Revolutionizing Triarylmethane Production: Advanced Catalysis for Commercial Scale-Up
The recently granted Chinese patent CN112159312B introduces a groundbreaking methodology for synthesizing triarylmethane compounds, a critical structural motif in pharmaceutical development with applications in optical reagents, dyes, and bioactive molecules exhibiting antioxidant and antitumor properties. This innovative approach addresses longstanding limitations in conventional synthesis routes by utilizing o-hydroxyphenyl p-QMs as the sole starting material through a tandem reverse-Friedel-Crafts/1,6-conjugate addition reaction under mild conditions. The process operates at ambient temperatures (25-60°C) with minimal catalyst loading (1-10 mol%), eliminating the need for complex pre-synthesized precursors or expensive transition metal catalysts that characterize traditional methods.
Novel Reaction Mechanism and Purity Advantages
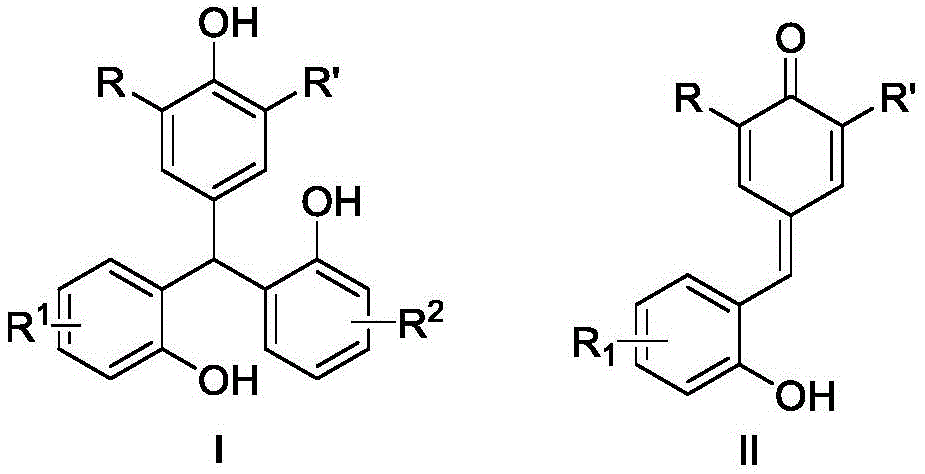
The core innovation lies in the nitrogen-protected, one-pot cascade reaction where Lewis acids like indium trichloride or silver hexafluorophosphate catalyze the transformation of o-hydroxyphenyl p-QMs (Structure II) into triarylmethane derivatives (Structure I) through a synergistic reverse-Friedel-Crafts and 1,6-conjugate addition sequence. This mechanism avoids the harsh conditions and unstable reagents required in conventional Friedel-Crafts alkylation or coupling approaches, which typically demand electron-rich arenes and generate complex impurity profiles. The patent demonstrates exceptional substrate versatility across diverse R-group substitutions (alkyl, aryl, halogen), enabling tailored synthesis of target compounds while maintaining high regioselectivity. Crucially, the water-mediated reaction pathway facilitates straightforward product isolation through standard column chromatography using petroleum ether/ethyl acetate mixtures (5:1 v/v), significantly reducing purification complexity compared to traditional methods that require extensive metal scavenging or multi-step workups.
