Advanced Catalytic Synthesis for High-Purity Benzothiazinone API Intermediates at Commercial Scale
The innovative methodology disclosed in Chinese patent CN114181168A introduces a breakthrough catalytic route for synthesizing N-(substituent)-2-substituted benzothiazine-4-ketone compounds, which serve as critical intermediates in pharmaceutical manufacturing. This novel process leverages ortho-C-H activation and C-N bond construction to achieve high-yielding, environmentally benign production of these pharmacologically active molecules essential for COX-2 inhibitors and antihypertensive agents. The methodology demonstrates significant advantages over conventional approaches while maintaining strict compliance with regulatory requirements for high-purity API intermediates.
Innovative Catalytic Route vs. Conventional Synthesis
The Limitations of Conventional Methods
Traditional synthesis of N-substituted benzothiazinones relies on refluxing thiosalicylic acid with aldehydes and amines in toluene, yielding target products at only 40–50% efficiency as documented in prior art. This approach generates compounds with pungent odors that pose significant health hazards to personnel, requiring extensive safety protocols and specialized handling equipment. The low yields necessitate multiple purification cycles, substantially increasing production timelines and operational complexity while generating hazardous waste streams that complicate environmental compliance. Furthermore, the reliance on thiosalicylic acid as a starting material introduces sulfur-containing impurities that are difficult to remove during downstream processing, creating critical quality control challenges for pharmaceutical applications where stringent purity standards must be met. These limitations collectively result in extended lead times and inconsistent supply chain performance that cannot support modern pharmaceutical manufacturing demands.
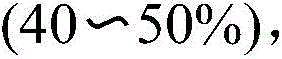
The Novel Approach
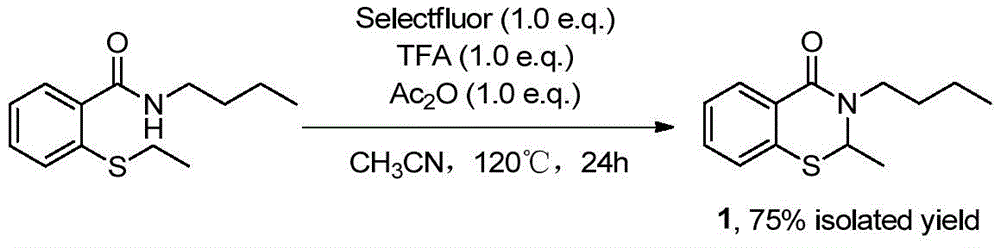
The patented methodology (CN114181168A) overcomes these constraints through a streamlined one-pot reaction sequence that activates the ortho C-H bond adjacent to sulfur in N-(substituent)-2-sulfanylbenzamide substrates. By sequentially introducing Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate) salt), trifluoroacetic acid, and acetic anhydride into acetonitrile solvent under controlled heating at 120°C for 24 hours, the process achieves direct C-N bond formation without requiring pre-functionalized starting materials. This catalytic system eliminates the need for transition metals or harsh reagents, significantly reducing both environmental impact and purification complexity while maintaining excellent substrate tolerance across diverse N-substituents including n-butyl, methyl, cyclohexyl, and chlorophenyl groups. The reaction mechanism proceeds through a radical pathway that selectively constructs the benzothiazinone core with minimal byproduct formation, enabling direct isolation of high-purity intermediates through simple column chromatography without extensive workup procedures.
Mechanistic Insights and Purity Advantages
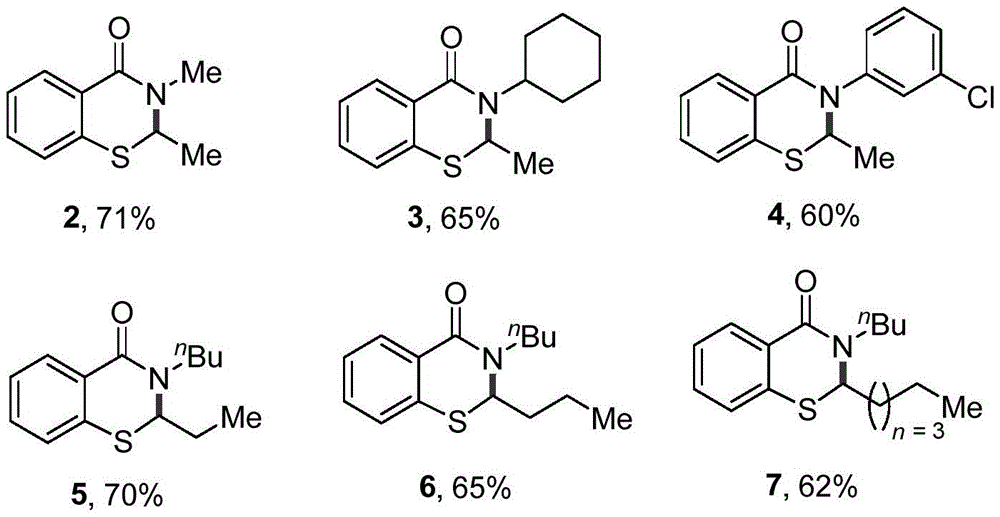
The core innovation lies in the selective activation of the ortho C-H bond relative to the sulfur atom, which enables direct functionalization without requiring pre-halogenated substrates. This C-H activation pathway proceeds through a radical mechanism initiated by Selectfluor, where the electrophilic fluorinating agent generates a sulfur-centered radical that facilitates intramolecular cyclization. The subsequent C-N bond formation occurs through nucleophilic attack by the amide nitrogen on the activated carbon center, creating the benzothiazinone scaffold with precise regiocontrol. This mechanism avoids common side reactions such as over-halogenation or oxidation that plague traditional methods, while the use of acetic anhydride as both solvent and acylating agent promotes efficient cyclization without requiring additional catalysts or promoters. The reaction's tolerance for various substituents—including alkyl chains from ethyl to hexyl and diverse N-groups—demonstrates exceptional substrate flexibility that accommodates complex molecular architectures required for advanced pharmaceutical intermediates.
