Revolutionizing Pharmaceutical Intermediate Production with Advanced N-Acylindole Synthesis Technology
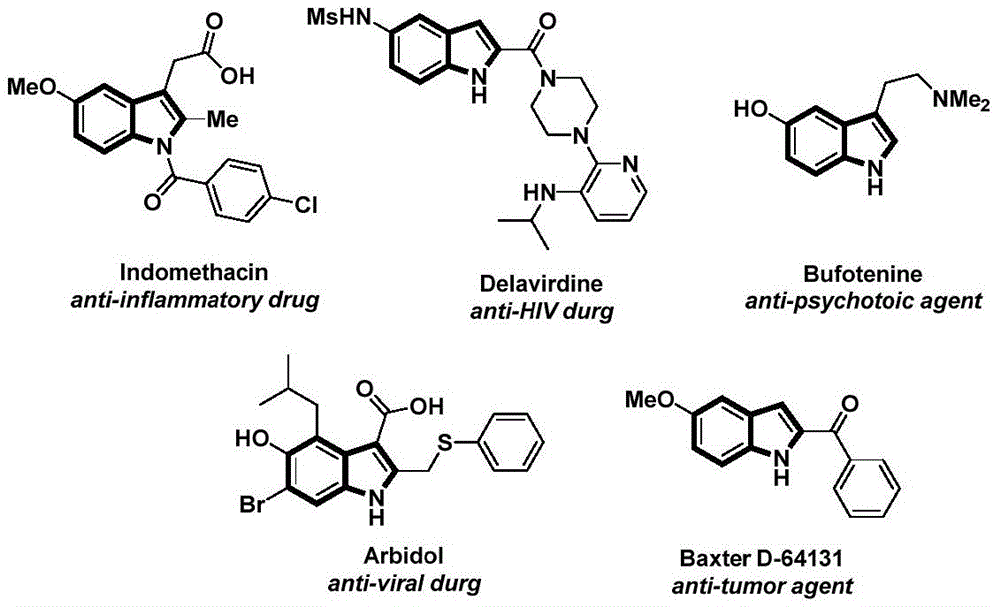
The innovative methodology disclosed in Chinese patent CN112898192A introduces a novel palladium-catalyzed carbonylation approach for synthesizing N-acylindole compounds, a critical class of pharmaceutical intermediates with applications in anti-inflammatory and antiviral drug development. This one-step process utilizes readily available starting materials including palladium catalysts and phenol-based carbon monoxide surrogates to construct complex molecular architectures under mild reaction conditions. The strategic elimination of high-pressure carbon monoxide equipment through the use of tricarboxylic acid phenol ester (TFBen) as a CO substitute represents a significant operational advancement while maintaining excellent substrate compatibility across diverse functional groups. As evidenced by the structural diversity of pharmaceutical agents shown in Figure 1, these compounds serve as essential building blocks for therapeutics including anti-HIV and anti-tumor medications.
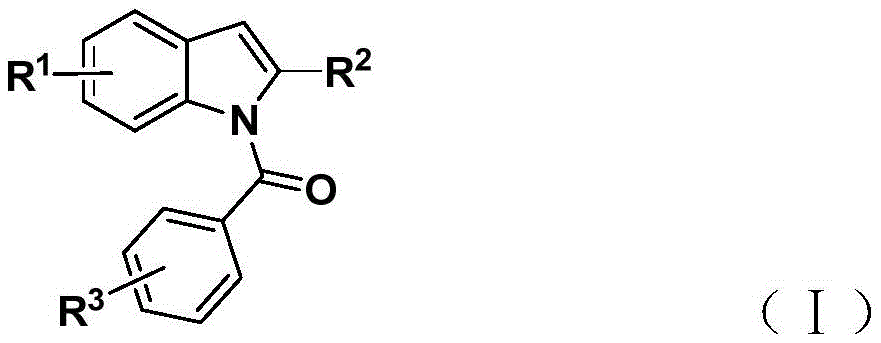
Mechanistic Advancements in N-Acylindole Synthesis
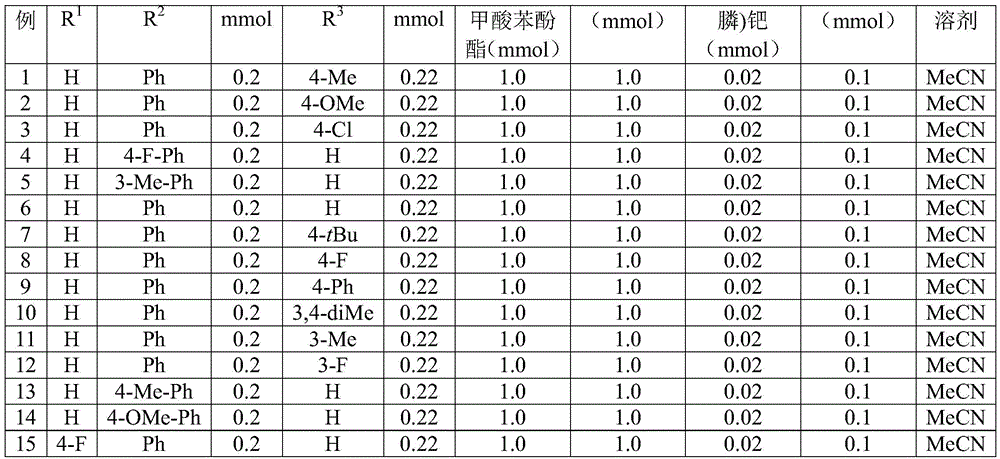
The reaction mechanism begins with palladium insertion into the aryl iodide bond to form an aryl palladium intermediate, followed by carbon monoxide insertion from the phenol tricarboxylate source to generate an acyl palladium species. This critical transformation occurs under ambient pressure conditions at 60°C in acetonitrile solvent, eliminating the safety hazards and specialized equipment requirements associated with traditional high-pressure carbonylation processes. The subsequent addition of 2-alkynyl aniline facilitates reductive elimination to form an amide intermediate, which then undergoes silver oxide-mediated cyclization to yield the final N-acyl indole product. This cascade reaction demonstrates remarkable functional group tolerance as illustrated in Figures 2 and 3, accommodating various substituents including halogens, alkyl groups, and alkoxy moieties without requiring protective group strategies. The precise control over regioselectivity prevents undesired isomer formation that commonly plagues conventional indole syntheses.