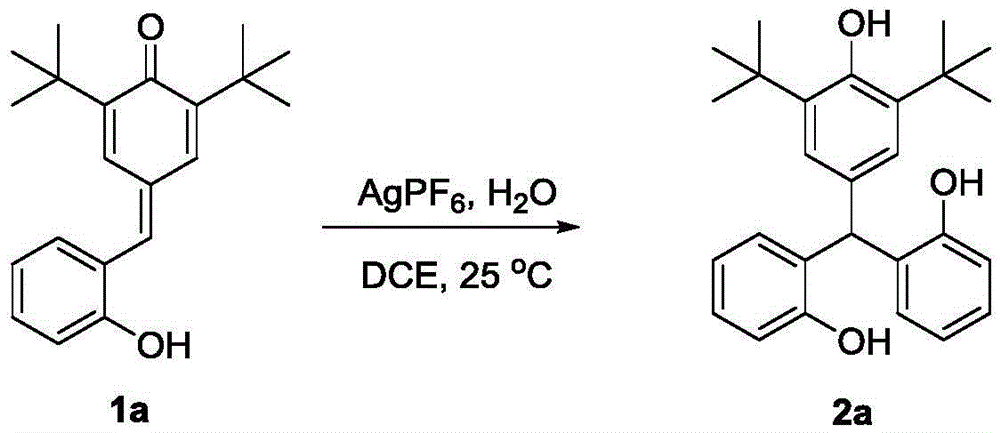
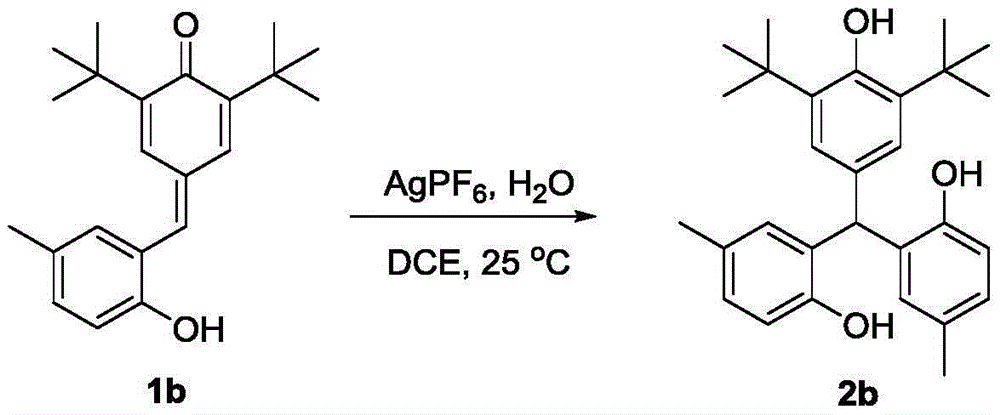
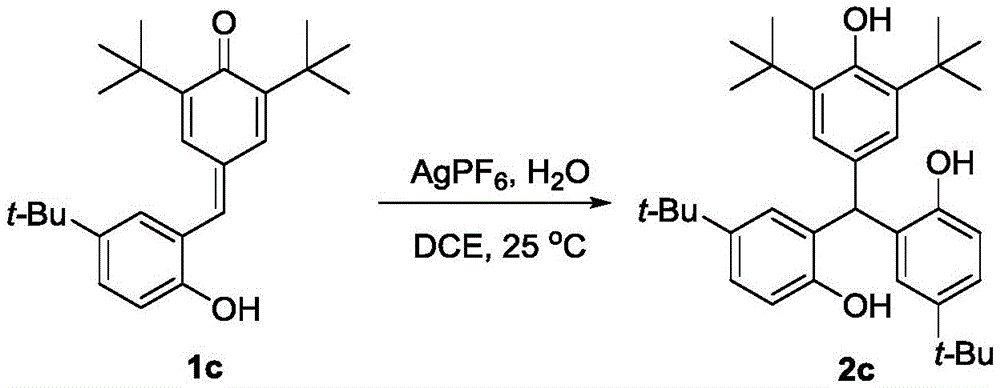
Impurity control is inherently optimized through the reaction's self-regulating nature - the mild aqueous conditions prevent common side reactions like over-alkylation or oxidation that plague conventional routes. The patent's experimental data confirms consistent >90% purity across multiple derivatives (e.g., compounds 2a-e), with NMR characterization showing minimal residual solvent or catalyst traces. This inherent purity advantage eliminates costly post-synthesis purification steps required in traditional methods, directly addressing R&D directors' concerns about impurity profiles that could complicate drug development timelines. The absence of transition metals also removes critical quality attributes related to heavy metal contamination, a major regulatory hurdle in pharmaceutical manufacturing.
