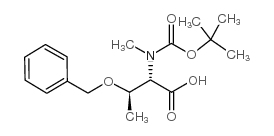
Boc-N-methyl-O-benzyl-L-threonine
- CAS No.64263-80-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity protected amino acid derivative designed for efficient peptide synthesis and pharmaceutical intermediate applications with reliable stability.
Request a Quote & Full COAProduct Technical Details
Product Overview
Boc-N-methyl-O-benzyl-L-threonine represents a highly specialized protected amino acid derivative essential for modern peptide chemistry and pharmaceutical development. This compound features a tert-butyloxycarbonyl (Boc) group on the nitrogen and a benzyl group on the side chain oxygen, providing robust protection during complex synthetic sequences. Our facility produces this chemical intermediate under strict quality control standards to ensure consistency for research and industrial scale applications.
As a key building block in the synthesis of bioactive peptides, this molecule offers superior stability against racemization during coupling reactions. The N-methylation enhances metabolic stability in final peptide therapeutics, making it a preferred choice for medicinal chemists designing novel drug candidates. We prioritize industrial purity levels to minimize downstream purification burdens.
Key Specifications
| Parameter | Value |
|---|---|
| CAS Number | 64263-80-5 |
| Molecular Formula | C17H25NO5 |
| Molecular Weight | 323.38 g/mol |
| Appearance | White powder |
| Assay | ≥98.0% |
| Storage Condition | Store at room temperature in a dry place |
Industrial Applications
This protected amino acid is primarily utilized in solid-phase peptide synthesis (SPPS) and solution-phase peptide construction. It serves as a critical intermediate for generating N-methylated peptide bonds, which are known to improve membrane permeability and enzymatic resistance in therapeutic peptides. Pharmaceutical companies rely on this building block for developing advanced neurological therapeutics and complex organic synthesis projects.
- Ideal for constructing metabolically stable peptide analogs
- Compatible with standard Boc and Fmoc synthesis strategies
- Ensures high coupling efficiency in sterically hindered sequences
- Supports large-scale manufacturing of peptide active pharmaceutical ingredients
Quality And Handling
Every batch undergoes rigorous analytical testing including HPLC and NMR verification to confirm structural integrity and purity levels. We maintain a closed-loop manufacturing process to prevent contamination and ensure product safety. For optimal stability, keep the container tightly closed in a cool, dry, and well-ventilated area away from strong oxidizing agents. Proper handling protocols should be followed according to standard chemical safety guidelines.
We offer flexible packaging options including 25kg drums tailored to customer requirements. Our global supply chain ensures timely delivery with complete documentation such as Certificates of Analysis (COA) and safety data sheets. Partner with us for reliable access to high-performance peptide building blocks that meet international regulatory standards.