A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
