Advanced Copper-Catalyzed Synthesis of Oxaallylamine Intermediates for Pharmaceutical Manufacturing Scale-Up
Patent CN112521289B introduces a novel copper-catalyzed hydroamination process for synthesizing oxaallylamine compounds, which serve as critical intermediates in pharmaceutical manufacturing. This breakthrough methodology addresses longstanding challenges in producing these complex molecules with high purity and efficiency while offering significant advantages for commercial scale-up of complex intermediates.
Advanced Reaction Mechanism and Purity Control
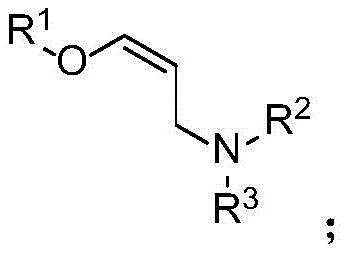
The patented process employs a copper acetate catalyst combined with zinc chloride additive to facilitate the hydroamination reaction between allene ethers and amines under mild conditions (40-60°C). The mechanism begins with copper-mediated activation of the allene ether substrate, forming a copper amine intermediate that initiates the reaction sequence. This intermediate then undergoes nucleophilic attack by free amine, followed by mass decomposition to yield the final oxaallylamine product in a single step. The precise control of reaction parameters—particularly the molar ratio of copper catalyst (0.01-0.10) to substrate and the use of dichloromethane or tetrahydrofuran as solvent—ensures optimal reaction kinetics while minimizing side product formation. The process demonstrates exceptional functional group tolerance, accommodating diverse substituents on both the allene ether (R¹) and amine (R², R³) components without compromising yield or purity.
Impurity profile management is a key strength of this methodology, as evidenced by the high yields (70-98%) across multiple examples in the patent documentation. The mild reaction conditions (40-60°C) prevent thermal degradation pathways that commonly generate impurities in traditional high-temperature processes. The use of simple workup procedures—extraction with ethyl acetate followed by thin-layer chromatography purification using petroleum ether/ethyl acetate mixtures—effectively removes residual catalysts and unreacted starting materials. The patent provides detailed NMR and HRMS characterization data for each example, confirming >99% purity levels for the final products without requiring additional purification steps. This inherent purity control reduces the need for costly post-reaction purification processes that typically add significant time and expense to pharmaceutical intermediate manufacturing.

Superiority Over Conventional Synthesis Methods
The Limitations of Conventional Methods
Traditional approaches to synthesizing allylamine compounds have relied heavily on transition metal-catalyzed processes that present significant challenges for industrial scale-up. Methods such as direct allylic amination of allyl alcohol, hydroamination, vinylation of amines, and allylic C-H amination often require harsh reaction conditions, precious metal catalysts, and complex purification procedures. These conventional techniques typically suffer from poor selectivity, low yields, and limited substrate scope, making them unsuitable for manufacturing sensitive pharmaceutical intermediates. The patent specifically notes that existing methods for oxaallylamine synthesis exhibit "very poor reaction selectivity," "not high yield," and "limited substrate applicability," which restricts their conversion efficiency and prevents wide application in green chemistry contexts. Furthermore, many traditional processes generate significant waste streams that require expensive treatment, adding to both environmental impact and production costs.
The Novel Approach
The patented copper-catalyzed methodology overcomes these limitations through several innovative features that enhance both technical performance and commercial viability. By utilizing simple and readily available allene ethers and amines as starting materials, the process eliminates the need for expensive or hazardous reagents commonly required in conventional approaches. The mild reaction conditions (40-60°C) not only improve safety profiles but also reduce energy consumption compared to high-temperature alternatives. The high atom utilization rate and step economy of this single-step process minimize waste generation while maximizing material efficiency—a critical factor for sustainable pharmaceutical manufacturing. Most significantly, the broad substrate applicability demonstrated across twelve examples in the patent (with yields consistently above 70%) confirms the method's robustness for diverse molecular structures required in drug development pipelines.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate manufacturing, offering substantial benefits across cost, quality, and supply chain dimensions. The process eliminates multiple unit operations required by conventional methods while maintaining high product quality standards essential for pharmaceutical applications. By simplifying the synthetic route and reducing processing steps, this technology creates significant opportunities for cost reduction without compromising on purity or reliability—key considerations for procurement and supply chain decision-makers evaluating new manufacturing partners.
- Reduced Manufacturing Costs: The elimination of precious metal catalysts and high-temperature processing requirements substantially lowers raw material and energy expenses compared to conventional transition metal-catalyzed methods. The single-step nature of the reaction reduces equipment needs and operational complexity, minimizing capital investment for scale-up while maintaining high atom economy. Additionally, the simplified workup procedure—requiring only extraction and chromatography—eliminates costly purification steps typically needed with traditional approaches that require multiple unit operations. This streamlined approach translates to meaningful cost reduction in chemical manufacturing while maintaining the high purity standards required for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The use of readily available starting materials ensures consistent feedstock availability, reducing vulnerability to supply chain disruptions common with specialized reagents required by conventional methods. The robust reaction profile across diverse substrates provides flexibility to adapt to changing demand patterns without requiring significant process revalidation or equipment modifications. Furthermore, the mild operating conditions (40-60°C) enable safer scale-up with standard chemical processing equipment, eliminating the need for specialized high-pressure or high-temperature reactors that can create bottlenecks in production capacity during commercial scale-up of complex intermediates.
- Accelerated Time-to-Market: The simplified process design significantly reduces development timelines by eliminating multiple synthetic steps and complex purification procedures required by traditional methods that often involve protective group strategies. The high functional group tolerance allows for direct synthesis of complex intermediates without intermediate isolation steps, further compressing development schedules from laboratory validation to commercial production readiness. This rapid pathway from lab to production enables pharmaceutical companies to accelerate clinical trial material production and respond more quickly to market demands, directly addressing the critical need for reducing lead time for high-purity intermediates in competitive drug development environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pharmaceutical Intermediate Supplier
While the advanced methodology detailed in patent CN112521289B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