Impurity profile management is significantly enhanced through this methodology due to the absence of transition metal residues that typically require extensive purification steps in alternative synthetic routes. The structural confirmation data provided in the patent through comprehensive NMR analysis (1H and 13C) demonstrates high fidelity in product formation across multiple derivatives as shown in Figures 4 through 7. The consistent spectral patterns observed across different substituents indicate minimal side product formation during the cyclization step. Column chromatography purification remains the primary post-processing step as specified in the patent description, but the simplified reaction pathway reduces the number of potential impurities compared to multi-step conventional approaches. This inherent selectivity directly contributes to higher final product purity without requiring additional costly purification stages that would otherwise increase manufacturing complexity and reduce overall yield efficiency.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis methodology addresses critical pain points in pharmaceutical intermediate manufacturing by transforming complex multi-step processes into a streamlined two-stage operation that significantly enhances supply chain resilience while reducing production costs. The elimination of specialized high-pressure equipment requirements and the use of commercially available reagents create immediate opportunities for cost reduction in API manufacturing without compromising product quality or process reliability. By leveraging standard laboratory equipment and ambient pressure conditions, manufacturers can achieve faster implementation timelines while minimizing capital expenditure requirements for new production facilities.
- Cost reduction in API manufacturing: The substitution of hazardous high-pressure carbon monoxide with stable phenol-based surrogates eliminates both capital costs for specialized pressure equipment and ongoing operational expenses associated with safety protocols and maintenance. This approach also reduces raw material costs by utilizing inexpensive potassium carbonate as a base instead of more expensive alternatives required in conventional methods. The simplified two-stage process minimizes solvent consumption and waste generation compared to traditional multi-step syntheses, directly contributing to lower environmental compliance costs. Furthermore, the broad substrate compatibility demonstrated across multiple functional groups allows manufacturers to produce diverse derivative compounds using identical equipment and process parameters, maximizing facility utilization and reducing changeover expenses.
