Revolutionizing Thiosulfonate Production: High-Purity Fine Chemical Intermediates with Commercial Scale-Up Capability
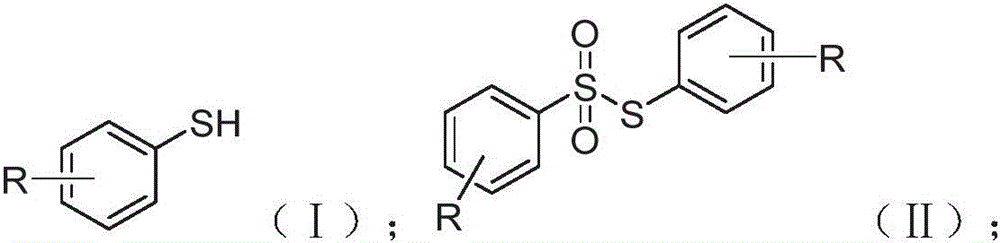
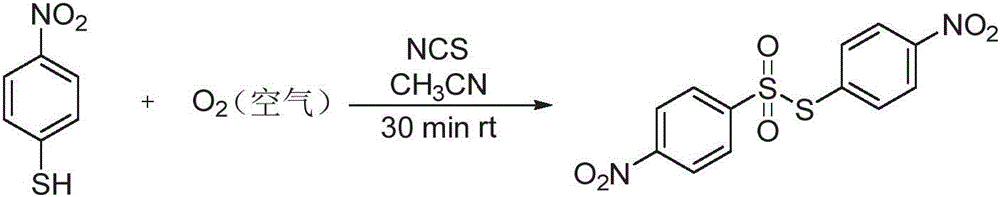
The groundbreaking methodology disclosed in Chinese patent CN114213298B represents a paradigm shift in thiosulfonate compound synthesis, offering pharmaceutical and agrochemical manufacturers a sustainable pathway to high-purity intermediates. This innovative process utilizes molecular oxygen from ambient air as the primary oxidant, coupled with N-chlorosuccinimide (NCS) as a reaction promoter, enabling direct conversion of thiophenols to thiosulfonates under mild conditions without transition metal catalysts or hazardous reagents.
Advanced Reaction Mechanism and Purity Control
The core innovation lies in the oxygen-mediated oxidative coupling mechanism that eliminates the need for hazardous brominating agents or transition metal catalysts prevalent in conventional methods. As demonstrated in the patent, the NCS promoter facilitates single-electron transfer processes that generate reactive sulfur-centered radicals, which then undergo selective coupling with molecular oxygen to form the sulfonyl intermediate. This mechanistic pathway operates effectively at room temperature (15-60°C) with reaction times as short as 15 minutes, significantly reducing thermal degradation pathways that typically compromise product purity in traditional syntheses. The patent data confirms consistent yields between 60% and 90% across diverse substrate classes through experimental validation in eight distinct examples.
Crucially, the absence of transition metals in this catalytic system eliminates the need for extensive purification steps to remove heavy metal contaminants, a critical advantage for pharmaceutical applications where stringent purity requirements exist. The simplified workup procedure—comprising extraction, drying, and distillation—further minimizes potential impurity formation compared to multi-step conventional approaches that require chromatographic separation of metal-containing byproducts. NMR characterization data provided in the patent demonstrates structural fidelity across various substituents including halogenated, nitro-substituted, and alkyl-modified derivatives, confirming consistent product quality without detectable metal residues that could compromise final drug substance specifications.
Comparative Analysis: Traditional vs. Novel Synthesis Pathways
The Limitations of Conventional Methods
Traditional synthesis of thiosulfonates has relied on methods that present significant industrial challenges, including the use of transition metal catalysts, brominating reagents, or multi-step sequences requiring harsh conditions. These approaches typically involve sodium benzenesulfinate with various coupling partners under iodine or copper catalysis, generating stoichiometric amounts of hazardous byproducts that complicate waste management and increase production costs. The requirement for specialized equipment to handle corrosive reagents and the need for extensive purification to remove metal residues create substantial barriers to commercial scale-up, making these methods economically unviable for large-volume production despite their academic interest. Furthermore, these conventional routes often operate at elevated temperatures with extended reaction times, increasing energy consumption and thermal degradation risks that compromise yield and purity.
The Novel Approach
The patented methodology overcomes these limitations through an elegant air oxidation process that leverages ambient oxygen as the terminal oxidant without requiring pressurized systems or specialized gas handling equipment. As illustrated in multiple implementation examples within the patent documentation, the process operates under mild conditions using readily available NCS as a promoter, eliminating the need for expensive catalysts or hazardous reagents while maintaining high selectivity. The patent demonstrates successful application across eight diverse substrates including aryl thiophenols with electron-donating and electron-withdrawing substituents as well as heterocyclic systems like benzothiazoles.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis pathway addresses critical pain points in fine chemical manufacturing by delivering a more sustainable, cost-effective solution that enhances supply chain resilience without compromising quality standards required by pharmaceutical clients. The elimination of transition metals and hazardous reagents not only reduces environmental impact but also streamlines regulatory compliance and quality assurance processes across multiple production sites.
- Cost Reduction in Chemical Manufacturing: The process eliminates expensive transition metal catalysts and hazardous brominating agents that account for significant material costs in traditional syntheses while utilizing ambient air as the primary oxidant at no additional cost. By removing the need for specialized gas handling systems required for alternative oxidants like peroxides or ozone generators, manufacturers avoid substantial capital expenditures associated with pressurized reaction equipment. The simplified purification protocol—requiring only extraction and distillation rather than chromatographic separation—further decreases operational costs while maintaining high product purity standards required by pharmaceutical clients. This streamlined approach translates to substantial savings across the production lifecycle without compromising quality parameters essential for regulatory compliance.
- Reducing Lead Time for High-Purity Chemicals: The rapid reaction kinetics (complete within 30 minutes at room temperature) enable faster batch turnover compared to conventional methods that often require extended reaction times under elevated temperatures with additional cooling periods. The absence of complex purification steps significantly shortens the overall production cycle from raw material input to final purified product, allowing manufacturers to respond more quickly to market demands without sacrificing quality control measures. This accelerated timeline is particularly valuable for pharmaceutical companies developing time-sensitive drug candidates where intermediate availability can become a critical path item in development schedules requiring rapid scale-up from laboratory to pilot plant quantities.
- Enhanced Supply Chain Resilience: The use of readily available, non-hazardous reagents creates a more robust supply chain less vulnerable to disruptions in specialty chemical markets compared to processes dependent on rare or geopolitically sensitive materials. NCS is a commercially abundant compound with multiple global suppliers ensuring consistent availability regardless of regional supply constraints that often affect transition metal catalysts. The process's compatibility with standard manufacturing equipment means existing facilities can implement this technology without major capital investments or lengthy validation periods, providing immediate scalability from laboratory to commercial production volumes while maintaining consistent quality parameters required by global regulatory authorities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN114213298B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