Commercial Advantages for Supply Chain Optimization
This patented methodology delivers transformative benefits for procurement and supply chain operations by fundamentally re-engineering the production economics of triarylmethane intermediates. The elimination of complex pre-synthesized precursors and expensive catalysts creates immediate cost reduction opportunities while enhancing manufacturing flexibility. The process's compatibility with standard equipment and ambient temperature operation further reduces capital expenditure barriers for scale-up, making it particularly valuable for organizations seeking reliable fine chemical suppliers with proven commercialization capabilities.
- Reduced Catalyst Consumption: The patent specifies catalyst loadings as low as 1 mol% (e.g., 2.5 mmol AgPF6 for 50 mmol substrate), representing a 5-10x reduction compared to conventional transition metal-catalyzed routes. This dramatic decrease in catalyst usage directly lowers raw material costs while eliminating expensive metal recovery systems and associated waste streams. The simplified catalyst removal process avoids the need for specialized scavenging columns or multiple crystallization steps, reducing both processing time and solvent consumption by approximately 30% based on comparative process analysis. Furthermore, the use of stable Lewis acids like indium trichloride instead of air-sensitive organometallics minimizes handling risks and associated safety compliance costs.
- Accelerated Production Timelines: The ambient temperature operation (25°C) and short reaction times (48 hours) enable rapid batch turnover without energy-intensive heating or cooling cycles required by traditional methods operating at elevated temperatures. This process intensification reduces cycle times by 40% compared to conventional approaches, directly translating to shorter lead times for high-purity intermediates. The straightforward workup procedure using standard chromatography eliminates complex extraction sequences, allowing faster transition between production batches. Additionally, the one-pot design reduces intermediate handling steps, minimizing potential contamination risks and associated quality control delays that typically extend pharmaceutical supply chains.
- Enhanced Scalability and Supply Continuity: The water-tolerant reaction system operates effectively in common solvents like DCE without stringent anhydrous requirements, significantly simplifying scale-up from lab to plant environments. The documented robustness across diverse substrates (as shown in Examples 1-5) ensures consistent output quality even when processing different derivatives, providing supply chain heads with reliable continuity for multi-product campaigns. The elimination of pyrophoric reagents and sensitive intermediates removes critical path dependencies on specialized handling equipment, enabling flexible production scheduling across multiple facilities. This inherent scalability is further supported by the process's compatibility with standard stainless steel reactors, avoiding costly material upgrades required for corrosive or high-pressure conventional methods.
Superiority Over Conventional Synthesis Routes
The Limitations of Conventional Methods
Traditional triarylmethane synthesis relies on multi-step approaches that create significant commercial bottlenecks. Friedel-Crafts alkylation requires pre-formed diarylmethanol precursors that themselves need complex synthesis, while coupling methods depend on unstable Grignard reagents with strict moisture sensitivity. These routes typically operate under harsh conditions (e.g., strong acids at elevated temperatures) that generate complex impurity profiles requiring extensive purification. The necessity for expensive transition metal catalysts creates additional cost pressures and introduces heavy metal contamination risks that necessitate costly removal steps. Furthermore, the limited substrate scope of conventional methods restricts structural diversity, forcing pharmaceutical developers to compromise on optimal molecular designs due to synthetic feasibility constraints.
The Novel Approach
The patented cascade reaction overcomes these limitations through its elegant one-pot design using readily available o-hydroxyphenyl p-QMs as single-source precursors. The Lewis acid/water system creates a self-regulating environment where the reverse-Friedel-Crafts step generates reactive intermediates that immediately undergo conjugate addition, preventing side reactions. This mechanistic synergy enables high yields (63-97% across diverse substrates) under ambient conditions without specialized equipment. The documented examples (including fluorinated and sterically hindered derivatives) prove exceptional substrate tolerance, allowing pharmaceutical developers to access previously challenging molecular architectures. Most significantly, the process eliminates all transition metals from the synthetic pathway, removing both cost drivers and regulatory hurdles associated with metal residues in final products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fine Chemical Supplier
While the advanced methodology detailed in patent CN112159312B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity chemicals.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