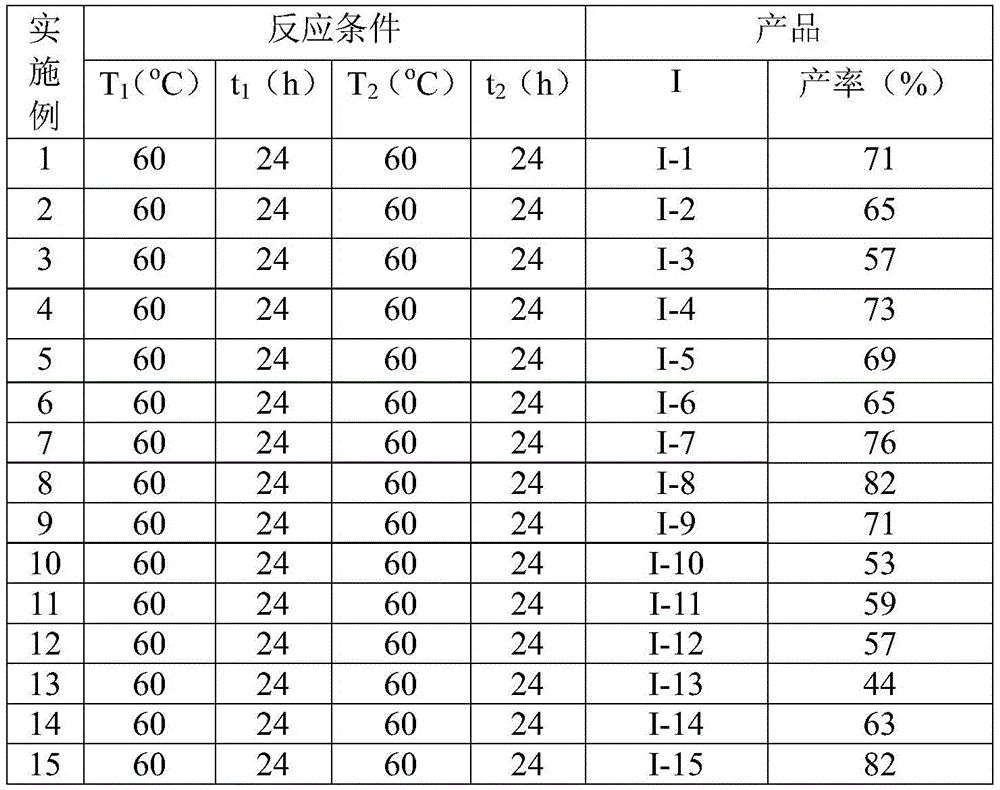
- Reducing lead time for high-purity intermediates: The straightforward reaction protocol operating at moderate temperatures (60°C) enables faster cycle times compared to conventional methods requiring extreme conditions or lengthy protection/deprotection sequences. The consistent reaction duration of 48 hours across diverse substrates as documented in Table 2 creates predictable production scheduling that enhances supply chain planning accuracy. The simplified post-processing procedure involving standard filtration and column chromatography eliminates complex purification steps that typically cause production bottlenecks in traditional syntheses. This operational simplicity translates directly to shorter manufacturing lead times while maintaining the high purity standards required for pharmaceutical applications as confirmed by the comprehensive NMR characterization data provided in the patent examples.
- Commercial scale-up of complex intermediates: The mild reaction conditions (60°C) and standard solvent system (acetonitrile) enable seamless transition from laboratory to production scale without requiring specialized engineering modifications to existing manufacturing infrastructure. The demonstrated scalability is evidenced by consistent yields across various substituents as shown in Table 2, indicating robust process performance under different material inputs. The absence of sensitive reagents or extreme process parameters ensures reliable batch-to-batch consistency during scale-up operations. This inherent scalability provides manufacturers with confidence in maintaining product quality while increasing production volumes to meet growing market demands for these critical pharmaceutical intermediates.
Superior Process Performance vs Conventional Methods
The Limitations of Conventional Methods
Traditional approaches for synthesizing N-acyl indole compounds typically involve multi-step sequences requiring harsh reaction conditions that compromise both efficiency and safety profiles. These conventional methods often necessitate cryogenic temperatures or high-pressure carbon monoxide systems that introduce significant operational hazards and require specialized equipment not commonly available in standard pharmaceutical manufacturing facilities. The need for multiple protection/deprotection steps creates additional complexity that increases both processing time and the potential for impurity formation throughout the synthetic pathway. Furthermore, limited substrate compatibility frequently forces manufacturers to develop customized processes for each derivative compound, resulting in inconsistent production timelines and variable yield profiles that undermine supply chain reliability. These inherent limitations create substantial barriers to efficient large-scale production while increasing both capital investment requirements and operational costs.
The Novel Approach
The patented methodology overcomes these challenges through an elegant one-pot cascade reaction that integrates carbonylation and cyclization steps under mild ambient pressure conditions at 60°C as illustrated in Figure 5. This innovative approach utilizes commercially available palladium catalysts and phenol-based carbon monoxide surrogates to achieve efficient molecular construction without requiring specialized high-pressure equipment. The broad functional group tolerance demonstrated across multiple substituents including halogens, alkyl groups, and alkoxy moieties enables manufacturers to produce diverse derivative compounds using identical process parameters as documented in Tables 1 and 2. The consistent yield ranges observed across different substrates (44%-82%) confirm the robustness of this methodology while eliminating the need for customized process development for each compound variant. This strategic simplification directly addresses key manufacturing pain points by reducing both processing complexity and equipment requirements while maintaining excellent product quality standards.
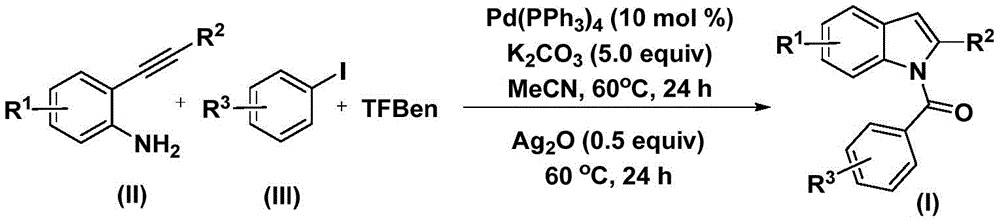

Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112898192A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