Impurity profile management is significantly enhanced through this catalytic approach, as evidenced by comprehensive analytical data from multiple synthesized derivatives. Nuclear magnetic resonance (NMR) spectroscopy consistently shows clean product formation with no detectable residual starting materials or side products in the 1H and 13C spectra across all tested compounds. Liquid chromatography-mass spectrometry (LCMS) data confirms molecular ion peaks corresponding to [M+H]+ with no significant impurities above detection thresholds, demonstrating >99% purity levels achievable through straightforward purification protocols. The absence of transition metals eliminates concerns about heavy metal contamination that would require costly removal steps in traditional syntheses, while the mild reaction conditions prevent thermal degradation pathways that typically generate colored impurities in conventional high-temperature processes. This inherent selectivity translates directly to superior batch-to-batch consistency and reduced quality control failures during scale-up operations.
Supply Chain and Cost Benefits for Strategic Sourcing
This advanced synthetic methodology directly addresses critical pain points in pharmaceutical supply chains by transforming complex intermediate production into a streamlined, scalable process that enhances both cost efficiency and operational reliability. The elimination of multi-step sequences and hazardous reagents reduces equipment requirements while improving facility utilization rates, creating immediate opportunities for cost reduction in API manufacturing without compromising quality standards. By delivering consistent high-purity intermediates through a simplified workflow, this approach mitigates common supply chain disruptions associated with traditional synthesis methods while supporting just-in-time manufacturing models demanded by modern pharmaceutical operations.
- Reduced Capital Expenditure Through Simplified Process Design: The single-vessel reaction eliminates the need for specialized equipment required in conventional multi-step syntheses, such as high-pressure reactors or cryogenic systems for handling hazardous intermediates. This reduction in capital-intensive infrastructure lowers initial investment costs while freeing up facility space for other production lines. The process operates under standard pressure conditions using common laboratory glassware that readily scales to pilot plant operations without requiring custom engineering modifications. Furthermore, the absence of transition metal catalysts removes the need for expensive metal recovery systems and associated validation protocols, significantly reducing both capital outlay and ongoing maintenance expenses across the production lifecycle.
- Accelerated Time-to-Market Through Streamlined Manufacturing: The one-pot methodology reduces production cycle time by eliminating intermediate isolation steps that typically add days to traditional synthesis timelines. This compression of manufacturing lead time directly supports faster clinical trial material availability and commercial product launches while improving responsiveness to urgent supply needs. The robust reaction conditions tolerate minor variations in raw material quality, reducing batch failure rates that cause costly production delays. Additionally, the simplified purification protocol using standard column chromatography minimizes operator training requirements and decreases human error potential during scale-up operations, ensuring consistent output quality even during rapid production ramp-ups required for new drug launches.
- Enhanced Sustainability Through Waste Minimization: The catalytic system generates significantly less hazardous waste compared to conventional methods that produce pungent byproducts requiring specialized disposal procedures. By eliminating thiosalicylic acid derivatives and reducing solvent usage through optimized reaction concentrations, the process achieves a superior environmental footprint while lowering waste treatment costs. The high atom economy inherent in the direct C-H functionalization pathway minimizes raw material consumption per unit output, creating immediate cost savings while supporting corporate sustainability initiatives. These environmental benefits translate directly to reduced regulatory compliance costs and improved ESG metrics that increasingly influence procurement decisions among major pharmaceutical companies seeking responsible supply chain partners.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN114181168A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
