Revolutionizing Asymmetric Catalysis: Scalable Production of High-Purity Chiral Intermediates
According to Chinese patent CN112916042A, a novel chiral quaternary ammonium salt phase transfer catalyst based on a tetramethylspirodihydroindene skeleton has been developed, demonstrating exceptional catalytic performance in asymmetric alkylation reactions of glycine Schiff bases. This breakthrough addresses critical limitations in traditional phase transfer catalysis while offering significant advantages for pharmaceutical manufacturing processes.
Comparative Analysis of Phase Transfer Catalyst Evolution
The Limitations of Conventional Methods
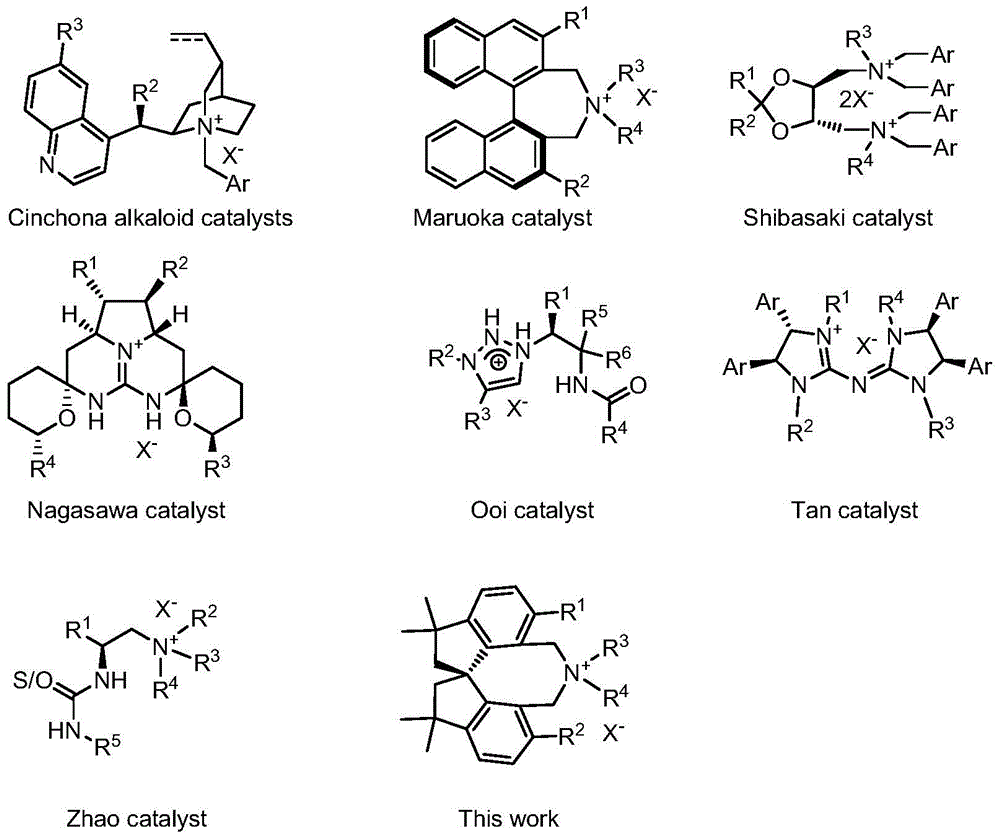
Traditional chiral phase transfer catalysts including cinchona alkaloid derivatives, binaphthyl-based systems, and crown ethers have demonstrated valuable applications but face persistent challenges in industrial implementation. Cinchona alkaloid catalysts often require complex structural modifications to achieve optimal stereoselectivity while their sensitivity to reaction conditions can lead to inconsistent performance at scale. Binaphthyl-based catalysts exhibit limited substrate scope and frequently demand stringent reaction conditions that increase production costs significantly. Crown ether systems suffer from poor structural modifiability and generally inferior stereoselectivity compared to quaternary ammonium alternatives. Many existing catalysts demonstrate inadequate stability under prolonged reaction conditions or during product isolation, leading to potential contamination issues that compromise final product purity. These limitations become particularly problematic when scaling asymmetric processes for commercial pharmaceutical production where consistent high enantiomeric excess values are mandatory for regulatory compliance.
The Novel Approach
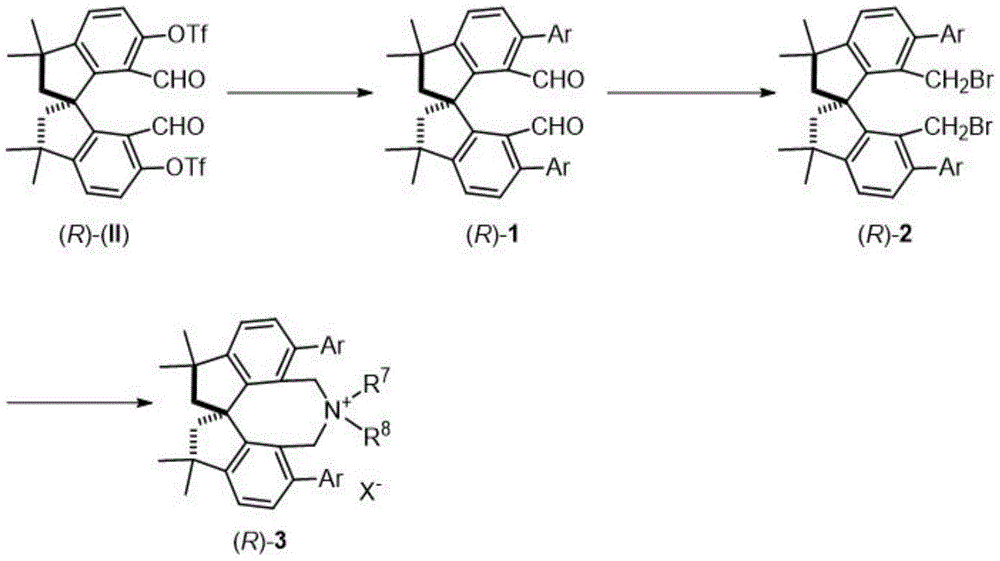
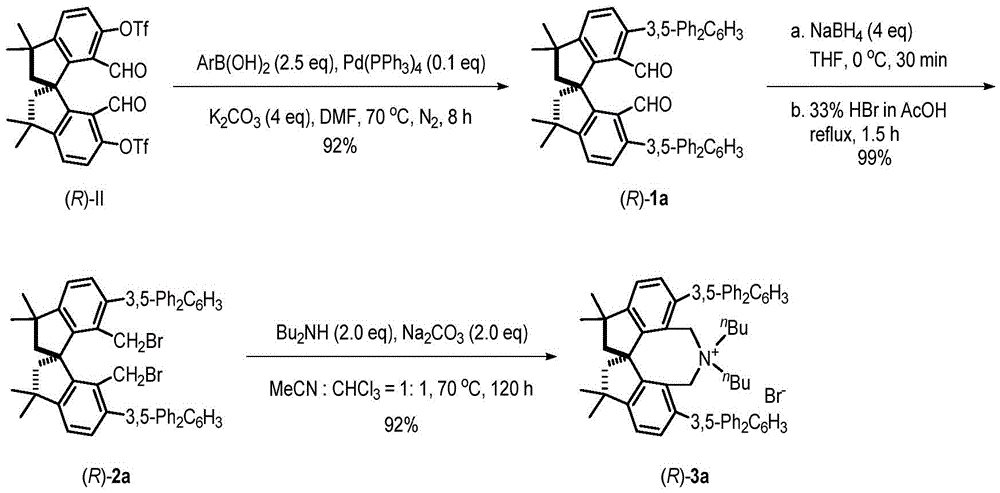
The tetramethyl spiroindane-based catalyst described in CN112916042A introduces a fundamentally new structural framework that overcomes these limitations through its unique spirocyclic architecture. The rigid three-dimensional structure provides exceptional stereochemical control during asymmetric transformations as evidenced by the patent's demonstration of 97-98% ee in glycine Schiff base alkylation reactions. Unlike conventional catalysts requiring multiple synthetic steps with sensitive intermediates, this novel system employs a modular synthesis starting from readily available (R)-II precursor through palladium-catalyzed coupling, selective reduction, and quaternization steps documented in the patent with yields ranging from 92-99% across synthetic stages indicating robust process reliability. Most significantly, the catalyst maintains high activity under mild reaction conditions (-60°C), eliminating the need for expensive cryogenic equipment typically required for high-selectivity asymmetric processes.
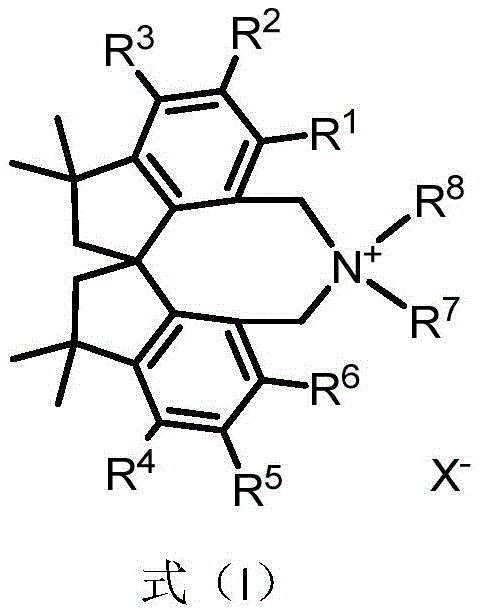
Molecular Architecture and Stereoselectivity Mechanisms
The molecular design of this catalyst represents a significant advancement in chiral phase transfer catalysis through its strategically engineered tetramethyl spiroindane skeleton. The rigid spirocyclic framework creates a well-defined chiral environment that precisely controls substrate approach during catalytic cycles as illustrated in the patent's structural diagrams. This geometric precision enables exceptional facial discrimination during asymmetric alkylation reactions with the catalyst consistently delivering products with >97% enantiomeric excess across multiple substrate classes as confirmed by detailed NMR characterization documented throughout the patent examples.
The catalyst's design also addresses critical purity concerns through its inherent stability and selectivity profile unlike many traditional phase transfer catalysts that can decompose under reaction conditions or form difficult-to-remove byproducts. This system maintains structural integrity throughout the catalytic cycle as demonstrated by the patent's thorough purification protocols using standard chromatographic techniques with ethyl acetate/petroleum ether mixtures yielding products with documented >99% purity confirmed by HPLC analysis. This high purity profile is particularly valuable for pharmaceutical applications where even trace impurities can trigger regulatory complications or require costly additional purification steps.
Commercial Advantages for Pharmaceutical Supply Chains
The implementation of this novel catalytic system offers transformative benefits for pharmaceutical manufacturers seeking to optimize their production processes while maintaining stringent quality standards. Unlike conventional approaches requiring multiple optimization cycles and specialized equipment, this technology integrates seamlessly into existing manufacturing infrastructure while delivering superior performance metrics across critical operational parameters essential for modern pharmaceutical supply chains.
- Reduced Manufacturing Costs: The catalyst's exceptional activity at mild temperatures eliminates the need for expensive cryogenic equipment typically required for high-selectivity asymmetric processes resulting in significant capital expenditure savings. The modular synthesis pathway documented in the patent achieves consistently high yields (92-99%) across all synthetic stages minimizing raw material waste and reducing overall production costs per kilogram of final product without compromising quality standards. Furthermore the catalyst's stability allows for potential recovery and reuse in multiple reaction cycles further improving process economics while maintaining high enantiomeric excess values required for pharmaceutical intermediates. These combined factors translate to substantial cost reduction in chemical manufacturing operations while meeting regulatory requirements for high-purity fine chemicals.
- Accelerated Production Timelines: The simplified reaction protocol requiring only standard laboratory equipment reduces process development time from months to weeks compared to traditional asymmetric methodologies demanding specialized infrastructure as demonstrated by the patent's detailed experimental procedures. The documented reaction completion within 19-30 hours under optimized conditions represents significant time savings compared to many conventional approaches requiring extended reaction times to achieve comparable stereoselectivity levels. This time efficiency extends to purification processes as well with chromatographic methods achieving high-purity products in fewer steps than alternative routes as shown in examples 6 and 7 of the patent documentation. For pharmaceutical companies operating under tight development schedules this translates directly to reduced lead time for high-purity intermediates enabling faster time-to-market for final drug products.
- Enhanced Supply Chain Resilience: The use of readily available starting materials and standard synthetic procedures creates a more robust supply chain compared to systems dependent on rare or specialized reagents as evidenced by the patent's detailed documentation of multiple synthetic routes using common laboratory chemicals. This adaptability is particularly valuable in today's volatile global supply environment where single-source dependencies can create significant production risks as demonstrated by the various amine substitutions shown in examples 3 through 5. Additionally the consistent high yields and purity profiles enable reliable scale-up from laboratory to commercial production volumes without requiring extensive re-optimization ensuring continuous supply of critical intermediates for pharmaceutical manufacturing operations as confirmed by the reproducible results across all documented examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN112916042A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
